Advanced Synthesis Of Organophosphorus Fluorine Compounds For Commercial Scale-Up And Ligand Design
Advanced Synthesis Of Organophosphorus Fluorine Compounds For Commercial Scale-Up And Ligand Design
The landscape of organophosphorus chemistry is undergoing a significant transformation driven by the demand for specialized fluorinated intermediates in pharmaceutical and agrochemical sectors. A pivotal development in this field is documented in patent CN115403621A, which discloses a novel and efficient synthetic methodology for generating organophosphorus fluorine compounds. This technology addresses long-standing challenges in the deoxydifluoromethylation of phosphine oxides, a reaction class that has historically suffered from limited substrate scope and harsh reaction conditions. By leveraging a transition-metal-free catalytic system utilizing trifluoromethyltrimethylsilane (TMSCF3), this innovation offers a robust pathway to access valuable trivalent phosphorus species. For R&D directors and procurement strategists, understanding the nuances of this patent is essential, as it represents a potential paradigm shift in how high-value phosphine ligands and fine chemical intermediates are manufactured. The following analysis dissects the technical merits and commercial implications of this breakthrough, providing a comprehensive roadmap for integrating these advanced synthetic capabilities into global supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the fluorination of phosphine oxide compounds has been fraught with significant technical hurdles that impede large-scale commercialization. Conventional approaches often rely on highly reactive and hazardous fluorinating agents that pose severe safety risks in an industrial setting, requiring specialized containment infrastructure and rigorous safety protocols. Furthermore, existing methodologies such as silicon trifluoromethylation or deoxytrifluoromethylation frequently exhibit poor substrate adaptability, failing to accommodate sterically hindered or electronically diverse aromatic systems. This limitation restricts the chemical space available to medicinal chemists designing next-generation catalysts. Additionally, many traditional routes suffer from low yields and generate substantial amounts of toxic waste, complicating downstream purification and increasing the overall cost of goods sold. The reliance on transition metal catalysts in some legacy processes introduces another layer of complexity, necessitating expensive and time-consuming metal scavenging steps to meet the stringent purity requirements of the pharmaceutical industry.
The Novel Approach
In stark contrast to these legacy issues, the method described in CN115403621A introduces a streamlined and economically viable solution for the deoxydifluoromethylation of diphenyl(dialkyl)phosphine oxides. The core of this innovation lies in the strategic combination of TMSCF3 as the fluorinating source with a simple yet effective additive system comprising potassium carbonate, sodium iodide, and water in a tetrahydrofuran solvent. This reagent cocktail enables the efficient conversion of pentavalent phosphine oxides into trivalent organophosphorus fluorine compounds under relatively mild thermal conditions. 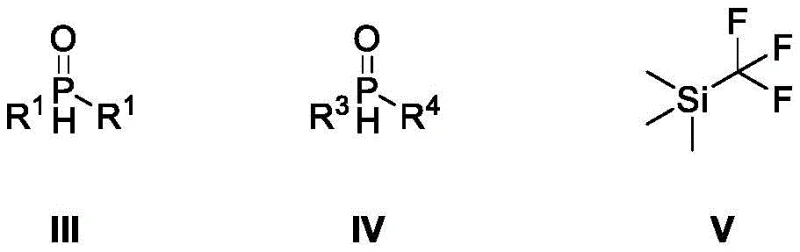 As illustrated in the structural overview of the reactants, the process accommodates a vast array of substituents, ranging from halogenated phenyl groups to aliphatic chains, demonstrating exceptional versatility. The elimination of transition metals not only simplifies the reaction setup but also drastically reduces the environmental footprint, aligning perfectly with modern green chemistry principles. This novel approach transforms a previously niche and difficult transformation into a reliable, scalable process suitable for the production of reliable organophosphorus fluorine compound supplier inventories.
As illustrated in the structural overview of the reactants, the process accommodates a vast array of substituents, ranging from halogenated phenyl groups to aliphatic chains, demonstrating exceptional versatility. The elimination of transition metals not only simplifies the reaction setup but also drastically reduces the environmental footprint, aligning perfectly with modern green chemistry principles. This novel approach transforms a previously niche and difficult transformation into a reliable, scalable process suitable for the production of reliable organophosphorus fluorine compound supplier inventories.
Mechanistic Insights into TMSCF3-Mediated Deoxydifluoromethylation
To fully appreciate the value proposition of this technology for R&D teams, one must delve into the mechanistic underpinnings that drive its high efficiency. The reaction proceeds through a nucleophilic substitution pathway where the activated TMSCF3 species attacks the phosphorus center of the phosphine oxide. The presence of sodium iodide and water plays a critical role in generating the active difluorocarbene or equivalent nucleophilic fluorinating species in situ, facilitating the cleavage of the phosphorus-oxygen double bond. This mechanism is particularly advantageous because it avoids the formation of stable P-O bonds that are notoriously difficult to break without aggressive reagents. The thermal energy provided at 150°C ensures sufficient activation energy for the reaction to proceed to completion within a 12-hour timeframe, balancing reaction rate with thermal stability of the sensitive intermediates. Understanding this mechanism allows process chemists to fine-tune reaction parameters for specific substrates, optimizing the balance between conversion rates and byproduct formation.
From an impurity control perspective, the transition-metal-free nature of this catalytic cycle is a game-changer for product purity. In traditional metal-catalyzed cross-couplings, trace metals can coordinate with the phosphorus lone pair, leading to catalyst poisoning in downstream applications or failing regulatory tests for residual metals in APIs. By circumventing this issue entirely, the new method ensures that the resulting organophosphorus fluorine compounds possess a cleaner impurity profile. 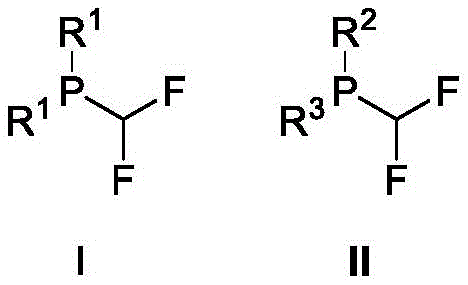 The structural diversity shown in the product schemes highlights the robustness of the method against various electronic environments, whether electron-rich dimethylamino groups or electron-deficient halophenyl rings are present. This consistency in performance across different substrates suggests a highly predictable reaction manifold, which is crucial for scaling up complex organophosphorus ligands without unexpected batch-to-batch variations. The ability to generate these trivalent phosphorus species directly from stable oxides opens new avenues for ligand design that were previously inaccessible due to synthetic bottlenecks.
The structural diversity shown in the product schemes highlights the robustness of the method against various electronic environments, whether electron-rich dimethylamino groups or electron-deficient halophenyl rings are present. This consistency in performance across different substrates suggests a highly predictable reaction manifold, which is crucial for scaling up complex organophosphorus ligands without unexpected batch-to-batch variations. The ability to generate these trivalent phosphorus species directly from stable oxides opens new avenues for ligand design that were previously inaccessible due to synthetic bottlenecks.
How to Synthesize Organophosphorus Fluorine Compounds Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize yield and safety. The protocol involves charging a reactor with the phosphine oxide substrate and TMSCF3 in a precise molar ratio, followed by the addition of the base and additive package. Maintaining an inert nitrogen atmosphere is critical throughout the 12-hour heating period at 150°C to prevent oxidation of the sensitive trivalent phosphorus products. Upon completion, the reaction mixture is subjected to standard workup procedures, typically involving column chromatography to isolate the pure colorless liquids or solids. The detailed standardized synthesis steps see the guide below for exact stoichiometric ratios and safety precautions.
- Prepare the reaction mixture by combining diphenyl(dialkyl)phosphine oxide, TMSCF3, potassium carbonate, sodium iodide, and water in tetrahydrofuran solvent.
- Heat the reaction mixture to 150°C under a nitrogen atmosphere and maintain stirring for 12 hours to ensure complete conversion.
- Upon completion, isolate the target organophosphorus fluorine compound using standard column chromatography separation techniques.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic methodology offers tangible benefits that extend far beyond mere chemical curiosity. The primary advantage lies in the drastic simplification of the manufacturing process, which directly translates to cost reduction in fine chemical intermediates manufacturing. By eliminating the need for expensive transition metal catalysts and the associated purification infrastructure, manufacturers can achieve substantial cost savings on raw materials and processing time. Furthermore, the use of commercially available and relatively inexpensive reagents like TMSCF3 and potassium carbonate ensures a stable and predictable supply chain, mitigating the risks associated with sourcing exotic or controlled chemicals. This reliability is paramount for maintaining continuous production schedules in the fast-paced pharmaceutical sector.
- Cost Reduction in Manufacturing: The economic impact of removing transition metals from the process cannot be overstated. Traditional methods often require palladium or other precious metals, which not only carry high upfront costs but also necessitate complex recovery systems to meet environmental regulations. This new method bypasses these expenses entirely, allowing for a leaner cost structure. Additionally, the high yields reported across a broad substrate scope mean less raw material waste and higher throughput per batch. The simplified purification process, often requiring only standard chromatography rather than specialized metal scavenging resins, further reduces operational expenditures. These factors combine to create a highly competitive cost profile for producing high-purity organophosphorus fluorine compounds.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of robust, non-hazardous reagents that are widely available in the global chemical market. Unlike dangerous fluorinating agents that may be subject to strict transportation regulations or supply shortages, the components of this reaction system are stable and easy to handle. This ease of handling reduces logistical complexities and storage costs, ensuring that production facilities can maintain optimal inventory levels without safety concerns. The scalability of the process from gram to kilogram scales ensures that suppliers can respond rapidly to fluctuating market demands, reducing lead time for high-purity organophosphorus fluorine compounds and preventing production bottlenecks.
- Scalability and Environmental Compliance: From an environmental standpoint, this method aligns with increasingly stringent global regulations regarding chemical waste and emissions. The absence of heavy metals simplifies wastewater treatment and reduces the burden of hazardous waste disposal. The reaction conditions, while requiring elevated temperatures, do not necessitate extreme pressures or cryogenic cooling, making the process energy-efficient and easier to scale in standard stainless steel reactors. This environmental compatibility not only lowers compliance costs but also enhances the corporate sustainability profile of manufacturers adopting this technology. The ability to scale up complex organophosphorus ligands with minimal environmental impact positions this method as a future-proof solution for the green chemistry era.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and background analysis provided in the patent documentation, offering clarity on the practical aspects of the technology. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers reflect the consensus on the method's superiority regarding safety, efficiency, and product quality compared to prior art.
Q: What are the primary advantages of this deoxydifluoromethylation method over traditional fluorination?
A: Unlike traditional methods that often rely on dangerous fluorinating reagents or transition metal catalysts, this novel approach utilizes TMSCF3 with simple inorganic additives. This eliminates the need for expensive metal removal steps, significantly reducing production costs and environmental hazards while maintaining high substrate adaptability.
Q: What is the substrate scope for the R1 group in the phosphine oxide starting material?
A: The method demonstrates excellent versatility, accommodating a wide range of substituents including electron-withdrawing groups like 4-fluorophenyl and 4-chlorophenyl, as well as bulky groups like tert-butyl and naphthyl. This broad scope ensures the process is viable for synthesizing diverse libraries of chiral phosphine ligands.
Q: How does the absence of transition metals impact the purity of the final ligand?
A: The transition-metal-free nature of this catalytic system prevents heavy metal contamination in the final product. This is critical for pharmaceutical applications where strict residual metal limits apply, thereby simplifying the purification workflow and ensuring the resulting ligands meet stringent quality specifications for sensitive catalytic cycles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Organophosphorus Fluorine Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic strategies outlined in CN115403621A for the production of advanced organophosphorus intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent, high-quality materials regardless of volume. Our state-of-the-art facilities are equipped to handle the specific thermal and atmospheric requirements of this deoxydifluoromethylation process, guaranteeing stringent purity specifications and rigorous QC labs testing for every batch. We are committed to delivering reliable organophosphorus fluorine compound supplier solutions that meet the exacting standards of the global pharmaceutical and agrochemical industries.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs, demonstrating how this metal-free route can optimize your budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you navigate the complexities of modern organophosphorus synthesis and secure a competitive advantage in your supply chain.
