Advanced Chiral Synthesis of D-p-methylsulfonylphenylserine Ethyl Ester for Commercial Scale-up
Introduction to Next-Generation Veterinary Intermediate Synthesis
The pharmaceutical and agrochemical industries are constantly seeking more efficient and environmentally benign pathways for producing critical active pharmaceutical ingredients (APIs) and their precursors. A significant breakthrough in this domain is documented in patent CN115677546B, which details a novel chiral synthesis method for D-p-methylsulfonylphenylserine ethyl ester. This compound serves as a pivotal intermediate in the manufacture of Florfenicol, a broad-spectrum veterinary antibiotic widely used to treat respiratory and intestinal infections in livestock and aquaculture. The disclosed technology represents a paradigm shift from traditional copper-mediated processes to a cleaner, epoxide-based route that leverages modern organic catalysis principles. By utilizing p-methylsulfonylbenzaldehyde and ethyl chloroacetate as primary starting materials, the invention establishes a robust framework for constructing the chiral center early in the synthesis, thereby enhancing stereochemical control and overall process efficiency. This report analyzes the technical merits and commercial implications of this innovation for global supply chain stakeholders.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of D-p-methylsulfonylphenylserine ethyl ester has relied heavily on condensation reactions involving glycine and copper sulfate. While this traditional pathway is well-established, it suffers from severe inherent drawbacks that impact both operational costs and environmental compliance. The process typically involves the formation of a copper salt complex, followed by esterification and a cumbersome resolution step using tartaric acid to separate the desired D-enantiomer from the L-enantiomer. A critical bottleneck in this legacy method is the necessity to remove copper ions from the reaction mixture, a step that often requires the use of sodium sulfide. This generates highly toxic hydrogen sulfide gas, posing significant safety risks to plant personnel and necessitating expensive scrubbing systems to meet environmental regulations. Furthermore, the overall yield of the D-isomer in these conventional processes is notoriously low, often hovering between 40% and 50%, due to losses incurred during the racemization and resolution cycles. The generation of substantial volumes of heavy metal-contaminated wastewater further complicates the waste management profile, driving up the total cost of ownership for manufacturers.
The Novel Approach
In stark contrast to the copper-dependent legacy routes, the methodology described in patent CN115677546B introduces a streamlined, three-step synthesis that bypasses the need for heavy metal catalysts entirely. The new approach initiates with a Darzens-like condensation to form a chiral epoxy ester intermediate, utilizing DBU (1,8-diazabicyclo[5.4.0]undec-7-ene) and sodium ethoxide as promoters. This strategy effectively installs the necessary stereochemistry at the outset, reducing the reliance on downstream resolution techniques that sacrifice half of the material. Subsequent steps involve a regioselective ring-opening reaction with alpha-phenylethylamine, followed by a catalytic hydrogenolysis to remove the chiral auxiliary group. This sequence not only simplifies the operational workflow by eliminating the copper removal and extensive racemization steps but also drastically reduces the environmental footprint by avoiding toxic gas emissions. The result is a process that is inherently safer, cleaner, and capable of delivering significantly higher theoretical yields, addressing the core inefficiencies that have long plagued the production of this key veterinary intermediate.
Mechanistic Insights into DBU-Catalyzed Epoxidation and Hydrogenolysis
The cornerstone of this innovative synthesis lies in the initial formation of the chiral epoxide, specifically (2S, 3R)-3-[4-(methylsulfonyl)phenyl]oxirane-2-carboxylic acid ethyl ester. This transformation is achieved through the reaction of p-methylsulfonylbenzaldehyde with ethyl chloroacetate in the presence of a strong base system comprising sodium ethoxide and the organic superbase DBU. The mechanism likely proceeds via the generation of an enolate from the chloroacetate, which then attacks the aldehyde carbonyl, followed by an intramolecular nucleophilic substitution to close the epoxide ring. The choice of DBU is critical here, as it facilitates the deprotonation and stabilization of intermediates without promoting excessive side reactions that could lead to polymerization or decomposition. Optimization data within the patent indicates that maintaining the reaction temperature between 30°C and 50°C for a duration of 5 hours provides the optimal balance between reaction rate and selectivity. Cooling the reaction mixture to 5°C post-reaction is essential for maximizing the recovery of the solid epoxide intermediate, ensuring high purity before it proceeds to the next stage.
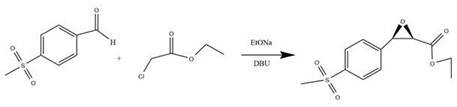
Following the isolation of the chiral epoxide, the synthesis proceeds through a sophisticated ring-opening and reduction sequence to yield the final serine derivative. The epoxide undergoes nucleophilic attack by alpha-phenylethylamine, which serves as a chiral resolving agent and nitrogen source, opening the strained three-membered ring to form an amino-alcohol intermediate. This step is typically conducted at moderate temperatures around 50°C to ensure regioselectivity. The subsequent removal of the phenylethyl group is achieved via catalytic hydrogenolysis using 10% Palladium on Carbon (Pd/C) under hydrogen pressure. This step is crucial for revealing the primary amine functionality of the serine ester while recovering the valuable chiral amine for potential recycling. The patent specifies that hydrogen pressures of 3 MPa and temperatures around 90°C are optimal for this reduction, ensuring complete cleavage of the benzyl-type bond without affecting the sensitive ester or sulfone moieties. This two-stage transformation effectively converts the rigid epoxide scaffold into the flexible amino acid backbone required for Florfenicol synthesis.
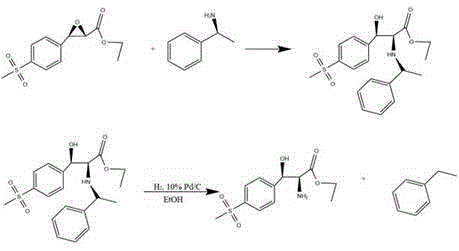
How to Synthesize D-p-methylsulfonylphenylserine Ethyl Ester Efficiently
Implementing this novel synthesis route requires precise control over reaction parameters to replicate the high yields and purity reported in the patent literature. The process is designed to be operationally simple, relying on standard unit operations such as heating, cooling, filtration, and distillation, which facilitates easy adoption in existing multipurpose chemical plants. The key to success lies in the strict adherence to the stoichiometric ratios and thermal profiles defined in the experimental examples, particularly during the exothermic epoxidation step and the high-pressure hydrogenation phase. By following the optimized conditions, manufacturers can achieve a total calculated yield exceeding 70%, a substantial improvement over legacy methods. For detailed operational protocols, safety guidelines, and specific equipment requirements necessary for scaling this chemistry, please refer to the standardized synthesis guide provided below.
- Perform a DBU-catalyzed condensation between p-methylsulfonylbenzaldehyde and ethyl chloroacetate to form the chiral epoxide intermediate.
- Execute a ring-opening reaction using alpha-phenylethylamine followed by catalytic hydrogenation with Pd/C to remove the chiral auxiliary.
- Purify the crude product through acid-base extraction and crystallization to obtain the final high-purity ester.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this new synthetic route offers compelling economic and logistical benefits that extend beyond simple yield improvements. The elimination of copper sulfate and the associated copper removal steps fundamentally alters the cost structure of production by removing the need for expensive heavy metal scavengers and the disposal of hazardous copper-laden sludge. This simplification translates directly into reduced raw material costs and lower waste treatment fees, enhancing the overall margin profile of the intermediate. Furthermore, the avoidance of hydrogen sulfide generation mitigates significant regulatory and safety liabilities, ensuring uninterrupted production schedules free from environmental compliance shutdowns. The robustness of the process, demonstrated by its tolerance to slight variations in temperature and pressure, also suggests a high degree of reliability for long-term supply contracts, reducing the risk of batch failures that can disrupt downstream API manufacturing.
- Cost Reduction in Manufacturing: The new process achieves cost optimization primarily through the elimination of costly resolution agents and the reduction of processing steps. By avoiding the copper-glycine condensation and the subsequent tartaric acid resolution, the material consumption per kilogram of product is significantly lowered. Additionally, the recovery of solvents and the potential recycling of the alpha-phenylethylamine chiral auxiliary further contribute to a leaner cost base. The higher overall yield means that less starting material is required to produce the same amount of final product, effectively amplifying the purchasing power of raw material budgets and driving down the unit cost of goods sold.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the use of commodity chemicals such as p-methylsulfonylbenzaldehyde and ethyl chloroacetate, which are readily available from multiple global suppliers, reducing dependency on single-source specialty reagents. The simplified workflow, which removes the complex and time-consuming copper removal and racemization loops, shortens the overall cycle time for each production batch. This increased throughput capacity allows manufacturers to respond more agilely to fluctuations in market demand for Florfenicol, ensuring that inventory levels remain stable even during peak seasons. The reduced generation of hazardous waste also minimizes the risk of logistical bottlenecks associated with the transport and disposal of toxic byproducts.
- Scalability and Environmental Compliance: From a scalability perspective, the process is highly amenable to scale-up as it avoids the use of cryogenic conditions or extremely high pressures that often limit reactor size. The reaction conditions are mild enough to be handled in standard glass-lined or stainless steel reactors found in most fine chemical facilities. Environmentally, the shift away from heavy metals and toxic gases aligns perfectly with increasingly stringent global environmental, social, and governance (ESG) standards. This 'green' credential not only future-proofs the supply chain against tightening regulations but also enhances the brand value of the final veterinary product by associating it with sustainable manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral synthesis technology. These answers are derived directly from the experimental data and beneficial effects outlined in the patent documentation, providing a clear understanding of the process capabilities and limitations. Stakeholders are encouraged to review these points to assess the feasibility of integrating this route into their existing production portfolios.
Q: How does this new synthesis method improve upon traditional copper-based routes?
A: The new method eliminates the use of copper sulfate and glycine, thereby avoiding the generation of toxic hydrogen sulfide gas during copper removal and significantly simplifying the wastewater treatment process while improving overall yield from roughly 40-50% to over 70%.
Q: What are the critical reaction conditions for the epoxidation step?
A: Optimal results are achieved by maintaining the catalytic reaction temperature between 30°C and 50°C for approximately 5 hours, followed by cooling to 5°C to maximize the precipitation and purity of the chiral epoxide intermediate.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes common reagents like sodium ethoxide and DBU, avoids complex resolution steps requiring large amounts of resolving agents, and demonstrates robust yields under standard hydrogenation pressures, making it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable D-p-methylsulfonylphenylserine Ethyl Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the production of effective veterinary medicines. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications for every batch of D-p-methylsulfonylphenylserine ethyl ester we produce. We are committed to leveraging innovations like the one described in CN115677546B to deliver superior products that meet the evolving needs of the global pharmaceutical market.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us demonstrate how our advanced synthesis capabilities can drive value for your organization.
