Advanced Nickel-Catalyzed Synthesis of 2-Cyclohexylbenzamide for Scalable Pharmaceutical Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct C(sp2)-C(sp3) bonds, a critical transformation for accessing diverse bioactive scaffolds. Patent CN110724064A introduces a groundbreaking nickel-catalyzed strategy for the direct synthesis of 2-cyclohexyl-substituted benzamides, addressing long-standing challenges in C-H functionalization. This technology leverages the unique redox properties of nickel to activate inert C-H bonds in cyclohexane, coupling them directly with benzamide derivatives without the need for pre-halogenated substrates. For R&D directors and process chemists, this represents a paradigm shift from atom-inefficient cross-coupling reactions to streamlined cross-dehydrogenative coupling (CDC) protocols. The ability to utilize cyclohexane simultaneously as a solvent and a reactant drastically simplifies the reaction matrix, offering a compelling value proposition for the manufacturing of high-purity pharmaceutical intermediates. By circumventing the use of expensive noble metals and toxic halides, this innovation aligns perfectly with modern green chemistry principles while enhancing economic viability for large-scale production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of ortho-alkylated benzamides has relied heavily on transition metal catalysis involving complex directing groups and pre-activated electrophiles. One prominent prior art approach utilizes quinoline as a directing group to coordinate with nickel, facilitating ortho-C-H activation; however, this method suffers from significant drawbacks regarding substrate scope and safety. As illustrated in the reaction scheme below, this pathway necessitates the use of toxic halogenated alkylating agents, which pose severe environmental and handling hazards in a commercial setting. 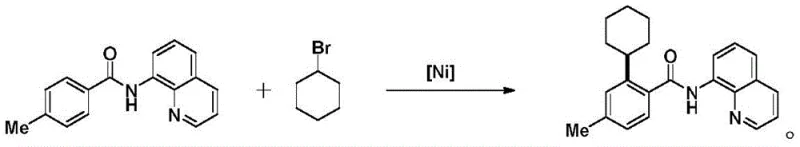 Furthermore, the requirement to install and subsequently remove the quinoline moiety adds multiple synthetic steps, increasing the overall cost of goods sold (COGS) and extending the manufacturing lead time. Another conventional strategy employs cobalt catalysis with pyridine-based directing groups to achieve similar transformations. While effective in academic settings, this approach often demands rigorous exclusion of air and moisture, along with substrates that require tedious pre-activation. These complexities create bottlenecks in supply chain reliability, as the sourcing of specialized directing group precursors can be inconsistent and costly, thereby limiting the scalability of such processes for industrial applications.
Furthermore, the requirement to install and subsequently remove the quinoline moiety adds multiple synthetic steps, increasing the overall cost of goods sold (COGS) and extending the manufacturing lead time. Another conventional strategy employs cobalt catalysis with pyridine-based directing groups to achieve similar transformations. While effective in academic settings, this approach often demands rigorous exclusion of air and moisture, along with substrates that require tedious pre-activation. These complexities create bottlenecks in supply chain reliability, as the sourcing of specialized directing group precursors can be inconsistent and costly, thereby limiting the scalability of such processes for industrial applications.
The Novel Approach
In stark contrast to these cumbersome legacy methods, the technology disclosed in CN110724064A offers a remarkably streamlined solution that bypasses the need for directing groups entirely. The novel protocol achieves direct C-H alkylation by employing a simple nickel chloride catalyst system in conjunction with a robust oxidant and ligand combination. 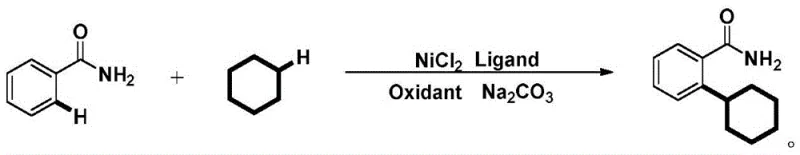 This approach allows for the direct coupling of unmodified benzamide with cyclohexane, effectively turning a common solvent into a valuable chemical building block. The elimination of halogenated reagents not only improves the safety profile of the reaction but also simplifies the downstream purification process, as there are no halide salts to remove from the waste stream. By operating at moderate temperatures between 120°C and 135°C, the process maintains high energy efficiency while delivering competitive yields. This simplicity translates directly into operational excellence, enabling procurement teams to source generic, commodity-grade raw materials rather than relying on bespoke, high-cost specialty chemicals. The result is a manufacturing route that is inherently more resilient to market fluctuations and supply disruptions.
This approach allows for the direct coupling of unmodified benzamide with cyclohexane, effectively turning a common solvent into a valuable chemical building block. The elimination of halogenated reagents not only improves the safety profile of the reaction but also simplifies the downstream purification process, as there are no halide salts to remove from the waste stream. By operating at moderate temperatures between 120°C and 135°C, the process maintains high energy efficiency while delivering competitive yields. This simplicity translates directly into operational excellence, enabling procurement teams to source generic, commodity-grade raw materials rather than relying on bespoke, high-cost specialty chemicals. The result is a manufacturing route that is inherently more resilient to market fluctuations and supply disruptions.
Mechanistic Insights into Nickel-Catalyzed Cross-Dehydrogenative Coupling
The core of this technological advancement lies in the sophisticated interplay between the nickel catalyst, the ligand environment, and the oxidant system. The reaction likely proceeds through a radical-mediated mechanism where the nickel center facilitates the homolytic cleavage of the oxidant, generating reactive radical species capable of abstracting hydrogen atoms from the cyclohexane ring. This generates a cyclohexyl radical which then adds to the nickel-coordinated benzamide intermediate. The use of ligands such as tricyclohexylphosphine or 1,10-phenanthroline is critical, as they stabilize the active nickel species and modulate its electronic properties to favor the desired C-C bond formation over competing side reactions. Understanding this mechanistic nuance is vital for R&D teams aiming to adapt this chemistry to broader substrate scopes, as the steric and electronic nature of the ligand can be tuned to accommodate different substitution patterns on the benzamide ring. The presence of sodium carbonate as a base further assists in the deprotonation steps necessary to regenerate the active catalytic cycle, ensuring turnover numbers remain high throughout the 18-hour reaction window.
From an impurity control perspective, this mechanism offers distinct advantages over traditional cross-coupling reactions. Since the process avoids the use of aryl halides, the formation of homocoupling byproducts (biaryls) typically associated with oxidative addition/reductive elimination cycles is minimized. The primary impurities are likely to be over-alkylated species or oxidation byproducts of the cyclohexane, both of which are generally easier to separate via standard chromatographic techniques or crystallization. The patent data indicates that optimizing the molar ratio of the oxidant to the substrate is key to suppressing these side reactions; specifically, maintaining an oxidant-to-benzamide ratio between 1.5:1 and 3.5:1 ensures complete conversion without excessive degradation of the product. This level of control over the impurity profile is essential for meeting the stringent purity specifications required for pharmaceutical intermediates, reducing the burden on quality control laboratories and accelerating batch release times.
How to Synthesize 2-Cyclohexyl-substituted Benzamide Efficiently
To implement this synthesis effectively, operators must adhere to precise stoichiometric ratios and thermal profiles as outlined in the patent examples. The process begins with the preparation of the catalytic mixture, followed by the sequential addition of reagents to ensure proper initiation of the radical chain reaction. Detailed standardized operating procedures regarding the specific order of addition and workup protocols are critical for reproducibility.
- Prepare the catalytic system by mixing nickel chloride and a phosphine or nitrogen ligand in cyclohexane solvent at room temperature.
- Add the oxidant (such as tert-butyl peroxide) and sodium carbonate base to the mixture, followed by the benzamide substrate.
- Heat the reaction mixture to 120°C-135°C for 18 hours, then perform extraction with ethyl acetate and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this nickel-catalyzed methodology presents a transformative opportunity to optimize cost structures and enhance supply security. The shift from precious metal catalysts to base metal nickel results in a drastic reduction in raw material expenditure, as nickel salts are orders of magnitude cheaper than palladium or rhodium alternatives. Moreover, the ability to use cyclohexane as both the solvent and the reactant eliminates the need to purchase and store large volumes of separate alkylating agents, thereby reducing inventory holding costs and minimizing the facility's chemical footprint. This consolidation of materials simplifies logistics and reduces the complexity of hazardous material handling, leading to substantial operational savings. The robustness of the reaction conditions also implies a lower risk of batch failures due to sensitive reagent degradation, ensuring a more consistent and reliable output of high-purity intermediates for downstream drug synthesis.
- Cost Reduction in Manufacturing: The replacement of expensive noble metal catalysts with affordable nickel chloride significantly lowers the direct material costs associated with the catalytic system. Additionally, the avoidance of pre-functionalized substrates and toxic halides removes the cost burden of synthesizing or purchasing these complex starting materials. The simplified workup procedure, which involves basic extraction and distillation rather than complex scavenging of heavy metals, further reduces processing time and utility consumption. These factors combine to deliver a highly cost-efficient manufacturing process that improves overall margin potential for the final API or intermediate.
- Enhanced Supply Chain Reliability: By relying on commodity chemicals such as benzamide, cyclohexane, and sodium carbonate, the supply chain becomes far less vulnerable to the volatility often seen in the market for specialized organometallic reagents. These bulk chemicals are produced globally in massive quantities, ensuring continuous availability and stable pricing even during periods of geopolitical tension or logistical disruption. This stability allows for better long-term planning and contract negotiation, securing the continuity of supply for critical pharmaceutical projects. The reduced dependency on single-source suppliers for exotic ligands or catalysts further diversifies the supply base, mitigating risk.
- Scalability and Environmental Compliance: The absence of halogenated waste streams simplifies effluent treatment and disposal, aligning the process with increasingly strict environmental regulations. The use of cyclohexane, a common industrial solvent, facilitates easier solvent recovery and recycling, contributing to a more sustainable manufacturing lifecycle. The reaction's tolerance to standard laboratory glassware and heating equipment suggests that scale-up from kilogram to tonne scale can be achieved with minimal capital investment in specialized reactors. This ease of scale-up ensures that production capacity can be rapidly expanded to meet surging market demand without compromising on safety or quality standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this nickel-catalyzed synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation.
Q: What are the primary advantages of this nickel-catalyzed method over traditional palladium catalysis?
A: This method utilizes inexpensive nickel chloride instead of precious metals like palladium or rhodium, significantly reducing raw material costs. Furthermore, it employs cyclohexane directly as both solvent and reactant, eliminating the need for pre-functionalized alkylating agents.
Q: Does this process require complex directing groups on the benzamide substrate?
A: No, unlike prior art methods that require quinoline or pyridine directing groups which necessitate additional synthetic steps for installation and removal, this protocol operates efficiently on simple benzamide substrates without extra functionalization.
Q: What represent the optimal reaction conditions for maximum yield?
A: Experimental data indicates that using tricyclohexylphosphine as the ligand with tert-butyl peroxide as the oxidant at 135°C provides the highest yields, reaching up to 71% under optimized molar ratios of catalyst and base.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Cyclohexylbenzamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient C-H activation technologies in modern drug discovery and development. Our team of expert process chemists has extensively evaluated the nickel-catalyzed route described in CN110724064A and confirmed its potential for robust commercial manufacturing. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of high-quality intermediates. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 2-cyclohexyl-substituted benzamide meets the exacting standards required for pharmaceutical applications. We are committed to leveraging this advanced chemistry to deliver superior value to our global partners.
We invite procurement leaders and R&D directors to collaborate with us to explore how this cost-effective synthesis route can be integrated into your supply chain. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and project timelines. We encourage you to contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments. Let us help you accelerate your development programs with reliable, scalable, and economically viable chemical solutions.
