Breakthrough Synthesis of 2,5-Dichloro-3-Methoxypyridine: Enabling Commercial Scale-Up of Complex Heterocycles
The pharmaceutical and fine chemical industries have long faced a significant bottleneck regarding the availability of 2,5-dichloro-3-methoxypyridine (CAS: 1214366-19-4), a valuable heterocyclic building block. Historically, the absence of a robust, scalable synthetic route has driven market prices to exorbitant levels, reportedly reaching $300-500 per gram for small quantities. The recent disclosure in patent CN116283748A introduces a transformative three-step methodology that resolves these supply chain constraints through a clever application of N-oxide chemistry. This new approach not only establishes a viable pathway for the first time but also addresses the critical issue of regioselectivity that plagued previous attempts at synthesizing similar dichloropyridine scaffolds. By leveraging inexpensive starting materials like 5-chloro-3-hydroxypyridine and utilizing standard reagents such as methyl iodide and phosphorus oxychloride, this invention paves the way for the commercial scale-up of complex heterocycles essential for next-generation drug discovery programs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of dichloro-hydroxypyridine derivatives relied heavily on direct electrophilic chlorination strategies, which proved fundamentally flawed for this specific substitution pattern. As noted in the background art referencing Synthesis (1990), attempting to chlorinate 3-hydroxypyridine using sodium hypochlorite in aqueous sodium hydroxide resulted in a chaotic reaction profile. This conventional method yielded a complex quaternary mixture containing unreacted starting material, unwanted regioisomers, and over-chlorinated trichloro-species. The inability to remove these impurities through simple crystallization rendered the process commercially unviable, as it demanded fresh, high-quality hypochlorite solutions for even marginal reproducibility. Furthermore, the lack of control over the chlorination position meant that extensive and costly purification protocols were required, drastically reducing overall throughput and making the method unsuitable for industrial factory production requirements where consistency is paramount.
The Novel Approach
The methodology presented in CN116283748A circumvents these historical failures by adopting a stepwise functionalization strategy that prioritizes electronic control over brute-force halogenation. Instead of attacking the ring directly, the process begins with the protection of the 3-hydroxyl group via O-methylation, creating a stable 5-chloro-3-methoxypyridine intermediate. This is followed by a strategic N-oxidation step using m-chloroperoxybenzoic acid (mCPBA), which fundamentally alters the electronic density of the pyridine ring. This activation allows for a highly selective nucleophilic substitution in the final step, where phosphorus oxychloride introduces the second chlorine atom exclusively at the 2-position. This route eliminates the formation of isomeric byproducts and trichloro-impurities, offering a clean, linear progression from cheap raw materials to the high-value target molecule with minimal waste generation.
Mechanistic Insights into N-Oxide Activated Regioselective Chlorination
The core brilliance of this synthesis lies in the utilization of the pyridine N-oxide as a transient activating group to direct the incoming chloride nucleophile. In the second step, the lone pair on the pyridine nitrogen attacks the peracid oxygen of mCPBA, forming the N-oxide species (Compound 2). This oxidation withdraws electron density from the ring carbons, particularly activating the ortho (2 and 6) positions towards nucleophilic attack. When this N-oxide intermediate is subjected to reflux in phosphorus oxychloride (POCl3), the mechanism proceeds via an addition-elimination pathway where the chloride ion attacks the electron-deficient C2 position. The presence of the methoxy group at the 3-position further influences the electronics, ensuring that the substitution occurs cleanly at C2 rather than other potential sites, thereby solving the regioselectivity issues inherent in direct chlorination methods.
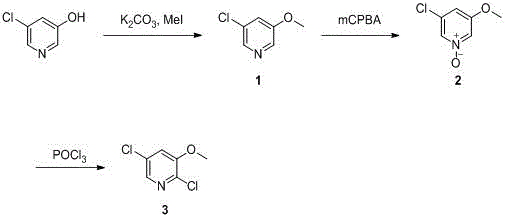
Furthermore, the process design incorporates critical impurity control mechanisms through the physical properties of the intermediates. The initial O-methylation step converts the polar phenolic starting material into a less polar ether (Compound 1), which can be effectively purified via column chromatography using a petroleum ether and ethyl acetate system to remove any unreacted phenol or di-methylated byproducts. Crucially, the patent specifies that the subsequent N-oxide intermediate does not require purification before the final chlorination. This telescoping capability suggests that the byproducts of the oxidation step (such as m-chlorobenzoic acid) do not interfere with the POCl3 reflux, likely because they are either soluble in the reaction medium or removed during the aqueous workup of the final step. This streamlined approach minimizes material loss and ensures that the final white solid product meets stringent purity specifications without the need for complex distillation or recrystallization sequences typically associated with halogenated pyridines.
How to Synthesize 2,5-Dichloro-3-Methoxypyridine Efficiently
The synthesis protocol outlined in the patent provides a clear, reproducible roadmap for laboratory and pilot-scale production, emphasizing precise temperature control and stoichiometry to maximize yield. The process begins with the heating of 5-chloro-3-hydroxypyridine with potassium carbonate and methyl iodide in DMF at 60°C for 6 hours, followed by a standard extractive workup. The resulting intermediate is then oxidized at room temperature (25°C) over 12 hours, a mild condition that prevents degradation of the sensitive N-oxide functionality. Finally, the crude material is refluxed in POCl3 for 3 hours to complete the transformation. For detailed operational parameters, safety considerations, and exact stoichiometric ratios required for GMP-compliant manufacturing, please refer to the standardized synthesis guide below.
- Perform O-methylation of 5-chloro-3-hydroxypyridine using methyl iodide and potassium carbonate in DMF at 60°C to yield 5-chloro-3-methoxypyridine.
- Oxidize the pyridine nitrogen using m-chloroperoxybenzoic acid (mCPBA) in dichloromethane to form the N-oxide intermediate without purification.
- Reflux the crude N-oxide in phosphorus oxychloride (POCl3) to effect regioselective chlorination at the 2-position, followed by purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the implementation of this synthetic route represents a significant opportunity to optimize costs and secure supply continuity for this critical intermediate. The shift from a non-existent or unreliable supply source to a defined, three-step chemical process allows for accurate forecasting and inventory planning. By utilizing commodity chemicals such as potassium carbonate, methyl iodide, and phosphorus oxychloride, the dependency on exotic or single-source reagents is eliminated, thereby mitigating supply chain risks associated with raw material shortages. Additionally, the ability to telescope the oxidation and chlorination steps without intermediate purification reduces solvent consumption and waste disposal costs, contributing to a more sustainable and economically efficient manufacturing profile.
- Cost Reduction in Manufacturing: The primary driver for cost reduction in fine chemical manufacturing via this route is the elimination of complex separation processes required by older chlorination methods. Since the new method avoids the formation of intractable isomer mixtures, the yield loss associated with discarding off-spec material is drastically minimized. Furthermore, the use of inexpensive starting materials like 5-chloro-3-hydroxypyridine, combined with the ability to skip purification of the N-oxide intermediate, significantly lowers the cost of goods sold (COGS). This economic efficiency allows suppliers to offer competitive pricing structures, moving the compound from a research-grade luxury item to a commercially viable bulk chemical.
- Enhanced Supply Chain Reliability: Reliability is bolstered by the robustness of the reaction conditions, which utilize standard heating and reflux techniques compatible with existing stainless steel reactor infrastructure. Unlike methods requiring cryogenic temperatures or specialized catalysts that may have long lead times, this process operates at moderate temperatures (50-70°C for step 1, reflux for step 3) that are easily maintained in multi-purpose plants. This compatibility ensures that production can be scaled up rapidly to meet demand surges without the need for capital-intensive equipment modifications, thus reducing lead time for high-purity intermediates and ensuring consistent delivery schedules for downstream API manufacturers.
- Scalability and Environmental Compliance: The environmental profile of this synthesis is favorable for large-scale operations due to the manageable nature of the byproducts and solvents involved. The process avoids the use of heavy metal catalysts or toxic gases, relying instead on liquid reagents that can be contained and treated using standard effluent handling systems. The high selectivity of the final chlorination step means that the generation of hazardous chlorinated organic waste is kept to a minimum compared to non-selective chlorination routes. This alignment with green chemistry principles facilitates easier regulatory approval for commercial scale-up of complex heterocycles, ensuring long-term operational continuity without environmental compliance bottlenecks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 2,5-dichloro-3-methoxypyridine, based on the specific data points and advantages detailed in patent CN116283748A. Understanding these nuances is critical for R&D teams evaluating this intermediate for new drug candidates and for procurement specialists negotiating supply agreements. The answers provided reflect the current state of the art as defined by the patented methodology, ensuring that all stakeholders have access to accurate and actionable information.
Q: Why is the N-oxide strategy superior to direct chlorination for this compound?
A: Direct chlorination of 3-hydroxypyridine derivatives often yields complex mixtures of isomers and over-chlorinated byproducts that are difficult to separate. The N-oxide strategy activates the 2-position specifically for nucleophilic attack by chloride, ensuring high regioselectivity for the 2,5-dichloro pattern.
Q: What are the critical reaction conditions for the final chlorination step?
A: The final step requires refluxing the N-oxide intermediate in phosphorus oxychloride (POCl3) for 2 to 4 hours. This harsh condition is necessary to replace the N-oxide oxygen with a chlorine atom while maintaining the integrity of the methoxy group at the 3-position.
Q: Is purification required between the oxidation and chlorination steps?
A: No, the patent explicitly states that the crude N-oxide product from the mCPBA oxidation step can be used directly in the subsequent POCl3 reflux step. This telescoping of steps significantly reduces solvent usage and processing time.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,5-Dichloro-3-Methoxypyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of 2,5-dichloro-3-methoxypyridine in the development of advanced pharmaceutical agents and agrochemicals. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and speed. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of this complex heterocycle meets the highest international standards for residue and impurity profiles. We are committed to bridging the gap between innovative patent chemistry and reliable industrial supply.
We invite you to contact our technical procurement team to discuss how we can support your specific project requirements with a Customized Cost-Saving Analysis. Whether you need specific COA data for regulatory filings or route feasibility assessments for process optimization, our experts are ready to provide the detailed technical support necessary to accelerate your timeline. Partner with us to secure a stable, high-quality supply of this critical intermediate and drive your development programs forward with confidence.
