Advanced Photochemical Synthesis of Benzyl Phosphine Oxides for Scalable Pharmaceutical Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and environmentally benign pathways to access complex organophosphorus scaffolds, which serve as critical building blocks for bioactive molecules. Patent CN113292599B introduces a groundbreaking methodology for the synthesis of benzyl phosphine oxide compounds, a class of molecules characterized by their heterocyclic aromatic hydrocarbon phosphine oxide moieties and highly active hydroxyl groups. These structural features endow the compounds with significant biological activity, making them exceptionally valuable as pharmaceutical intermediates with broad applications in antiviral, antitumor, and antibacterial drug development. The core innovation lies in a visible-light-driven protocol that operates under remarkably mild conditions, utilizing air as the sole oxidant and eliminating the need for toxic heavy metal catalysts or hazardous peroxides. This technological leap not only enhances the safety profile of the manufacturing process but also streamlines the purification workflow, offering a compelling value proposition for reliable pharmaceutical intermediate suppliers aiming to optimize their production pipelines.
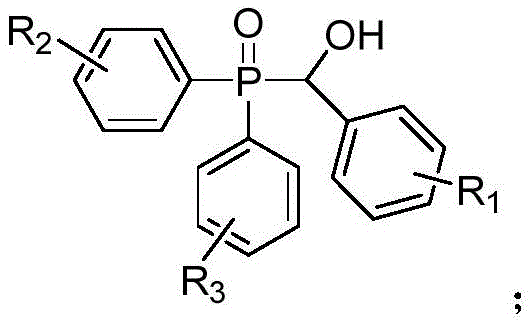
Traditionally, the construction of carbon-phosphorus bonds in benzyl phosphine oxide systems has been fraught with significant operational challenges and safety concerns. Conventional synthetic strategies typically rely on the use of stoichiometric amounts of organic peroxides to drive the oxidative coupling, often necessitating elevated temperatures that can compromise the integrity of sensitive functional groups. Furthermore, many established protocols depend on transition metal catalysts, such as copper or TEMPO-mediated systems, which introduce the risk of heavy metal contamination in the final product—a critical failure point for GMP-compliant API manufacturing. The removal of these metal residues requires additional, costly purification steps, such as scavenging or recrystallization, which inevitably erodes overall process efficiency and yield. In stark contrast, the novel approach detailed in the patent leverages visible light photocatalysis to generate reactive radical species directly from molecular oxygen, bypassing the need for external chemical oxidants. This paradigm shift allows the reaction to proceed at a moderate temperature of 60°C under ambient air pressure, drastically simplifying the reactor requirements and enhancing the inherent safety of the operation by removing explosive peroxide hazards from the process inventory.
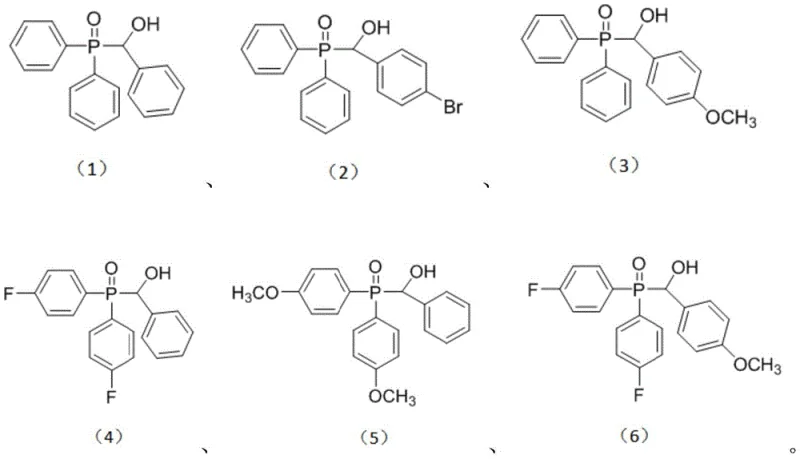
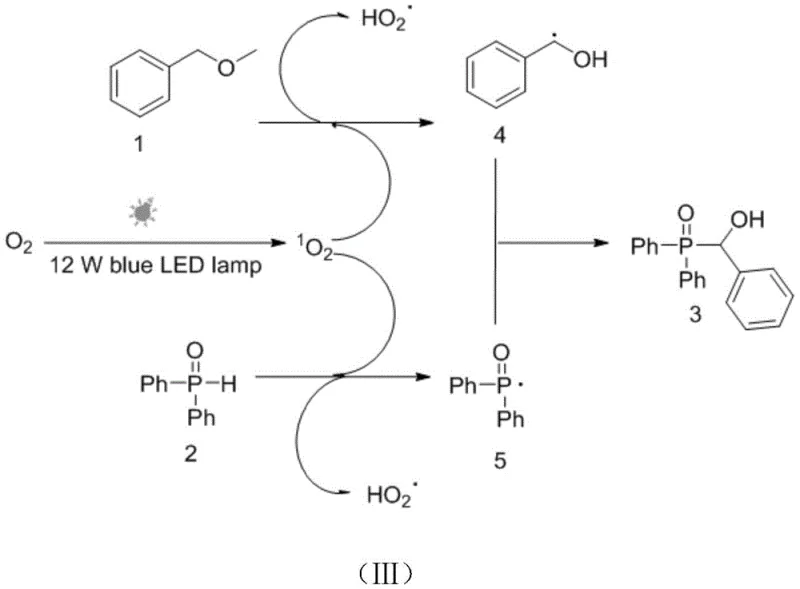
The mechanistic elegance of this transformation is rooted in a radical-radical cross-coupling pathway initiated by singlet oxygen species generated in situ. Upon irradiation with a 12W blue LED lamp or a light source within the 400-800nm range, molecular oxygen is excited to its singlet state, which subsequently abstracts hydrogen atoms from both the benzyl methyl ether substrate and the diaryl phosphine oxide reagent. This dual hydrogen abstraction generates two distinct radical intermediates: a benzylic radical stabilized by the adjacent aromatic ring and a phosphinoyl radical centered on the phosphorus atom. These transient species then undergo a rapid and selective cross-coupling event to forge the new C-P bond, yielding the target benzyl phosphine oxide product with high fidelity. The mildness of this radical generation mechanism is crucial for impurity control; unlike thermal radical initiators that can lead to non-selective bond cleavage and polymerization, the photochemical activation offers precise temporal and spatial control over radical concentration. This results in a cleaner reaction profile with fewer side products, facilitating easier downstream processing and ensuring that the final material meets the stringent purity specifications required for high-purity pharmaceutical intermediates.
How to Synthesize Benzyl Phosphine Oxide Efficiently
The practical implementation of this synthesis route is designed for operational simplicity, requiring only commercially available starting materials and standard laboratory equipment. The process begins by combining the diaryl phosphine oxide compound and the benzyl methyl ether compound in a pressure-resistant reaction vessel, typically utilizing a significant excess of the ether substrate which also serves as the reaction medium. The mixture is then subjected to irradiation under an air atmosphere, where the interplay between light energy and molecular oxygen drives the transformation without the need for inert gas protection or specialized high-pressure equipment. While the general procedure is straightforward, precise control over reaction parameters such as light wavelength, temperature, and molar ratios is essential to maximize conversion and minimize reaction time. For a comprehensive understanding of the standardized operating procedures and specific workup protocols, please refer to the detailed guide below.
- Mix diaryl phosphine oxide compound and benzyl methyl ether compound in a pressure-resistant tube with a molar ratio of 1: (50-200).
- Irradiate the mixture with a light source (400-800nm, preferably 600nm or blue LED) under an air atmosphere.
- Stir the reaction at 40-100°C (optimally 60°C) for approximately 36 hours, then purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this metal-free photochemical technology offers substantial opportunities for cost optimization and supply chain resilience. By completely eliminating the requirement for precious metal catalysts such as palladium, copper, or rhodium, manufacturers can achieve significant cost reduction in pharmaceutical intermediate manufacturing, as the volatility of noble metal prices no longer impacts the bill of materials. Furthermore, the absence of metal catalysts removes the necessity for expensive metal scavenging resins and the associated validation testing for residual metals, thereby shortening the release timeline for batch certification. The reliance on air as the terminal oxidant further decouples the process from the supply chain risks associated with hazardous chemical oxidants like peroxides, which often require special storage, handling, and transportation protocols. This simplification of the raw material portfolio enhances supply chain reliability, ensuring continuous production even during periods of logistical disruption for specialty reagents.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the drastic simplification of the reaction setup and the elimination of costly catalytic systems. Without the need for transition metals, the raw material costs are inherently lower, and the downstream purification process is significantly streamlined, reducing solvent consumption and waste disposal fees. The ability to run the reaction at moderate temperatures (60°C) rather than high thermal loads also translates to lower energy consumption per kilogram of product, contributing to a leaner manufacturing cost structure. Additionally, the high conversion rates and yields reported (up to 81%) mean that less starting material is wasted, maximizing the atom economy and overall process efficiency.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route is bolstered by the use of commodity chemicals that are widely available from multiple global suppliers. Benzyl methyl ethers and diaryl phosphine oxides are stable, shelf-stable reagents that do not suffer from the short shelf-life or sensitivity issues common to many organometallic catalysts. Operating under an air atmosphere removes the dependency on bulk nitrogen or argon supplies, further insulating the production process from utility shortages. This accessibility of inputs ensures that production schedules can be maintained with high predictability, reducing lead time for high-purity pharmaceutical intermediates and allowing for more agile response to market demand fluctuations.
- Scalability and Environmental Compliance: Scaling this technology from benchtop to commercial production is facilitated by the benign nature of the reagents and the absence of explosive peroxides. The photochemical nature of the reaction can be adapted to flow chemistry setups or large-scale photoreactors, enabling the commercial scale-up of complex pharmaceutical intermediates with consistent quality. From an environmental standpoint, the process aligns with green chemistry principles by utilizing visible light as a renewable energy source and generating water as the primary byproduct of the oxidation, rather than toxic heavy metal waste streams. This favorable environmental profile simplifies regulatory compliance and waste treatment, making it an attractive option for facilities aiming to reduce their carbon footprint and adhere to stricter environmental regulations.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of integrating this technology into their existing workflows, we have compiled answers to common inquiries regarding the reaction scope, safety parameters, and product quality. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a transparent view of the process capabilities. Understanding these details is crucial for project managers assessing the risk-benefit ratio of adopting new synthetic methodologies for critical API intermediates.
Q: What are the primary advantages of this photochemical method over traditional metal-catalyzed routes?
A: This method eliminates the need for expensive transition metal catalysts and hazardous peroxides, operating under mild conditions (60°C) with air as the oxidant, which significantly reduces environmental impact and purification costs.
Q: What is the expected yield and purity profile for these benzyl phosphine oxide derivatives?
A: The patented process demonstrates high conversion rates with isolated yields reaching up to 81% for unsubstituted derivatives, producing high-purity white solids suitable for downstream pharmaceutical applications after standard chromatographic purification.
Q: Is this synthesis protocol scalable for industrial production of API intermediates?
A: Yes, the use of simple reagents, ambient air pressure, and visible light irradiation makes the process highly amenable to scale-up, avoiding the safety hazards associated with high-pressure peroxide reactions found in conventional methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzyl Phosphine Oxide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this photochemical synthesis route for the next generation of organophosphorus therapeutics. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to industrial manufacturing is seamless and efficient. Our facility is equipped with state-of-the-art photoreactors and rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of benzyl phosphine oxide delivered meets the highest standards of quality and consistency required by global regulatory bodies.
We invite you to collaborate with our technical procurement team to explore how this innovative methodology can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic advantages specific to your target molecule. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to delivering superior value through advanced chemical engineering and supply chain excellence.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
