Advanced Catalytic Synthesis of 2-Keto Aryl Benzoxazoles for Commercial Pharmaceutical Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking more efficient and sustainable pathways to access complex heterocyclic scaffolds, particularly those with proven biological activity. Patent CN115557911A introduces a groundbreaking methodology for the synthesis of 2-keto aryl benzoxazole compounds, a structural motif prevalent in numerous bioactive molecules including FAAH inhibitors and protease inhibitors. This innovation represents a significant departure from traditional synthetic routes by utilizing readily available aryl carboxylic acids and benzoxazoles as primary building blocks. The disclosed method employs a sophisticated dual-catalytic system involving palladium acetate and copper chloride, facilitated by specific phosphine ligands and activating agents. By shifting the paradigm from hazardous acid halides to stable carboxylic acids, this technology not only enhances the safety profile of the manufacturing process but also opens new avenues for cost-effective production of high-value pharmaceutical intermediates. The robustness of this approach is evidenced by its ability to tolerate a wide range of functional groups, ensuring versatility for diverse drug discovery programs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 2-keto aryl benzoxazole core has relied heavily on the cyclization of o-aminophenols with aryl acid halides or the coupling of benzoxazoles with aryl halides. These conventional strategies suffer from inherent drawbacks that hinder their applicability in modern green manufacturing. The use of acid chlorides, for instance, introduces significant safety hazards due to their corrosive nature and sensitivity to moisture, necessitating stringent handling protocols and specialized equipment. Furthermore, the generation of stoichiometric amounts of halide waste contradicts the principles of atom economy and environmental sustainability. Alternative methods involving decarboxylative coupling often require pre-functionalized substrates or harsh conditions that limit substrate scope and increase overall process complexity. The reliance on toxic halogenated reagents not only escalates waste disposal costs but also poses challenges in meeting increasingly rigorous regulatory standards for residual impurities in active pharmaceutical ingredients.
The Novel Approach
In stark contrast, the methodology described in patent CN115557911A offers a streamlined and environmentally benign alternative by directly coupling aryl carboxylic acids with benzoxazoles. This novel route leverages the stability and low cost of carboxylic acids, which are commodity chemicals available in vast structural diversity. The reaction proceeds efficiently under relatively mild thermal conditions using a synergistic Pd/Cu catalytic system, achieving high conversion rates without the need for exotic reagents. By employing di-tert-butyl dicarbonate as an activating agent, the process effectively transforms the inert carboxylic acid into a reactive species in situ, facilitating the C-C bond formation with the azole ring. This strategic shift eliminates the generation of halide byproducts and simplifies the downstream purification process. The result is a cleaner reaction profile that aligns perfectly with the industry's drive towards greener synthesis and reduced environmental footprint.
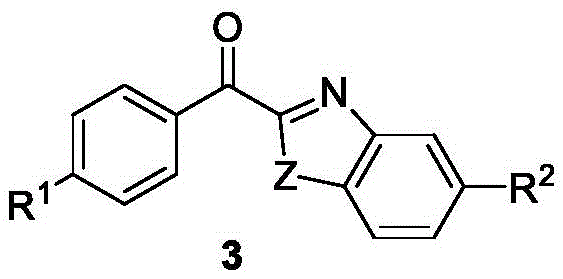
Mechanistic Insights into Pd/Cu Dual-Catalyzed Oxidative Coupling
The success of this transformation hinges on the intricate interplay between the palladium and copper catalysts, which work in concert to activate both coupling partners. The proposed mechanism likely involves the initial activation of the aryl carboxylic acid by di-tert-butyl dicarbonate to form a mixed anhydride or a similar activated intermediate. Simultaneously, the copper catalyst may facilitate the C-H activation of the benzoxazole ring at the C2 position, generating a nucleophilic organocopper species. The palladium catalyst then mediates the cross-coupling event, potentially through a transmetallation step followed by reductive elimination to forge the critical ketone-azole bond. The presence of the phosphine ligand, specifically diphenylcyclohexylphosphine, is crucial for stabilizing the palladium center and modulating its electronic properties to favor the desired catalytic cycle. This dual-metal strategy allows for the overcoming of high energy barriers associated with direct C-H functionalization, enabling the reaction to proceed with high selectivity and efficiency.
From an impurity control perspective, the choice of reagents and conditions plays a pivotal role in ensuring product purity. The use of sodium bicarbonate as a mild base helps to neutralize acidic byproducts without promoting unwanted side reactions such as hydrolysis of the sensitive ketone moiety. Furthermore, the specific ligand environment around the metal centers minimizes the formation of homocoupling byproducts, which are common pitfalls in oxidative coupling reactions. The reaction temperature of 120°C in cyclohexane provides sufficient thermal energy to drive the catalytic cycle to completion while maintaining a homogeneous reaction mixture that facilitates heat and mass transfer. Understanding these mechanistic nuances is essential for R&D teams aiming to optimize the process further or adapt it to continuous flow manufacturing setups, ensuring consistent quality and reproducibility at scale.
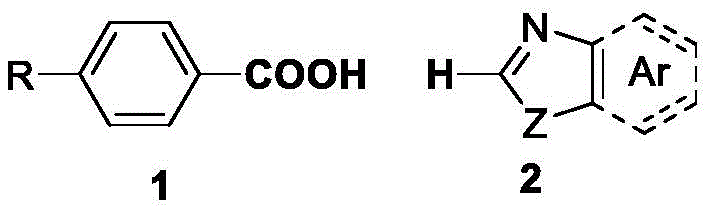
How to Synthesize 2-Keto Aryl Benzoxazole Efficiently
The practical execution of this synthesis is designed to be straightforward, making it accessible for both laboratory-scale optimization and pilot plant operations. The general procedure involves charging a reaction vessel with the aryl carboxylic acid and benzoxazole substrates in a defined molar ratio, typically favoring a slight excess of the acid to drive the equilibrium forward. The catalytic system, comprising palladium acetate and copper chloride along with the phosphine ligand, is introduced alongside the activating agent di-tert-butyl dicarbonate and the base. Cyclohexane serves as the reaction medium, providing a non-polar environment that supports the solubility of organic substrates while allowing for easy removal post-reaction. The mixture is then heated to 120°C and maintained for approximately 12 hours to ensure full conversion. Following the reaction, standard workup procedures involving filtration and column chromatography yield the target 2-keto aryl benzoxazole compounds with high purity. For a detailed breakdown of the standardized synthesis steps, please refer to the guide below.
- Combine aryl carboxylic acid and benzoxazole derivatives with palladium acetate and copper chloride catalysts in a reaction vessel.
- Add diphenylcyclohexylphosphine ligand, di-tert-butyl dicarbonate as an activator, sodium bicarbonate base, and cyclohexane solvent.
- Heat the mixture to 120°C for 12 hours, then purify the resulting 2-keto aryl benzoxazole product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers compelling economic and logistical benefits that extend beyond mere technical feasibility. The substitution of expensive and hazardous acid chlorides with stable, commodity-grade aryl carboxylic acids fundamentally alters the cost structure of the manufacturing process. This shift not only reduces the direct cost of goods sold but also mitigates the risks associated with the storage and handling of dangerous reagents. The simplified workflow, characterized by fewer unit operations and the absence of complex quenching steps required for acid halides, translates into shorter batch cycles and increased throughput. Moreover, the high yields reported across a broad substrate scope mean that less raw material is wasted, enhancing the overall material efficiency of the production line. These factors collectively contribute to a more resilient and cost-competitive supply chain capable of meeting the demanding timelines of the pharmaceutical sector.
- Cost Reduction in Manufacturing: The elimination of toxic halide precursors removes the need for specialized corrosion-resistant equipment and extensive safety measures, leading to significant capital expenditure savings. Additionally, the use of inexpensive solvents like cyclohexane and readily available catalysts ensures that operating expenses remain low. The high atom economy of the reaction minimizes waste disposal costs, further driving down the total cost of ownership for the manufacturing process. By streamlining the synthesis to a single pot operation where possible, labor costs and energy consumption are also optimized, providing a clear path to margin improvement.
- Enhanced Supply Chain Reliability: Sourcing aryl carboxylic acids is inherently more stable than relying on specialized acid chlorides, which often have limited suppliers and longer lead times. The robustness of the reaction conditions allows for flexibility in raw material sourcing, reducing dependency on single-source vendors. This diversification of the supply base enhances continuity and reduces the risk of production stoppages due to material shortages. Furthermore, the stability of the intermediates allows for safer transportation and storage, simplifying logistics and reducing insurance premiums associated with hazardous cargo.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard heating and stirring equipment that is readily available in most multipurpose chemical plants. The absence of heavy metal contamination issues, thanks to the efficient catalytic system and purification methods, simplifies compliance with strict environmental regulations regarding heavy metal residues in APIs. The green chemistry credentials of the method, characterized by reduced waste generation and the use of less toxic reagents, align with corporate sustainability goals and facilitate easier regulatory approval. This makes the technology not just a chemical solution, but a strategic asset for long-term sustainable growth.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a reliable foundation for decision-making. Understanding the nuances of catalyst loading, substrate compatibility, and purification requirements is critical for successful technology transfer. We encourage potential partners to review these details closely to assess the fit for their specific project needs.
Q: What are the primary advantages of using aryl carboxylic acids over acid chlorides in this synthesis?
A: Using aryl carboxylic acids eliminates the need for toxic and moisture-sensitive acid halides, significantly improving operational safety and reducing raw material costs while adhering to green chemistry principles.
Q: What is the typical yield range for this Pd/Cu catalyzed coupling reaction?
A: The patent data indicates high efficiency, with isolated yields frequently exceeding 80% for various substrates, demonstrating the robustness of the catalytic system across different electronic environments.
Q: Is this process scalable for industrial production of pharmaceutical intermediates?
A: Yes, the method utilizes common solvents like cyclohexane and standard heating conditions (120°C) without requiring specialized high-pressure equipment, making it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Keto Aryl Benzoxazole Supplier
The technological advancements presented in patent CN115557911A underscore the immense potential of 2-keto aryl benzoxazoles as versatile building blocks in modern drug discovery. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate such innovative laboratory methodologies into robust commercial processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from bench to plant is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing the quality and consistency required for clinical and commercial applications. Our dedication to process optimization allows us to offer competitive pricing without compromising on the integrity of the final product.
We invite you to collaborate with us to leverage this cutting-edge synthesis route for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can accelerate your development timeline and enhance your supply chain efficiency. Let us be your partner in turning complex chemical challenges into commercial successes.
