Scalable Production of High-Purity Condensed Ring Indole Intermediates via Safe Chloride Salt Catalysis
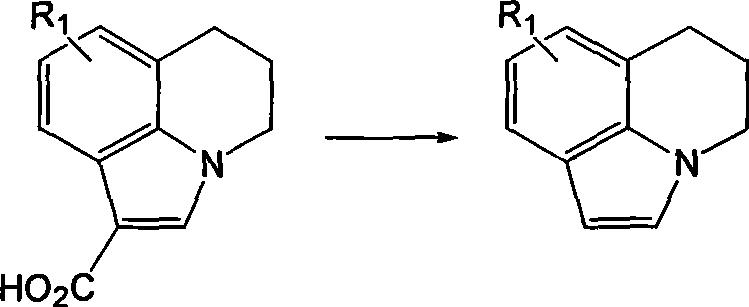
The pharmaceutical industry continuously seeks robust synthetic routes for heterocyclic intermediates, particularly those serving as key scaffolds for receptor antagonists. Patent CN101302216B introduces a significant advancement in the preparation of condensed ring indole compounds, which serve as vital intermediates for 5-HT3 receptor antagonists used in treating nausea and vomiting. This technology addresses long-standing issues in the field by replacing hazardous heavy metal reagents with benign chloride salts, offering a pathway that is not only chemically efficient but also environmentally sustainable. The core innovation lies in a thermal decarboxylation process that operates under relatively mild conditions, utilizing common inorganic salts to drive the reaction to completion with exceptional purity profiles. For R&D directors and process chemists, this represents a pivotal shift away from the toxic legacy of chromium and tin-based methodologies toward a greener, more manageable chemical process.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of these fused ring systems has been plagued by significant safety and environmental drawbacks associated with prior art methodologies. One prominent method reported by von Wijngaarden utilized quinoline as a solvent alongside copper oxide and chromium oxide reagents for decarboxylation. This approach presents severe liabilities, including the use of highly toxic solvents and the generation of hazardous chromium waste, which complicates downstream processing and disposal. The removal of trace chromium from the final product is notoriously difficult and costly, posing a major risk for pharmaceutical grade materials where heavy metal limits are strictly regulated. Furthermore, alternative radical cyclization methods employing tributyltin hydride and AIBN suffer from similar toxicity issues related to organotin compounds, alongside generally lower yields that render them economically unviable for large-scale operations. These conventional routes create substantial bottlenecks in supply chains due to the rigorous purification requirements needed to meet safety standards.
The Novel Approach
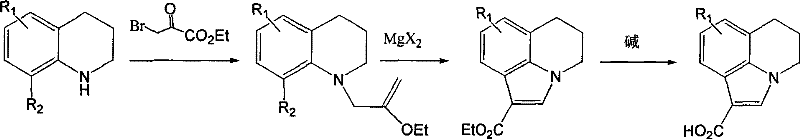
In stark contrast, the novel approach detailed in the patent utilizes a simple yet effective strategy involving chloride salts to facilitate the decarboxylation of fused ring indole carboxylic acids. By reacting the carboxylic acid precursor with a molar excess of salts such as sodium chloride, potassium chloride, or magnesium chloride in a solvent like toluene or DMSO, the reaction proceeds smoothly at temperatures between 80°C and 150°C. This method completely eliminates the need for toxic transition metal catalysts or hazardous radical initiators, thereby simplifying the reaction profile and reducing the environmental footprint. The use of inexpensive, commercially available inorganic salts drastically lowers the raw material costs while ensuring that the final product is free from problematic heavy metal residues. This streamlined process allows for a direct isolation of the product through simple aqueous workup and filtration, bypassing the complex purification steps required by older technologies.
Mechanistic Insights into Chloride Salt-Mediated Thermal Decarboxylation
The mechanistic underpinning of this transformation relies on the ability of chloride ions to stabilize transition states or intermediates during the thermal loss of carbon dioxide from the carboxylic acid moiety. While the exact mechanistic pathway may vary depending on the specific salt and solvent system employed, the presence of the halide salt appears to lower the activation energy required for decarboxylation compared to thermal treatment alone. The reaction likely proceeds through a concerted mechanism where the chloride salt interacts with the carboxylate group, facilitating the cleavage of the C-C bond and the subsequent release of CO2. This interaction is crucial for maintaining the integrity of the sensitive indole ring system under the elevated temperatures required for the reaction. The choice of solvent plays a complementary role, with polar aprotic solvents like DMSO or aromatic hydrocarbons like toluene providing the necessary medium to dissolve both the organic substrate and the inorganic salt, ensuring homogeneous reaction conditions that drive high conversion rates.
From an impurity control perspective, this mechanism offers distinct advantages by avoiding the formation of organometallic byproducts that are characteristic of tin or copper-mediated reactions. In traditional methods, side reactions often lead to complex mixtures containing metal-complexed species that are difficult to separate from the desired API intermediate. The chloride salt method minimizes these side pathways, resulting in a cleaner crude reaction mixture that requires less intensive purification. The high purity levels reported, often exceeding 99% area% by HPLC, suggest that the reaction is highly selective for the decarboxylation event without inducing degradation of the fused ring scaffold. This selectivity is paramount for pharmaceutical manufacturing, where the presence of genotoxic impurities or heavy metals can halt a drug development program. The robustness of this chemical transformation ensures consistent quality across different batches, a critical factor for regulatory compliance.
How to Synthesize Condensed Ring Indole Efficiently
To implement this synthesis effectively, process chemists should focus on optimizing the molar ratio of the chloride salt to the carboxylic acid substrate, with the patent suggesting a range of 1 to 10 equivalents for optimal results. The selection of the solvent is also critical, with options ranging from benzene derivatives to sulfoxides, allowing for flexibility based on boiling point requirements and solubility profiles. Detailed standard operating procedures for scaling this reaction from gram to kilogram scale involve precise temperature control and monitoring of reaction progress via HPLC to prevent over-heating or decomposition. The standardized synthesis steps outlined below provide a framework for reproducing the high yields and purity described in the patent examples.
- Prepare the fused ring indole carboxylic acid starting material (Formula II) according to established literature methods.
- Mix the carboxylic acid with a chloride salt (such as NaCl, KCl, or MgCl2) in a molar ratio of 1: 1 to 1:10 within a suitable organic solvent like toluene or DMSO.
- Heat the reaction mixture to a temperature between 80°C and 150°C under reflux conditions until the decarboxylation is complete, followed by aqueous workup and filtration.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this chloride salt-mediated process offers transformative benefits in terms of cost structure and operational reliability. The shift away from specialized, hazardous reagents to commodity chemicals fundamentally alters the cost dynamics of producing these valuable intermediates. By eliminating the need for expensive and regulated substances like tributyltin hydride or chromium oxides, manufacturers can achieve significant cost reductions in raw material procurement. Furthermore, the simplified workup procedure reduces the consumption of solvents and adsorbents typically required for metal scavenging, leading to additional savings in operational expenditures. This economic efficiency is compounded by the reduced liability associated with handling toxic materials, lowering insurance and waste disposal costs for the manufacturing facility.
- Cost Reduction in Manufacturing: The replacement of high-cost catalytic systems with ubiquitous inorganic salts like sodium chloride or magnesium chloride results in a drastic reduction in reagent costs. Unlike precious metal catalysts or specialized organometallic reagents that require strict inventory control and expensive disposal protocols, chloride salts are inexpensive and readily available in bulk quantities globally. This substitution removes a major cost driver from the bill of materials, allowing for more competitive pricing of the final intermediate. Additionally, the elimination of complex metal removal steps reduces the demand for specialized purification resins and silica gel, further driving down the overall cost of goods sold.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals for the key transformation step significantly de-risks the supply chain against shortages of specialized reagents. Global supply chains for basic inorganic salts are robust and stable, ensuring continuous production capability even during market fluctuations that might affect niche chemical suppliers. This reliability is crucial for maintaining uninterrupted supply of critical pharmaceutical intermediates to downstream customers. Moreover, the use of non-hazardous materials simplifies logistics and transportation requirements, reducing lead times and administrative burdens associated with shipping dangerous goods.
- Scalability and Environmental Compliance: The simplicity of the reaction conditions and the absence of toxic heavy metals make this process inherently scalable for industrial production. Facilities can easily adapt existing reactors for this chemistry without needing specialized containment systems for chromium or tin waste. This ease of scale-up accelerates the timeline from pilot plant to commercial manufacturing, enabling faster time-to-market for new drug candidates. From an environmental standpoint, the process aligns with green chemistry principles by reducing hazardous waste generation, thereby ensuring compliance with increasingly stringent environmental regulations and enhancing the corporate sustainability profile.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this decarboxylation technology in a production environment. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a reliable basis for process evaluation. Understanding these specifics helps stakeholders assess the feasibility of integrating this method into their existing manufacturing portfolios.
Q: What are the primary advantages of using chloride salts over traditional decarboxylation reagents?
A: Traditional methods often rely on toxic copper/chromium oxides or tributyltin hydride, which create severe environmental hazards and difficult purification challenges. The chloride salt method described in CN101302216B utilizes non-toxic, inexpensive inorganic salts that eliminate heavy metal contamination risks and simplify the post-reaction workup significantly.
Q: What purity levels can be achieved with this decarboxylation process?
A: The patent data indicates that this method consistently produces condensed ring indole compounds with high purity, typically exceeding 99.0% area% as measured by HPLC. This high purity is critical for pharmaceutical applications where impurity profiles must be strictly controlled.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is explicitly designed for industrial scalability. It uses commercially available solvents and reagents, operates at moderate temperatures (80-150°C), and avoids complex purification steps like column chromatography, making it highly viable for metric-ton scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Condensed Ring Indole Supplier
NINGBO INNO PHARMCHEM stands at the forefront of implementing advanced synthetic methodologies like the chloride salt decarboxylation process to deliver superior pharmaceutical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch meets the highest standards required by global regulatory bodies. Our commitment to quality assurance means that clients receive materials with consistent impurity profiles, essential for the successful development of 5-HT3 receptor antagonists.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can optimize your supply chain and reduce overall manufacturing costs. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the economic benefits of switching to this greener, more efficient process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project requirements, ensuring a strategic advantage in the competitive pharmaceutical landscape.
