Advanced Synthesis of Tri-Substituted Phenyl Compounds for Commercial PDE IV Inhibitor Production
Advanced Synthesis of Tri-Substituted Phenyl Compounds for Commercial PDE IV Inhibitor Production
The pharmaceutical landscape for treating inflammatory conditions such as rheumatic arthritis and allergic asthma is increasingly dependent on the efficient supply of high-purity Phosphodiesterase IV (PDE IV) inhibitors. Patent CN1166626C, granted in 2004, discloses a robust and versatile synthetic methodology for producing tri-substituted and tetra-substituted phenyl compounds that serve as potent PDE IV depressants. This intellectual property outlines a strategic approach to constructing complex benzamide scaffolds, specifically targeting N-(dihalophenyl or heterocyclic)-3-alkoxy-4-alkyl benzamides. For R&D directors and procurement specialists, understanding the nuances of this patent is critical, as it offers a pathway to bypass traditional bottlenecks associated with regioselective substitution on aromatic rings. The disclosed method leverages a sequence of esterification, nitration, amidation, and a unique diazotization-hydrolysis protocol to install oxygen functionalities with high precision. By analyzing the technical depth of CN1166626C, we can identify significant opportunities for optimizing the supply chain of these critical anti-inflammatory intermediates, ensuring both cost-effectiveness and regulatory compliance in the final drug substance manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for generating 3-alkoxy-4-alkyl benzamides often suffer from significant inefficiencies that impact both yield and environmental footprint. Prior art, such as the methods referenced in US5935978, frequently relies on the oxidation of benzaldehyde derivatives using strong oxidizing agents like potassium persulfate to generate the corresponding benzoic acids. This oxidation step is notoriously difficult to control on a large scale, often leading to over-oxidation byproducts and requiring rigorous purification steps to remove metal residues. Furthermore, conventional approaches to introducing the 3-alkoxy group typically involve nucleophilic substitution on pre-functionalized halobenzenes, which can result in poor regioselectivity and the formation of difficult-to-separate isomers. The reliance on harsh conditions and multiple purification stages not only drives up the cost of goods sold (COGS) but also complicates the waste management profile, creating substantial hurdles for supply chain managers aiming for green chemistry compliance. Additionally, the use of sensitive reagents in earlier steps can limit the scope of compatible functional groups, restricting the diversity of analogs that can be efficiently produced for structure-activity relationship (SAR) studies.
The Novel Approach
In stark contrast, the methodology detailed in CN1166626C introduces a paradigm shift by reversing the order of functional group installation to maximize efficiency and selectivity. The novel approach initiates with the esterification of readily available para-alkyl or alkoxy benzoic acids, followed by a highly regioselective nitration to secure the 3-nitro-4-substituted benzoate intermediate. This sequence ensures that the electron-donating groups direct the nitro group precisely to the desired position, minimizing isomeric impurities from the outset. A key innovation lies in the conversion of the nitro group to a hydroxyl group via a reduction-diazotization-hydrolysis cascade, rather than direct oxidation. This transformation allows for the mild introduction of the oxygen functionality under controlled acidic conditions, significantly reducing the risk of side reactions. Subsequent alkylation of the phenolic intermediate with haloalkanes under basic catalysis completes the scaffold construction. This route is characterized by its operational simplicity, where intermediate isolation often requires only basic aqueous washing and drying, eliminating the need for expensive column chromatography and enabling a streamlined flow suitable for continuous manufacturing environments.
Mechanistic Insights into Diazotization-Hydrolysis and Regioselective Nitration
The core chemical elegance of this patent lies in its masterful manipulation of electrophilic aromatic substitution and diazonium chemistry to achieve precise substitution patterns. The process begins with the nitration of a para-substituted benzoate, where the existing alkoxy or alkyl group acts as an ortho/para director. However, since the para position is occupied, the incoming nitro group is directed exclusively to the ortho position relative to the donor, which corresponds to the meta position relative to the ester group. This inherent electronic bias ensures high regioselectivity, a critical factor for maintaining high purity in the final active pharmaceutical ingredient (API). Following amidation with dihalo-anilines or heterocyclic amines, the nitro group serves as a masked precursor for the hydroxyl functionality. The reduction of the nitro group to an amine, typically using iron powder and hydrochloric acid or catalytic hydrogenation, sets the stage for the diazotization step. In this crucial transformation, the aromatic amine reacts with sodium nitrite under acidic conditions to form a diazonium salt, which is subsequently hydrolyzed upon heating in dilute acid to yield the phenol. This mechanism effectively swaps a nitrogen substituent for an oxygen substituent with retention of the aromatic core integrity.
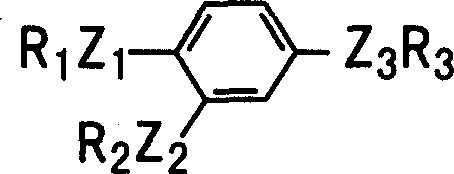
Understanding the structural versatility depicted in the general formula is essential for R&D teams exploring analog libraries. The patent defines a broad scope where R1 represents low alkyl groups, while R2 and R3 can vary from cycloalkyl to heteroaryl moieties, connected via diverse linkers such as ether, thioether, or amine bridges. The ability to tolerate dihalo-substituted pyridines and phenyl rings in the amide portion (Z3R3) highlights the robustness of the coupling conditions. Mechanistically, the final alkylation step involves the deprotonation of the newly formed phenol to generate a phenoxide ion, which then attacks the alkyl halide in an SN2 reaction. The choice of base, such as anhydrous potassium carbonate or sodium hydride, and the solvent system is critical to driving this reaction to completion without hydrolyzing the sensitive amide bond. This careful balance of reactivity ensures that the final tri-substituted phenyl compound retains its structural fidelity, providing a reliable foundation for biological activity as a PDE IV inhibitor. The detailed control over each mechanistic step translates directly into a cleaner impurity profile, reducing the burden on downstream purification processes.
How to Synthesize N-(Dihalophenyl)-3-alkoxy-4-alkyl Benzamides Efficiently
Implementing this synthesis route requires a disciplined approach to reaction monitoring and parameter control to ensure reproducibility at scale. The process generally follows a linear progression starting from commodity chemicals like p-toluic acid or anisic acid, making raw material sourcing straightforward and cost-effective. The initial esterification and nitration steps are exothermic and require careful temperature management, typically keeping the nitration mixture below 0°C to prevent dinitration or oxidative degradation. Following the formation of the nitro-benzoate, the amidation with dihalo-anilines is conducted in alcoholic solvents, where the nucleophilicity of the amine is sufficient to displace the ester alkoxy group without the need for activating the acid chloride, although acid chloride pathways are also disclosed as alternatives. The subsequent reduction and diazotization steps demand strict pH control and stoichiometric precision to avoid the accumulation of hazardous diazonium species. Finally, the alkylation is performed under reflux conditions to ensure complete conversion of the phenolic intermediate. Detailed standardized operating procedures for each of these unit operations are essential for technology transfer, ensuring that the high yields reported in the patent examples (often exceeding 80%) are maintained in commercial production.
- Esterify para-alkyl or alkoxy benzoic acid followed by nitration to obtain 3-nitro-4-substituted benzoates.
- React the nitro-benzoate with dihalogenated aniline or heterocyclic amine to form the amide linkage.
- Reduce the nitro group to an amine, perform diazotization and hydrolysis to introduce the hydroxyl group, and finally alkylate to achieve the target ether structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the synthesis route described in CN1166626C presents a compelling value proposition centered on cost reduction and supply reliability. The primary economic driver is the utilization of inexpensive, commodity-grade starting materials such as substituted benzoic acids and simple haloalkanes, which are widely available in the global chemical market. Unlike routes that require specialized, high-cost building blocks or precious metal catalysts, this method relies on base metals like iron for reduction and common mineral acids for catalysis, drastically lowering the raw material cost baseline. Furthermore, the process design inherently minimizes waste generation; the workup procedures predominantly involve aqueous washes and crystallization, avoiding the extensive use of organic solvents and silica gel associated with chromatographic purification. This reduction in solvent consumption not only lowers disposal costs but also aligns with increasingly stringent environmental regulations, mitigating the risk of production shutdowns due to compliance issues. The robustness of the chemistry also implies a lower rate of batch failures, ensuring a consistent flow of intermediates to the formulation stage.
- Cost Reduction in Manufacturing: The elimination of expensive oxidizing agents and transition metal catalysts significantly reduces the direct material costs associated with the synthesis. By replacing complex oxidation steps with a diazotization-hydrolysis sequence, the process avoids the need for specialized equipment resistant to highly corrosive oxidants, thereby lowering capital expenditure requirements. Additionally, the high regioselectivity of the nitration step minimizes the formation of isomeric byproducts, which reduces the yield loss typically incurred during purification. The ability to isolate intermediates through simple crystallization or filtration rather than distillation or chromatography further drives down energy consumption and labor costs. These cumulative efficiencies translate into a substantially lower cost of goods sold, providing a competitive margin advantage in the pricing of the final PDE IV inhibitor API.
- Enhanced Supply Chain Reliability: The reliance on widely available bulk chemicals ensures that the supply chain is resilient to market fluctuations and geopolitical disruptions. Since the synthesis does not depend on single-source specialty reagents or fragile biological catalysts, procurement teams can easily qualify multiple vendors for raw materials, securing a stable supply line. The simplicity of the reaction conditions also means that the process can be manufactured in a broader range of facilities, including those with standard glass-lined steel reactors, increasing the available manufacturing capacity. This flexibility allows for rapid scaling in response to demand surges without the long lead times associated with installing specialized catalytic infrastructure. Consequently, the risk of supply interruptions is minimized, guaranteeing continuity for downstream drug product manufacturing schedules.
- Scalability and Environmental Compliance: The process is inherently scalable due to its reliance on well-understood unit operations such as nitration, amidation, and alkylation, which are staples of the fine chemical industry. The thermal profiles of these reactions are manageable, allowing for safe scale-up from pilot plants to multi-ton commercial reactors with predictable heat transfer characteristics. From an environmental perspective, the avoidance of heavy metal catalysts simplifies the wastewater treatment process, as there is no need for complex metal scavenging or recovery systems. The reduced solvent intensity and the potential for solvent recycling further enhance the sustainability profile of the manufacturing process. This alignment with green chemistry principles not only reduces regulatory burdens but also enhances the corporate social responsibility standing of the manufacturing partner, making it a preferred choice for environmentally conscious pharmaceutical clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and claims within CN1166626C, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of adopting this technology for their specific portfolio needs. The answers reflect the practical realities of scaling organic synthesis while maintaining high quality standards.
Q: What are the key advantages of the synthesis route described in CN1166626C?
A: The patented route offers significant operational simplicity by utilizing standard unit operations like esterification and nitration with easy aqueous workups, eliminating the need for complex chromatographic purification and reducing overall production costs.
Q: How does this method improve impurity control compared to conventional oxidation routes?
A: By employing a diazotization-hydrolysis sequence to install the hydroxyl group rather than direct oxidation of aldehydes, the process minimizes over-oxidation byproducts and ensures higher regioselectivity for the 3-position substitution.
Q: Is this synthesis scalable for commercial manufacturing of PDE IV inhibitors?
A: Yes, the process relies on robust, exothermic reactions like nitration and amidation that are well-understood in chemical engineering, allowing for safe scale-up from kilogram to multi-ton production with consistent yield profiles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tri-substituted Phenyl Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in the development of next-generation anti-inflammatory therapeutics. Our technical team has extensively analyzed the methodology presented in CN1166626C and possesses the expertise to adapt and optimize this chemistry for large-scale production. We understand that transitioning from laboratory benchtop to commercial manufacturing requires more than just following a recipe; it demands a deep understanding of process safety, impurity control, and regulatory requirements. With our state-of-the-art facilities, we offer extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our commitment to quality is underscored by our stringent purity specifications and rigorous QC labs, which utilize advanced analytical techniques to verify the identity and purity of every batch of PDE IV inhibitor intermediates we produce.
We invite you to collaborate with us to leverage this innovative synthesis technology for your drug development programs. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. By partnering with NINGBO INNO PHARMCHEM, you gain access to a reliable supply chain partner dedicated to accelerating your time-to-market. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our manufacturing capabilities can support your strategic goals in the pharmaceutical sector.
