Advanced Asymmetric Synthesis of Key Moxifloxacin Intermediate for Commercial Scale-Up
The pharmaceutical industry continuously seeks more efficient pathways for producing high-value antibiotics, and the synthesis of fourth-generation quinolones remains a critical area of focus. Patent CN114085219A discloses a groundbreaking method for synthesizing (1S,6R)-8-benzyl-7,9-dioxo-2,8-diazabicyclo[4.3.0]nonane, a pivotal chiral intermediate in the manufacturing of Moxifloxacin. This novel approach addresses long-standing challenges in stereochemical control and process efficiency by leveraging advanced asymmetric catalysis. Unlike traditional methods that rely on the separation of racemic mixtures, this technology employs a strategic four-step sequence involving partial oxidation, protective group manipulation, and highly selective chiral hydrogenation. The result is a process capable of achieving an enantiomeric excess (ee) of up to 99.5%, setting a new benchmark for purity in antibiotic intermediate production. For R&D directors and procurement specialists, understanding the mechanistic advantages of this route is essential for evaluating next-generation supply chain partners who can deliver complex chiral building blocks with superior consistency and reduced environmental impact.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of (1S,6R)-8-benzyl-7,9-dioxo-2,8-diazabicyclo[4.3.0]nonane has relied heavily on classical resolution techniques, as exemplified by prior art such as WO9415938. These conventional pathways typically involve the synthesis of a racemic mixture followed by the separation of enantiomers using chiral amines or acids. 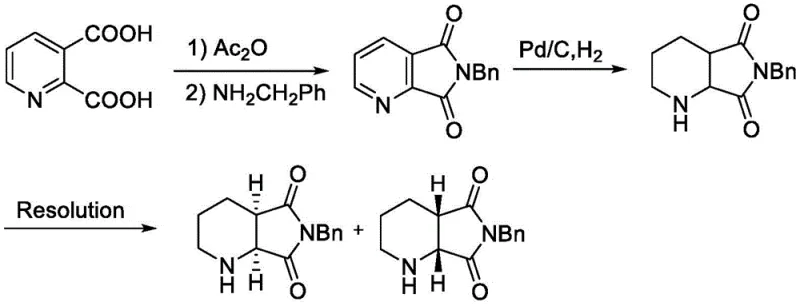 The fundamental flaw in this approach is the inherent 50% theoretical yield limit, as half of the synthesized material constitutes the unwanted enantiomer which is often discarded or requires complex recycling procedures. Furthermore, resolution processes frequently demand multiple crystallization steps to achieve acceptable optical purity, leading to significant solvent consumption, extended processing times, and increased waste generation. The reliance on stoichiometric amounts of resolving agents also drives up raw material costs, making the final API expensive to produce. For large-scale manufacturers, these inefficiencies translate into higher carbon footprints and supply chain vulnerabilities, particularly when the demand for broad-spectrum antibiotics like Moxifloxacin fluctuates globally.
The fundamental flaw in this approach is the inherent 50% theoretical yield limit, as half of the synthesized material constitutes the unwanted enantiomer which is often discarded or requires complex recycling procedures. Furthermore, resolution processes frequently demand multiple crystallization steps to achieve acceptable optical purity, leading to significant solvent consumption, extended processing times, and increased waste generation. The reliance on stoichiometric amounts of resolving agents also drives up raw material costs, making the final API expensive to produce. For large-scale manufacturers, these inefficiencies translate into higher carbon footprints and supply chain vulnerabilities, particularly when the demand for broad-spectrum antibiotics like Moxifloxacin fluctuates globally.
The Novel Approach
In stark contrast, the methodology described in CN114085219A introduces a streamlined asymmetric synthesis that bypasses the need for resolution entirely. The process begins with the partial oxidation of N-benzyl piperidine dicarboximide to generate a reactive intra-ring double bond, followed by the strategic introduction of a tert-butoxycarbonyl (Boc) group. 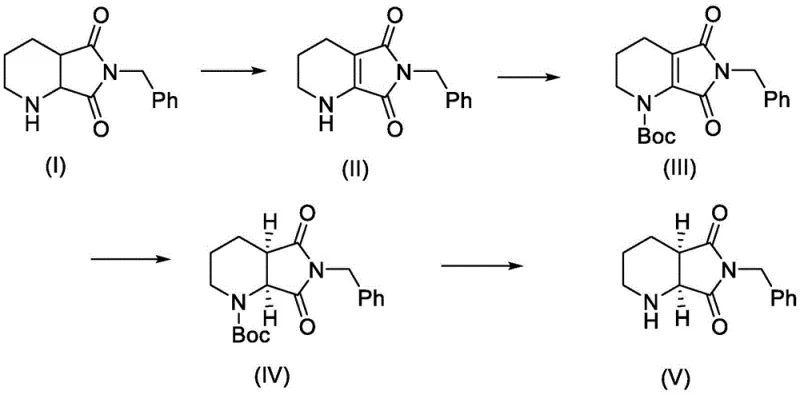 This activation step is crucial as it prepares the substrate for the subsequent key transformation: asymmetric catalytic hydrogenation. By utilizing specialized chiral Iridium catalysts, the process directly installs the required stereocenters with exceptional precision, theoretically allowing for 100% conversion of the starting material into the desired chiral product. This shift from separation-based purification to construction-based synthesis represents a paradigm shift in process chemistry, offering substantial reductions in raw material usage and waste. The mild reaction conditions and high selectivity ensure that the process is not only chemically elegant but also commercially viable for multi-ton production scales, addressing the critical need for cost reduction in pharmaceutical intermediate manufacturing.
This activation step is crucial as it prepares the substrate for the subsequent key transformation: asymmetric catalytic hydrogenation. By utilizing specialized chiral Iridium catalysts, the process directly installs the required stereocenters with exceptional precision, theoretically allowing for 100% conversion of the starting material into the desired chiral product. This shift from separation-based purification to construction-based synthesis represents a paradigm shift in process chemistry, offering substantial reductions in raw material usage and waste. The mild reaction conditions and high selectivity ensure that the process is not only chemically elegant but also commercially viable for multi-ton production scales, addressing the critical need for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Chiral Iridium-Catalyzed Asymmetric Hydrogenation
The core innovation of this patent lies in the application of specific chiral Iridium complexes to hydrogenate a sterically hindered intra-ring double bond. The catalysts employed, designated as Compounds A, B, and C, feature sophisticated P-N ligand systems that create a highly defined chiral environment around the metal center. 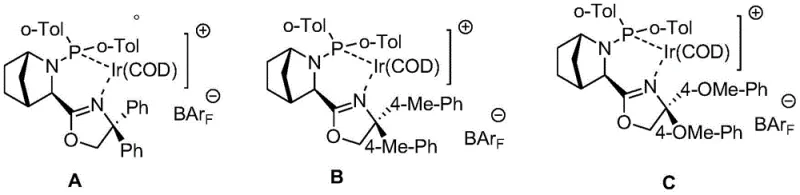 These ligands are designed to interact with the substrate through a combination of steric bulk and electronic coordination, guiding the approach of hydrogen to specific faces of the double bond. The patent highlights that while such catalysts were previously known for linear alkenes, their successful application to this rigid bicyclic system is a significant technical achievement. The presence of the electron-withdrawing Boc group on the adjacent nitrogen atom plays a synergistic role by lowering the electron density of the alkene, thereby facilitating coordination with the cationic Iridium species. This electronic activation overcomes the kinetic barriers associated with the steric congestion of the bicyclic framework, enabling reaction rates and selectivities that were previously unattainable with standard heterogeneous catalysts like Pd/C.
These ligands are designed to interact with the substrate through a combination of steric bulk and electronic coordination, guiding the approach of hydrogen to specific faces of the double bond. The patent highlights that while such catalysts were previously known for linear alkenes, their successful application to this rigid bicyclic system is a significant technical achievement. The presence of the electron-withdrawing Boc group on the adjacent nitrogen atom plays a synergistic role by lowering the electron density of the alkene, thereby facilitating coordination with the cationic Iridium species. This electronic activation overcomes the kinetic barriers associated with the steric congestion of the bicyclic framework, enabling reaction rates and selectivities that were previously unattainable with standard heterogeneous catalysts like Pd/C.
From an impurity control perspective, this mechanism offers distinct advantages over non-catalytic methods. The high specificity of the chiral catalyst minimizes the formation of diastereomeric byproducts, which are often difficult to separate in downstream processing. The patent data indicates that the asymmetric conversion rate exceeds 96%, with ee values reaching as high as 99.5% when using optimized catalyst variants. This level of stereocontrol simplifies the purification workflow, often eliminating the need for preparative chiral chromatography which is prohibitively expensive at scale. Furthermore, the mild conditions (20-80°C and 20-80 kg pressure) prevent thermal degradation of the sensitive lactam rings, ensuring a clean impurity profile. For quality assurance teams, this translates to a more robust process with fewer critical quality attributes (CQAs) to monitor, reducing the risk of batch failures and ensuring consistent supply of high-purity intermediates.
How to Synthesize (1S,6R)-8-benzyl-7,9-dioxo-2,8-diazabicyclo[4.3.0]nonane Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for replicating this high-efficiency route in a pilot or production setting. The process is divided into four distinct operational stages, each optimized for yield and safety. The initial oxidation step utilizes common oxidants such as potassium permanganate or manganese dioxide under controlled temperatures to generate the unsaturated intermediate. Following this, the Boc protection is performed using standard reagents like di-tert-butyl dicarbonate in the presence of a base, a routine operation in most fine chemical facilities.
- Partial oxidation of N-benzyl piperidine dicarboximide using oxidants like KMnO4 or MnO2 to form the intra-ring double bond.
- Introduction of the tert-butoxycarbonyl (Boc) group to the ortho-NH position to activate the double bond for hydrogenation.
- Asymmetric catalytic hydrogenation using chiral Iridium catalysts (A, B, or C) under 20-80 kg pressure to achieve high enantioselectivity.
- Acid-catalyzed removal of the Boc protecting group to yield the final high-purity bicyclic intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this asymmetric synthesis route offers compelling economic and logistical benefits that extend beyond simple yield improvements. The transition from a resolution-based process to a direct asymmetric synthesis fundamentally alters the cost structure of the intermediate. By eliminating the discard of the unwanted enantiomer, the effective utilization of raw materials is nearly doubled, leading to substantial cost savings in starting material procurement. Additionally, the reduction in unit operations—specifically the removal of multiple crystallization and recycling steps required for resolution—lowers energy consumption and labor costs. The use of readily available reagents and solvents further enhances the economic viability, ensuring that the supply of this critical Moxifloxacin precursor remains stable even during raw material market fluctuations.
- Cost Reduction in Manufacturing: The elimination of the resolution step removes the inherent 50% yield penalty associated with separating racemates, effectively doubling the output per kilogram of starting material without increasing input costs. This dramatic improvement in atom economy directly translates to a lower cost of goods sold (COGS) for the final API. Furthermore, the high selectivity of the chiral catalyst reduces the burden on downstream purification, minimizing solvent usage and waste disposal fees which are significant cost drivers in pharmaceutical manufacturing.
- Enhanced Supply Chain Reliability: The reliance on robust, well-defined chemical steps rather than unpredictable physical separations enhances process reliability. The catalysts described are stable and effective under moderate pressures, reducing the risk of equipment failure or batch variability. This consistency allows for more accurate production planning and inventory management, ensuring that downstream API manufacturers receive their intermediates on schedule. The simplicity of the route also means that technology transfer to multiple manufacturing sites is feasible, diversifying supply sources and mitigating geopolitical or logistical risks.
- Scalability and Environmental Compliance: The process operates under mild conditions and avoids the use of hazardous heavy metals often found in stoichiometric chiral auxiliaries. The catalytic nature of the hydrogenation step means that only small amounts of the expensive Iridium complex are needed, which can potentially be recovered and recycled. This aligns with green chemistry principles, reducing the environmental footprint of the manufacturing process. For companies facing increasing regulatory pressure to reduce waste and emissions, adopting this cleaner synthesis route provides a strategic advantage in maintaining compliance and corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making.
Q: What is the primary advantage of this new synthesis route over traditional resolution methods?
A: The primary advantage is the elimination of the 50% theoretical yield loss inherent in racemic resolution. By utilizing asymmetric hydrogenation with chiral Iridium catalysts, the process achieves an enantiomeric excess (ee) of up to 99.5% and significantly higher overall conversion rates compared to the separation of diastereomeric salts.
Q: Why is the introduction of the Boc group critical in this reaction sequence?
A: The Boc group serves as an electron-withdrawing group that reduces the electron cloud density of the intra-ring double bond. This activation is essential for overcoming steric hindrance during the asymmetric hydrogenation step, thereby enabling the chiral catalyst to effectively induce stereochemistry and improve both yield and selectivity.
Q: What are the typical reaction conditions for the key hydrogenation step?
A: The asymmetric hydrogenation is typically conducted at mild temperatures ranging from 20°C to 80°C under a hydrogen pressure of 20-80 kg. The reaction utilizes specialized chiral Iridium complexes (Compounds A, B, or C) in solvents such as DCM, methanol, or THF, ensuring robust performance suitable for industrial scaling.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (1S,6R)-8-benzyl-7,9-dioxo-2,8-diazabicyclo[4.3.0]nonane Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global supply of life-saving antibiotics. Our technical team has extensively analyzed the pathway described in CN114085219A and possesses the expertise to scale this complex asymmetric hydrogenation from laboratory benchtop to commercial production. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. Our state-of-the-art facilities are equipped with high-pressure hydrogenation reactors and rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to guarantee ee values exceeding 99%.
We invite pharmaceutical partners to collaborate with us to leverage this advanced synthesis technology for their Moxifloxacin projects. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential efficiencies of switching to this asymmetric route. We encourage you to contact us today to discuss your specific requirements,索取 specific COA data, and review our comprehensive route feasibility assessments. Let us help you secure a sustainable and cost-effective supply chain for your next-generation antibiotic formulations.
