Advanced Aqueous Synthesis of 2-Aminothiazole-4-ylacetic Acid Hydrochloride for Cephalosporins
Advanced Aqueous Synthesis of 2-Aminothiazole-4-ylacetic Acid Hydrochloride for Cephalosporins
The pharmaceutical industry constantly seeks more efficient and environmentally benign pathways for producing critical antibiotic intermediates. Patent CN101948446B introduces a groundbreaking method for preparing 2-aminothiazol-4-ylacetic acid hydrochloride, a pivotal building block for third-generation cephalosporins like Cefotiam. This innovation addresses long-standing challenges in the synthesis of thiazole derivatives by shifting from hazardous organic solvents to a safer, aqueous-based system. By utilizing thiourea and alpha-halo ethyl acetoacetate as primary raw materials, the process achieves high purity and yield under remarkably mild conditions. For R&D directors and procurement specialists, this represents a significant opportunity to optimize the supply chain for high-purity pharmaceutical intermediates while adhering to stricter environmental regulations.
The Limitations of Conventional Methods vs. The Novel Approach
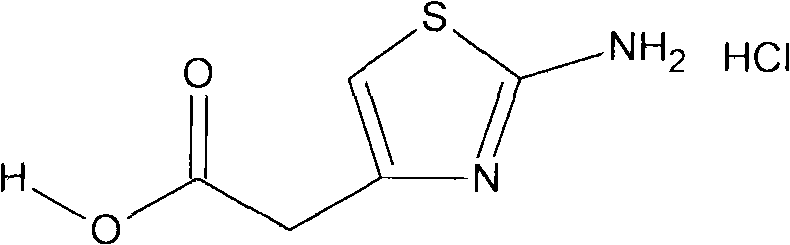
The Limitations of Conventional Methods
Historically, the synthesis of thiazole compounds has relied on cumbersome multi-step procedures that pose significant operational and environmental burdens. Traditional routes typically involve a three-step sequence where thiourea and gamma-halogenated acetoacetate undergo direct cyclization to form an ester, followed by alkaline saponification to generate a sodium salt, and finally acidification to yield the target acid. This legacy approach necessitates the use of volatile organic solvents and requires repeated acid-base treatments for purification, often involving complex techniques like capillary electrophoresis which are ill-suited for large-scale industrial application. Furthermore, the extensive post-treatment required to isolate the product generates substantial chemical waste, leading to high disposal costs and potential regulatory compliance issues for manufacturers aiming for green chemistry standards.
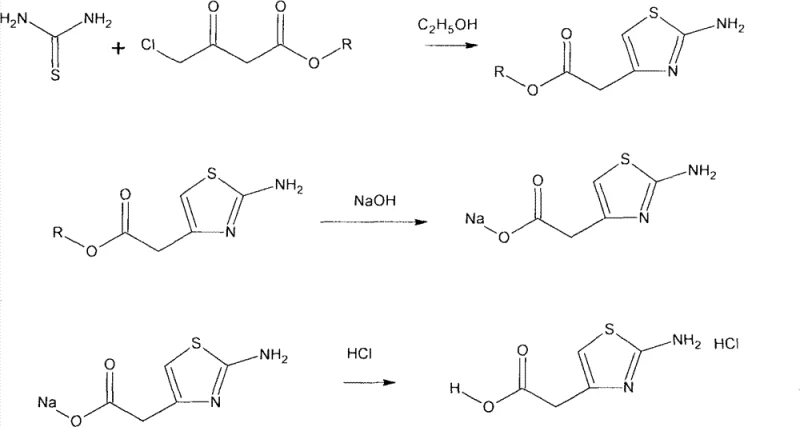
The Novel Approach
In stark contrast, the methodology disclosed in the patent streamlines production into a highly efficient two-step process conducted primarily in an aqueous phase. The first step involves the reaction of 4-chloro ethyl acetoacetate with thiourea in water, followed by a direct suspension of the resulting intermediate in cold concentrated hydrochloric acid for the second step. This elimination of the separate saponification step not only reduces the number of unit operations but also removes the need for expensive organic solvents and complex purification protocols. The result is a robust manufacturing route that delivers the target hydrochloride salt with exceptional purity and yield, making it an ideal solution for cost reduction in pharmaceutical intermediate manufacturing without compromising on quality or safety.
Mechanistic Insights into Aqueous Phase Cyclization and Hydrolysis
The core of this technological advancement lies in the precise control of reaction conditions during the cyclization and hydrolysis phases. In the initial step, thiourea acts as a nucleophile attacking the alpha-carbon of the chloroacetoacetate derivative. By maintaining the reaction temperature strictly between -5°C and 3°C, specifically optimizing at 0-3°C, the process minimizes side reactions such as polymerization or degradation of the sensitive thiazole ring. The subsequent addition of ammonia to adjust the pH to a range of 7 to 10 facilitates the precipitation of the intermediate ester as white crystals, effectively separating it from the aqueous mother liquor without the need for extraction. This pH-controlled crystallization is a critical mechanism for impurity rejection, ensuring that the intermediate entering the second step is of high quality.
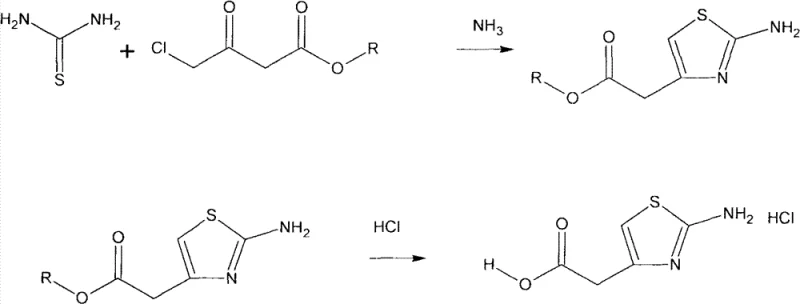
The second mechanistic phase involves the hydrolysis of the ester intermediate using cold concentrated hydrochloric acid. Suspending the white crystals in acid cooled to -5°C to 3°C prevents thermal shock and uncontrolled exothermic reactions. Upon heating the mixture to 50-60°C, the ester bond is cleaved, and the amino group is protonated to form the stable hydrochloride salt. This one-pot conversion from ester to acid hydrochloride avoids the isolation of the free acid, which can be unstable or difficult to handle. The rigorous temperature control throughout the 3 to 6-hour hydrolysis period ensures complete conversion while preventing the decomposition of the thiazole ring, thereby securing the high purity levels required for downstream antibiotic synthesis.
How to Synthesize 2-Aminothiazol-4-ylacetic Acid Hydrochloride Efficiently
Implementing this synthesis route requires careful attention to temperature gradients and reagent addition rates to maximize yield and safety. The process begins with the suspension of thiourea in water, followed by the controlled dropwise addition of the chloro-ester over a period of 2 to 3 hours. Maintaining the low temperature during this exothermic addition is paramount for controlling the reaction kinetics and preventing the formation of byproducts. Once the intermediate is isolated, it is immediately subjected to acid hydrolysis, where the concentration of hydrochloric acid and the heating profile determine the final crystal quality. For detailed operational parameters and standard operating procedures, please refer to the technical guide below which outlines the specific stoichiometry and timing derived from the patent examples.
- Suspend thiourea in water, cool to 0-3°C, and add 4-chloroacetoacetate dropwise while maintaining temperature, then adjust pH to 7-10 with ammonia to obtain the intermediate ester.
- Suspend the obtained white crystals in cold concentrated hydrochloric acid, heat to 50-60°C for hydrolysis, then cool and filter to isolate the final hydrochloride salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the shift to this aqueous-based synthesis offers profound strategic advantages beyond mere technical feasibility. The replacement of organic solvents with water fundamentally alters the cost structure of production by eliminating solvent purchase, recovery, and disposal expenses. Additionally, the simplification of the workflow from three steps to two reduces labor hours, equipment occupancy time, and energy consumption associated with distillation and drying processes. These factors collectively contribute to a more resilient and cost-effective supply chain, ensuring that critical cephalosporin intermediates remain available even during periods of raw material volatility or regulatory tightening on chemical emissions.
- Cost Reduction in Manufacturing: The elimination of organic solvents and the reduction of processing steps lead to substantial cost savings in raw material acquisition and waste management. By avoiding the use of expensive reagents for saponification and the subsequent acidification steps found in older methods, the overall variable cost per kilogram of product is drastically lowered. Furthermore, the high yield reported in the patent examples, consistently reaching roughly 92%, means less raw material is wasted, directly improving the gross margin for manufacturers adopting this technology.
- Enhanced Supply Chain Reliability: The starting materials, specifically thiourea and 4-chloroacetoacetate, are commodity chemicals that are widely available from multiple global suppliers, reducing the risk of single-source dependency. The robustness of the aqueous process also means that production is less susceptible to disruptions caused by solvent shortages or logistics issues related to hazardous material transport. This reliability ensures a steady flow of high-purity intermediates to downstream API manufacturers, safeguarding the continuity of antibiotic production lines.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward due to the use of water as the primary medium, which simplifies heat transfer and mixing in large reactors. The significant reduction in hazardous waste generation aligns with increasingly stringent global environmental regulations, minimizing the risk of production shutdowns due to compliance violations. This environmental compatibility makes the facility more sustainable and future-proof, appealing to partners who prioritize green chemistry initiatives in their supplier selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. They are derived from a detailed analysis of the patent specifications and are designed to clarify the operational benefits and chemical principles involved. Understanding these aspects is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing infrastructure.
Q: Why is water preferred over organic solvents for this cyclization reaction?
A: Water is preferred because it significantly reduces environmental pollution and production costs compared to traditional organic solvents. The patent data indicates that using water allows for milder reaction conditions and simplifies the post-treatment process, eliminating the need for complex solvent recovery systems.
Q: What is the typical purity achieved with this novel two-step method?
A: The novel method described in the patent achieves exceptionally high purity, with HPLC analysis showing results around 99.4% to 99.6%. This high purity is critical for downstream cephalosporin synthesis, ensuring minimal impurity carryover into the final antibiotic product.
Q: How does this process improve upon the conventional three-step synthesis?
A: Conventional methods often require a three-step sequence involving saponification and multiple acid-base treatments in organic solvents, which is complex and polluting. This new approach condenses the process into two efficient steps in an aqueous phase, drastically simplifying operations and improving overall yield to approximately 92%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Aminothiazol-4-ylacetic Acid Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global pharmaceutical supply chain. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the demanding volume requirements of multinational drug developers. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify every batch against the highest industry standards. Our capability to implement advanced aqueous synthesis technologies allows us to offer competitive pricing without sacrificing the quality essential for cephalosporin manufacturing.
We invite you to collaborate with us to optimize your supply chain for cephalosporin antibiotics. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs and quality requirements. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can support your long-term strategic goals.
