Advanced Continuous Synthesis of Aminoacetaldehyde Dimethyl Acetal for Industrial Pharmaceutical Applications
Advanced Continuous Synthesis of Aminoacetaldehyde Dimethyl Acetal for Industrial Pharmaceutical Applications
The pharmaceutical industry constantly seeks more efficient pathways for synthesizing critical intermediates used in life-saving medications. Patent CN111875510B introduces a groundbreaking methodology for the preparation of aminoacetaldehyde dimethyl acetal, a vital building block in the synthesis of complex heterocycles found in antiretroviral therapies such as Dolutegravir and anthelmintic agents like Praziquantel. This innovative process addresses long-standing inefficiencies in traditional manufacturing by integrating chlorination, acetalization, and ammonolysis into a streamlined, continuous workflow. By leveraging phase transfer catalysis and controlled pressure reactions, the technology enables the production of high-purity intermediates while drastically minimizing environmental impact and operational costs. The structural integrity and chemical versatility of this compound make it indispensable for modern medicinal chemistry.
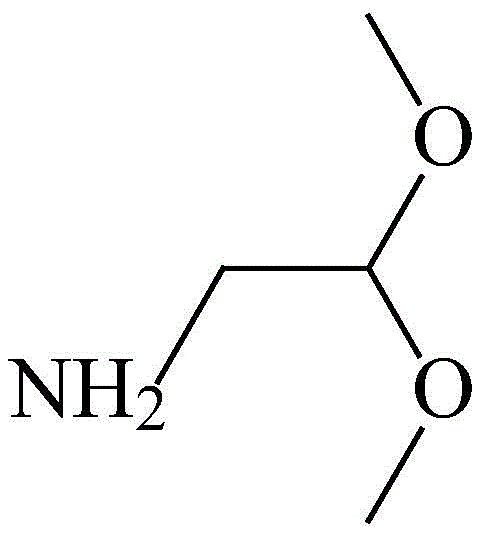
For procurement specialists and supply chain directors, the implications of this patent extend beyond mere chemical novelty; it represents a strategic opportunity to secure a reliable aminoacetaldehyde dimethyl acetal supplier capable of meeting rigorous quality standards. The shift from multi-step batch processing to a more integrated approach reduces the dependency on scarce, high-purity starting materials that often bottleneck production schedules. Furthermore, the ability to synthesize this compound with a total yield exceeding 80 percent and purity greater than 98 percent ensures that downstream API manufacturers receive materials that require minimal additional refinement. This technical advancement aligns perfectly with the global push towards greener chemistry and sustainable manufacturing practices in the fine chemical sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of aminoacetaldehyde dimethyl acetal has been plagued by significant operational inefficiencies and environmental burdens. Traditional routes typically rely on the direct reaction of high-purity chloroacetaldehyde dimethyl acetal with aqueous ammonia, a process that necessitates extensive post-treatment procedures including deamination, dehydration, neutralization, and multiple distillation steps. These legacy methods are not only energy-intensive due to the requirement of distilling large volumes of water but also generate substantial amounts of solid waste and wastewater, creating severe disposal challenges for chemical plants. Moreover, the reliance on pre-purified chloroacetaldehyde dimethyl acetal as a starting material inflates raw material costs and introduces supply chain vulnerabilities, as the synthesis of this precursor itself is often fraught with side reactions and low yields when performed via older acetaldehyde-based chlorination techniques.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN111875510B revolutionizes the synthesis landscape by adopting a continuous, one-pot strategy that bypasses the isolation of unstable intermediates. By initiating the reaction with readily available vinyl acetate and employing a phase transfer catalyst to facilitate chlorination, the process achieves superior control over reaction kinetics and selectivity. The subsequent in-situ conversion of the chlorinated species into the acetal, followed immediately by ammonolysis in a closed autoclave system, eliminates the need for intermediate purification and the associated solvent losses. This integrated approach not only simplifies the operational workflow but also significantly mitigates the generation of hazardous by-products, thereby offering a robust solution for cost reduction in pharmaceutical intermediate manufacturing while ensuring consistent product quality suitable for sensitive downstream applications.
Mechanistic Insights into Phase Transfer Catalyzed Chlorination and Ammonolysis
The core of this technological breakthrough lies in the sophisticated application of phase transfer catalysis (PTC) during the initial chlorination of vinyl acetate. In this step, a quaternary ammonium salt, such as tetrabutylammonium bromide or benzyltriethylammonium chloride, acts as a molecular shuttle, transporting reactive chloride species into the organic phase where they can efficiently interact with the vinyl double bond. This mechanism allows the reaction to proceed smoothly at mild temperatures between 5°C and 15°C, preventing the thermal degradation of the sensitive vinyl group and minimizing the formation of polychlorinated impurities. The precise control of chlorine introduction ensures that the electrophilic addition occurs selectively, generating a chlorinated solution that serves as the direct precursor for the subsequent acetalization step without requiring isolation.
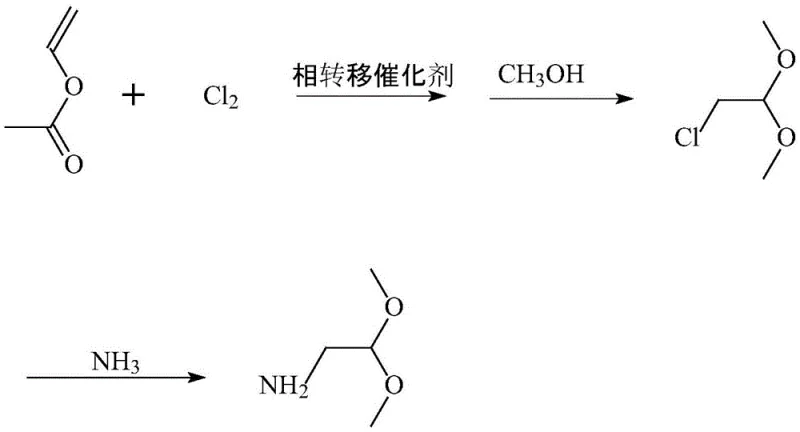
Following the formation of the chloro-intermediate, the process transitions into a high-pressure ammonolysis phase within an autoclave, where the crude chloroacetaldehyde dimethyl acetal reacts with liquid ammonia. This nucleophilic substitution is driven by elevated temperatures of 70°C to 100°C and pressures ranging from 2.0 to 3.0 MPa, conditions that favor the displacement of the chloride atom by the amino group while maintaining the stability of the acetal protecting groups. The final purification stage involves a carefully orchestrated vacuum distillation protocol, where methanol and excess ammonia are recovered at lower temperatures before the target product is distilled at 70°C to 90°C under high vacuum. This rigorous separation technique is critical for impurity control, ensuring that the final aminoacetaldehyde dimethyl acetal meets the stringent purity specifications required for the synthesis of complex active pharmaceutical ingredients.
How to Synthesize Aminoacetaldehyde Dimethyl Acetal Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for scaling this reaction from laboratory benchtop to industrial production vessels. The process begins with the careful addition of a phase transfer catalyst to vinyl acetate under cooling, followed by the controlled introduction of chlorine gas to generate the reactive chlorinated species. This solution is then immediately transferred into anhydrous methanol to form the acetal intermediate, which is subsequently pumped into a pressure reactor for ammonolysis. The detailed standardized synthesis steps below outline the specific molar ratios, temperature gradients, and pressure settings required to replicate the high yields and purity reported in the patent documentation.
- Chlorinate vinyl acetate using chlorine gas and a quaternary ammonium phase transfer catalyst at 5-15°C to form a chlorinated solution.
- React the chlorinated solution with anhydrous methanol at 0-15°C to generate crude chloroacetaldehyde dimethyl acetal without isolation.
- Transfer the crude product to an autoclave, react with liquid ammonia at 70-100°C and 2.0-3.0 MPa, then adjust pH and distill to obtain the final high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers transformative benefits that directly impact the bottom line and operational resilience. By eliminating the need to purchase or separately synthesize high-purity chloroacetaldehyde dimethyl acetal, manufacturers can drastically simplify their raw material sourcing strategies and reduce exposure to price volatility in the specialty chemical market. The continuous nature of the process means that production cycles are shorter and more predictable, allowing for better inventory management and faster response times to fluctuating market demands for antiretroviral and anthelmintic drug precursors. Furthermore, the reduction in waste generation translates to lower environmental compliance costs and a smaller carbon footprint, aligning corporate sustainability goals with economic efficiency.
- Cost Reduction in Manufacturing: The elimination of intermediate isolation and purification steps results in substantial savings in both energy consumption and solvent usage. By avoiding the energy-intensive distillation of water associated with aqueous ammonia methods and reducing the number of unit operations, the overall operational expenditure is significantly lowered. Additionally, the use of inexpensive and widely available raw materials like vinyl acetate and methanol further drives down the variable cost per kilogram of the final product, enhancing profit margins for large-scale producers.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals such as vinyl acetate and chlorine gas rather than specialized, high-purity intermediates creates a more robust and resilient supply chain. This diversification of raw material sources reduces the risk of production stoppages caused by shortages of niche reagents. The simplified workflow also decreases the complexity of logistics and storage requirements, ensuring a steady and uninterrupted flow of high-quality aminoacetaldehyde dimethyl acetal to downstream API manufacturers.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing standard industrial equipment such as autoclaves and distillation columns that are readily available in most fine chemical facilities. The significant reduction in three wastes (wastewater, waste gas, and solid waste) simplifies effluent treatment processes and ensures compliance with increasingly strict environmental regulations. This eco-friendly profile not only mitigates regulatory risks but also enhances the brand reputation of the manufacturer as a leader in sustainable chemical production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in patent CN111875510B, providing clarity on how this method outperforms legacy techniques in terms of yield, purity, and operational safety. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for commercial production.
Q: What are the primary advantages of this new synthesis route over conventional methods?
A: The patented method eliminates the need for isolating and purifying the intermediate chloroacetaldehyde dimethyl acetal, significantly reducing energy consumption and waste generation compared to traditional batch processes that require separate dehydration and neutralization steps.
Q: How does the process ensure high purity (>98%) of the final API intermediate?
A: High purity is achieved through precise temperature control during the exothermic chlorination step (5-15°C) and a rigorous vacuum distillation protocol at 70-90°C under high vacuum (<-0.09 MPa) which effectively separates the target amine from residual methanol and ammonia.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the process is specifically designed for industrial scalability, utilizing standard autoclave equipment for the ammonolysis step and achieving total yields exceeding 80%, making it economically viable for mass production of antiretroviral drug precursors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aminoacetaldehyde Dimethyl Acetal Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volumetric demands of global pharmaceutical partners. We are committed to delivering aminoacetaldehyde dimethyl acetal with stringent purity specifications, supported by our rigorous QC labs that employ advanced analytical techniques to verify every batch against the highest industry standards. Our dedication to process optimization allows us to offer competitive pricing without compromising on the quality or consistency that your R&D and production teams depend on.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific project needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can accelerate your drug development timeline and optimize your supply chain efficiency.
