Advanced Enzymatic Resolution for High-Purity Stereoisomer Carboxylates in Pharmaceutical Manufacturing
Advanced Enzymatic Resolution for High-Purity Stereoisomer Carboxylates in Pharmaceutical Manufacturing
The pharmaceutical and fine chemical industries constantly face the challenge of producing chiral intermediates with exceptional optical purity, a requirement that is critical for the efficacy and safety of active pharmaceutical ingredients (APIs). Patent CN1266904A introduces a groundbreaking process for the preparation of stereoisomer carboxylates, addressing the limitations of conventional resolution methods by leveraging the specificity of carboxylic ester hydrolases. This technology enables the synthesis of compounds where both the acid and alcohol components possess at least one chiral center, achieving high diastereomeric and enantiomeric excess simultaneously. By utilizing racemic starting materials and specific biocatalysts, this method offers a robust pathway for generating complex chiral structures that are essential precursors for medicines and crop protection agents. For R&D directors and procurement specialists, understanding this enzymatic approach provides a strategic advantage in securing reliable pharmaceutical intermediate suppliers who can deliver high-quality materials with reduced environmental impact.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for preparing optically pure carboxylic esters often rely on chemical resolution or non-enzymatic kinetic resolution, which frequently suffer from significant drawbacks regarding efficiency and selectivity. Prior art techniques, such as those involving lipase-catalyzed reactions with standard alkyl esters, often result in low conversion rates and poor enantio-selectivity, with E values ranging merely from 1.3 to 35.6 in many documented cases. Furthermore, these conventional processes typically require prolonged reaction times to reach equilibrium, leading to low space-time yields and increased operational costs. The inability to effectively control the stereochemistry of both the acid and alcohol moieties simultaneously often necessitates complex downstream purification steps, such as repeated crystallizations or expensive chromatographic separations, which drastically reduce the overall chemical yield. Additionally, the use of non-activated esters can lead to reversible reactions that limit the maximum theoretical conversion, making it difficult to achieve the high purity standards required for modern drug development without substantial material loss.
The Novel Approach
The innovative method described in the patent overcomes these historical barriers by employing activated racemic carboxylic acid esters, such as vinyl esters or oxime esters, in conjunction with highly selective carboxylic ester hydrolases. This approach facilitates an irreversible transesterification reaction, as the leaving group (e.g., vinyl alcohol) tautomerizes into a stable carbonyl compound like acetaldehyde, thereby driving the equilibrium completely towards the desired product. By reacting these activated esters with racemic alcohols in the presence of enzymes like Candida antarctica lipase B, the process achieves a dual kinetic resolution that enriches specific stereoisomers with remarkable efficiency. This strategy allows for the production of stereoisomer carboxylates with diastereomeric excess values exceeding 50% de and enantio-selectivity (E) values greater than 100, all within significantly shortened reaction times. The versatility of this method extends to a wide range of substrates, accommodating various aryl, alkyl, and heteroaryl substituents, making it a universally applicable solution for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Lipase-Catalyzed Dual Kinetic Resolution
The core of this technological advancement lies in the precise mechanistic action of carboxylic ester hydrolases, which act as chiral selectors to differentiate between the enantiomers of both the acid and alcohol components. The reaction proceeds through a nucleophilic attack by the serine residue in the enzyme's active site on the carbonyl carbon of the activated ester, forming an acyl-enzyme intermediate. This step is highly stereoselective, preferentially accepting one enantiomer of the racemic acid ester while rejecting the other. Subsequently, the accepted enantiomer of the racemic alcohol attacks this intermediate to form the final ester product, with the enzyme again exerting stereocontrol over the alcohol moiety. This dual recognition capability ensures that only specific stereoisomeric combinations, such as the (R,R) or (S,S) configurations, are formed in excess, while the unwanted isomers remain largely unreacted in the mixture. The use of activated esters is crucial here, as it prevents the hydrolysis of the product back to the starting materials, locking in the stereochemical integrity achieved during the catalytic cycle.
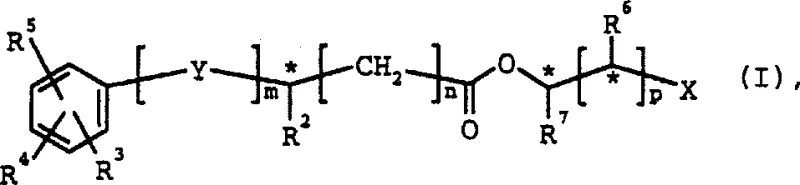
To visualize the structural complexity managed by this process, one must consider the general formula of the resulting stereoisomer carboxylates, which can contain multiple chiral centers denoted by asterisks in the molecular backbone. The patent specifies that variables such as n, m, and p can independently be 0 or 1, allowing for significant structural diversity in the final products. Substituents R3, R4, and R5 on the aromatic ring can vary widely, including halogens, nitro groups, or alkoxy chains, without compromising the enzyme's ability to discriminate between stereoisomers. This structural tolerance is vital for medicinal chemists who need to introduce specific functional groups to optimize the biological activity of a drug candidate. The mechanism ensures that regardless of these substitutions, the enzymatic pocket maintains its fidelity, delivering high-purity products that meet stringent regulatory specifications for impurity profiles. Such robustness in the face of structural variation is a key indicator of a mature and scalable synthetic platform suitable for industrial application.
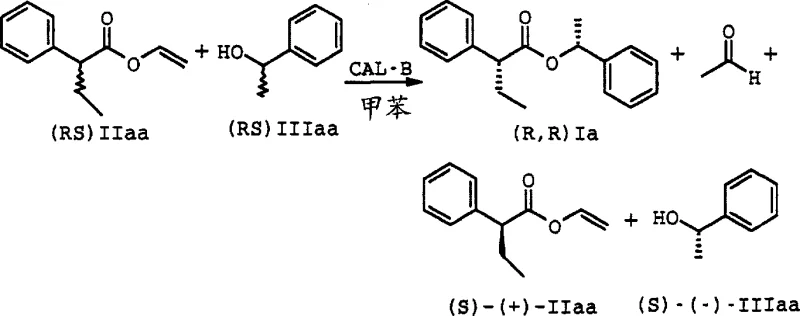
A concrete example of this mechanism in action is illustrated by the reaction between (RS)-2-phenylbutyric acid vinyl acetate and (RS)-1-phenylethyl alcohol, catalyzed by CAL-B lipase. In this specific transformation, the enzyme selectively converts the (R)-enantiomer of the acid ester and the (R)-enantiomer of the alcohol into the (R,R)-ester product, while leaving the (S)-enantiomers largely untouched. The reaction scheme demonstrates how the vinyl group serves as an excellent leaving group, facilitating the forward reaction and preventing reversibility. Analytical data from the patent confirms that this specific reaction yields the desired (R,R) isomer with greater than 98% ee for the alcohol moiety and 56% de overall after just 35 hours. This level of precision underscores the power of biocatalysis in resolving complex mixtures where chemical methods would fail to distinguish between such closely related stereoisomers, providing a clear pathway for obtaining single-isomer drugs.
How to Synthesize Stereoisomer Carboxylates Efficiently
Implementing this synthesis route requires careful attention to reaction conditions to maximize the benefits of the enzymatic catalyst. The process begins with the preparation of the activated racemic carboxylic acid ester, typically a vinyl ester, which is then mixed with a racemic alcohol in an aprotic organic solvent such as toluene or hexane. The choice of solvent is critical, as it influences the solubility of the substrates and the stability of the enzyme, with non-polar solvents often favoring higher enantio-selectivity. Once the mixture is prepared, a fixed form of carboxylic ester hydrolase, such as immobilized Candida antarctica lipase B, is added to the reactor. The reaction is typically conducted at temperatures ranging from 0°C to 90°C, with a preferred range of 10°C to 80°C to balance reaction rate and enzyme stability. Monitoring the conversion is essential, with the reaction ideally stopped at approximately 50% conversion to ensure the highest optical purity of the product, after which the enzyme is removed by filtration and the product is purified via standard techniques like crystallization.
- Prepare the reaction mixture by dissolving equimolar amounts of racemic activated carboxylic acid ester (e.g., vinyl ester) and racemic alcohol in an organic solvent like toluene.
- Add a carboxylic ester hydrolase, preferably Candida antarctica lipase B (CAL-B), to the solution and maintain the temperature between 0°C and 90°C.
- Monitor the reaction until approximately 50% conversion is reached, then separate the enzyme and purify the resulting stereoisomer carboxylate via crystallization or chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this enzymatic process translates into tangible strategic benefits that extend beyond mere technical feasibility. The ability to start from readily available racemic materials significantly reduces the cost of goods sold (COGS), as racemates are generally far cheaper and easier to source than their optically pure counterparts. This shift in raw material strategy mitigates supply chain risks associated with the scarcity of chiral pool resources, ensuring a more stable and continuous flow of intermediates for production lines. Furthermore, the high selectivity of the enzyme minimizes the formation of difficult-to-remove impurities, simplifying the purification workflow and reducing the consumption of solvents and silica gel typically required for extensive chromatographic separations. These operational efficiencies contribute to a greener manufacturing profile, aligning with increasingly strict environmental regulations and corporate sustainability goals without sacrificing output quality or speed.
- Cost Reduction in Manufacturing: The elimination of expensive chiral reagents and the reduction in downstream processing steps lead to substantial cost savings in API manufacturing. By utilizing robust biocatalysts that can often be recovered and reused, the process lowers the catalyst cost per kilogram of product, while the high conversion efficiency ensures that raw material waste is minimized. The simplified purification protocol, often requiring only a single recrystallization step to achieve high purity, further decreases energy consumption and labor costs associated with complex isolation procedures. This economic model supports the production of high-value intermediates at a price point that remains competitive in the global market.
- Enhanced Supply Chain Reliability: Relying on stable, commercially available enzymes and bulk chemical feedstocks enhances the resilience of the supply chain against disruptions. Unlike processes dependent on precious metal catalysts or exotic chiral auxiliaries, this biocatalytic method uses materials that are widely sourced and less susceptible to geopolitical supply constraints. The scalability of the reaction, which can be performed in both batch and continuous flow modes, allows manufacturers to flexibly adjust production volumes to meet fluctuating market demands without the need for extensive retooling. This adaptability ensures that delivery timelines are met consistently, fostering stronger partnerships between suppliers and pharmaceutical developers.
- Scalability and Environmental Compliance: The mild reaction conditions and aqueous-compatible nature of the enzymes facilitate easier scale-up from laboratory to commercial production scales. The process generates fewer hazardous byproducts compared to traditional chemical resolutions, reducing the burden on waste treatment facilities and lowering the overall environmental footprint. Compliance with green chemistry principles is inherently built into the methodology, as the use of renewable biocatalysts and the avoidance of toxic heavy metals align with modern regulatory expectations for sustainable chemical manufacturing. This forward-looking approach future-proofs the production facility against tightening environmental legislation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this stereoselective synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on the scope and limitations of the method. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this process into their existing manufacturing workflows. The responses cover aspects ranging from enzyme selection to substrate compatibility, ensuring a comprehensive overview of the technology's capabilities.
Q: What represents the primary advantage of this enzymatic process over traditional chemical resolution?
A: The primary advantage is the ability to achieve high diastereomeric excess (de) and enantiomeric excess (ee) simultaneously through dual kinetic resolution. Unlike traditional methods that often suffer from low selectivity or require harsh conditions, this biocatalytic approach utilizes specific lipases to distinguish between stereoisomers efficiently, resulting in superior purity with shorter reaction times and milder conditions.
Q: Which types of enzymes are most suitable for this stereoselective transesterification?
A: Carboxylic ester hydrolases, specifically lipases (EC 3.1.1.3) and esterases (EC 3.1.1.1), are most suitable. The patent highlights Candida antarctica lipase B (CAL-B) as particularly effective due to its high enantio-selectivity and stability in organic solvents. Other suitable sources include lipases from Pseudomonas, Bacillus, and Mucor species, which can be used in free or immobilized forms.
Q: How does the use of activated esters improve the reaction efficiency?
A: Using activated racemic carboxylic acid esters, such as vinyl esters or oxime esters, drives the reaction equilibrium towards product formation. The leaving group (e.g., vinyl alcohol tautomerizing to acetaldehyde) is removed from the equilibrium, preventing the reverse reaction. This irreversibility allows for higher conversion rates and better control over the stereochemical outcome compared to using standard alkyl esters.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Stereoisomer Carboxylate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity chiral intermediates in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to full-scale manufacturing is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs equipped with advanced chiral HPLC and GC capabilities to verify the enantiomeric and diastereomeric excess of every batch we produce. Our commitment to quality assurance means that clients can rely on us for consistent supply of materials that meet the exacting standards required for clinical and commercial drug applications.
We invite potential partners to engage with our technical procurement team to discuss how this enzymatic resolution technology can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how optimizing your synthetic route with our expertise can reduce overall production costs. We encourage you to contact us to obtain specific COA data and route feasibility assessments for your target molecules, allowing us to demonstrate our capability as a trusted partner in your supply chain. Let us collaborate to bring your high-value pharmaceutical projects to market faster and more economically.
