Advanced Synthesis of 2-Benzylfuran-4-Methanol for Scalable Agrochemical Manufacturing
The global demand for high-performance pyrethroid insecticides continues to drive the need for efficient, scalable synthesis routes for their critical building blocks. Patent CN103880791A introduces a transformative methodology for the production of 2-benzylfuran-4-methanol, also widely known in the industry as Elliott alcohol. This compound serves as the pivotal precursor for Resmethrin, a potent insecticide used extensively in agricultural and public health applications. The disclosed technology represents a significant departure from legacy manufacturing protocols, addressing long-standing challenges related to safety, yield, and operational complexity. By leveraging a strategic sequence of protection, organometallic addition, and catalytic hydrogenolysis, this innovation offers a pathway that is not only chemically elegant but also commercially viable for multi-ton production. For procurement leaders and technical directors seeking a reliable agrochemical intermediate supplier, understanding the nuances of this patent is essential for securing a competitive supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
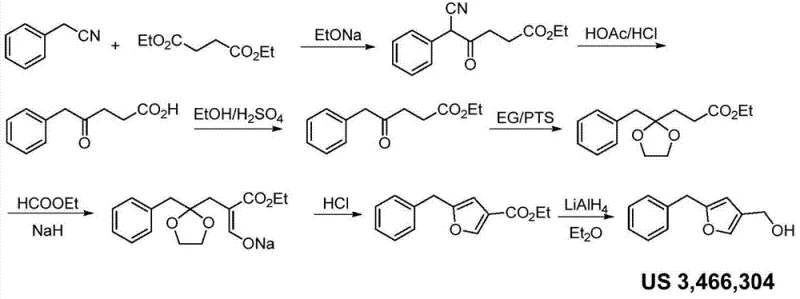
Historically, the industrial synthesis of 2-benzylfuran-4-methanol has been plagued by inefficient and hazardous chemical transformations. The foundational process described in US Patent 3,466,304, while effective in a laboratory setting, presents severe drawbacks for modern large-scale operations. This legacy route necessitates a Claisen condensation followed by a series of hydrolysis, esterification, and protection steps, culminating in a reduction using lithium aluminum hydride (LiAlH4). The reliance on LiAlH4 is particularly problematic due to its extreme reactivity with moisture, requiring rigorously anhydrous solvents and specialized handling equipment to prevent catastrophic safety incidents. Furthermore, the sheer number of synthetic steps involved in this traditional approach inherently accumulates impurities and reduces overall throughput, leading to substantial waste generation and elevated production costs.
Alternative historical methods, such as those detailed in EP 0,187,345 and WO 2002090341A1, attempt to bypass the LiAlH4 reduction but introduce their own set of logistical bottlenecks. These routes often depend on obscure starting materials like dichloroisobutene or require complex condensation reactions with aldoximes that suffer from poor regioselectivity and low yields. The difficulty in sourcing high-purity precursors for these older methods creates supply chain vulnerabilities, making it challenging for manufacturers to guarantee consistent delivery schedules. Additionally, the reaction conditions in these prior art processes often involve harsh temperatures or pressures that are difficult to maintain uniformly in large industrial reactors, further compromising the reliability of commercial scale-up of complex agrochemical intermediates.
The Novel Approach
In stark contrast to the convoluted pathways of the past, the method disclosed in CN103880791A streamlines the synthesis into a logical, three-stage sequence that prioritizes safety and efficiency. The core innovation lies in the initial protection of the hydroxymethyl group on the furan ring, which effectively masks the reactive alcohol functionality before the introduction of the phenyl group. This strategic move allows for the subsequent use of robust organometallic reagents, such as phenyl Grignard or phenyllithium, without the risk of unwanted side reactions at the hydroxyl site. The final stage combines deprotection and hydrogenolysis, potentially in a single operational step, utilizing safe catalytic systems like Pd/C or copper chromate under hydrogen pressure. This elimination of stoichiometric metal hydrides drastically simplifies the workup procedures and waste treatment requirements.

The versatility of this new approach is further enhanced by the flexibility in choosing protecting groups, such as tetrahydropyranyl (THP) ethers or benzyl ethers, allowing manufacturers to optimize based on local raw material availability and cost structures. The reaction conditions are mild, typically proceeding at temperatures between 20°C and 60°C for the addition steps, which reduces energy consumption compared to the high-temperature cyclizations of previous methods. By focusing on cost reduction in agrochemical intermediate manufacturing through step economy and safer reagents, this patent provides a blueprint for a more sustainable and economically attractive production model. The ability to achieve high yields with commercially available reagents ensures that the supply of this critical intermediate can be scaled rapidly to meet market demands without the technical barriers associated with legacy technologies.
Mechanistic Insights into Protective Group Strategy and Catalytic Hydrogenolysis
The success of this synthesis hinges on the precise manipulation of functional group reactivity, specifically the differential behavior of the hydroxymethyl groups on the furan ring. The inventors discovered that the position of the hydroxymethyl group significantly influences its susceptibility to hydrogenolysis; the alpha-position is readily cleaved to a methyl group, while the beta-position remains stable under specific conditions. However, to ensure absolute selectivity during the Grignard addition, the primary alcohol at the 4-position is first converted into an ether. When using dihydropyran (DHP) under acid catalysis, a tetrahydropyranyl ether is formed, which is stable to the basic conditions of the subsequent organometallic reaction. Alternatively, benzylation using benzyl chloride and a base like potassium carbonate provides a robust protecting group that can later be removed via hydrogenolysis. This dual-functionality of the benzyl group—acting as both a protector and a leaving group in the final step—exemplifies the atom-economic design of the process.
Following the formation of the secondary alcohol via nucleophilic attack of the phenyl anion on the aldehyde, the final transformation involves a sophisticated cascade of deprotection and reduction. If a THP group is used, mild acid hydrolysis restores the free alcohol, which is then subjected to hydrogenolysis to convert the adjacent benzylic alcohol into a methylene group, yielding the 2-benzyl motif. If a benzyl protecting group is employed, the process can be telescoped; catalytic hydrogenation simultaneously cleaves the benzyl ether and reduces the benzylic alcohol to the desired methyl linkage. The use of heterogeneous catalysts such as Pd/C or copper chromite allows for easy filtration and recycling, minimizing metal contamination in the final product. This mechanistic clarity ensures high-purity agrochemical intermediates that meet the stringent specifications required for downstream pesticide formulation.
How to Synthesize 2-Benzylfuran-4-Methanol Efficiently
The practical implementation of this synthesis route is designed for seamless integration into existing fine chemical manufacturing facilities. The process begins with the protection of 4-hydroxymethylfuran-2-carbaldehyde, followed by the addition of a phenyl nucleophile, and concludes with a catalytic finishing step. Detailed operational parameters, including solvent choices like THF or toluene and specific temperature ranges for each stage, are critical for maximizing yield and minimizing byproduct formation. For technical teams looking to adopt this methodology, the following guide outlines the standardized workflow derived from the patent examples, ensuring reproducibility and safety at scale.
- React 4-hydroxymethylfuran-2-carbaldehyde with a hydroxyl protecting reagent such as dihydropyran or benzyl halide to form the protected aldehyde intermediate.
- Perform a nucleophilic addition using a phenyl carbanion reagent, specifically phenylmagnesium bromide or phenyllithium, to the protected aldehyde to generate the secondary alcohol structure.
- Execute a final transformation involving the removal of the hydroxyl protecting group followed by catalytic hydrogenolysis to yield the target 2-benzylfuran-4-methanol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the CN103880791A synthesis route offers tangible strategic benefits beyond mere chemical curiosity. The shift away from hazardous reagents like lithium aluminum hydride eliminates the need for specialized storage and handling protocols, thereby reducing insurance premiums and operational overheads associated with safety compliance. Furthermore, the reliance on commodity chemicals such as benzyl chloride, dihydropyran, and phenylmagnesium bromide ensures that the raw material supply chain is resilient against market fluctuations. Unlike proprietary precursors required by older patents, these inputs are sourced from a broad base of global suppliers, mitigating the risk of single-source dependency and enhancing negotiation leverage for reducing lead time for high-purity agrochemical intermediates.
- Cost Reduction in Manufacturing: The streamlined nature of this process directly translates to lower manufacturing costs through significant step reduction. By avoiding the multi-step cyclization and esterification sequences of the US 3,466,304 route, the new method reduces solvent consumption, energy usage, and labor hours per kilogram of product. The elimination of expensive and dangerous stoichiometric reducing agents in favor of catalytic hydrogenation further drives down the variable cost of goods sold. Additionally, the higher selectivity of the reaction minimizes the formation of difficult-to-remove impurities, reducing the burden on purification units and increasing the overall recovery of saleable material.
- Enhanced Supply Chain Reliability: The use of robust, well-understood chemical transformations enhances the predictability of production campaigns. Traditional routes often suffer from batch-to-batch variability due to the sensitivity of reagents like LiAlH4 to trace moisture, leading to failed batches and supply disruptions. The new protocol operates under more forgiving conditions, ensuring consistent output quality and volume. This reliability is crucial for maintaining uninterrupted production of the final insecticide, Resmethrin, allowing downstream formulators to plan their inventory with greater confidence and security.
- Scalability and Environmental Compliance: From an environmental perspective, this method aligns with modern green chemistry principles by reducing waste generation and avoiding toxic heavy metal residues. The catalytic nature of the final step means that metal loading is minimal compared to stoichiometric reductions, simplifying wastewater treatment and effluent management. The process is inherently scalable, having been designed with industrial reactors in mind, facilitating a smooth transition from pilot plant to commercial tonnage without the need for extensive re-engineering of the process infrastructure.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating this technology, we have compiled answers to common inquiries regarding the synthesis and application of 2-benzylfuran-4-methanol. These responses are grounded in the specific technical disclosures of the patent, addressing concerns about safety, purity, and scalability. Understanding these details is vital for integrating this intermediate into your broader chemical portfolio and ensuring compliance with internal quality standards.
Q: Why is the new synthesis method for 2-benzylfuran-4-methanol superior to the US 3,466,304 process?
A: The conventional US 3,466,304 process relies on hazardous lithium aluminum hydride reductions and requires strictly anhydrous conditions across multiple cumbersome steps. The novel method utilizes safer catalytic hydrogenolysis and commercially available Grignard reagents, significantly simplifying the operational complexity and enhancing industrial safety profiles.
Q: What are the key raw materials required for this scalable production route?
A: The process initiates with 4-hydroxymethylfuran-2-carbaldehyde, a readily accessible furan derivative. Subsequent steps utilize standard protecting agents like dihydropyran or benzyl chloride, and common organometallic reagents such as phenylmagnesium bromide, ensuring a robust and reliable supply chain for large-scale manufacturing.
Q: How does this method address impurity control in agrochemical intermediate manufacturing?
A: By employing specific protective group strategies on the hydroxymethyl functionality prior to the Grignard addition, the method prevents side reactions at the alcohol position. This selectivity, combined with the high specificity of catalytic hydrogenolysis, results in intermediates with superior purity profiles compared to older cyclization-based routes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Benzylfuran-4-Methanol Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthesis routes requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN103880791A are fully realized in practice. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 2-benzylfuran-4-methanol meets the exacting requirements of the agrochemical industry. Our commitment to quality assurance ensures that the intermediates we supply contribute to the efficacy and safety of the final pesticide products.
We invite you to collaborate with us to optimize your supply chain for Resmethrin and related pyrethroids. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. We encourage you to contact us directly to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data and expert insight. Together, we can build a more efficient, safe, and sustainable supply chain for the next generation of crop protection solutions.
