Advanced Manufacturing of (S)-4-Phenyl-2-Oxazolidinone: A Green Route for Chiral Intermediates
Advanced Manufacturing of (S)-4-Phenyl-2-Oxazolidinone: A Green Route for Chiral Intermediates
The pharmaceutical and agrochemical industries are constantly seeking more efficient and safer pathways to produce high-value chiral intermediates. Patent CN112500361A introduces a significant technological breakthrough in the synthesis of (S)-4-phenyl-2-oxazolidinone, a critical chiral auxiliary widely used in asymmetric alkylation, aldol condensation, and conjugate addition reactions. This novel preparation method replaces hazardous traditional reagents with a streamlined borane reduction and base-catalyzed cyclization sequence, offering a robust solution for the production of complex organic amines. By shifting away from cytotoxic solvents and unstable oxidants, this technology aligns perfectly with modern green chemistry principles while maintaining exceptional product quality. For R&D directors and procurement specialists, understanding this shift is vital for optimizing supply chains and ensuring the consistent availability of high-purity building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of (S)-4-phenyl-2-oxazolidinone and its sulfur-containing derivatives has been plagued by significant safety and environmental challenges. Traditional routes often involve the reaction of L-phenylglycinol with carbon disulfide (CS2) under alkaline conditions, followed by oxidation with hydrogen peroxide. Carbon disulfide is notoriously hazardous; it is highly volatile, flammable, and possesses severe cytotoxicity, posing substantial risks to operator health and requiring expensive engineering controls for containment. Furthermore, the addition of hydrogen peroxide in these systems can lead to violent exothermic releases, creating unpredictable reaction profiles that are difficult to manage on a large scale. Alternative methods utilizing Lawesson's reagent for thionation are also problematic due to the high cost of the reagent and the generation of difficult-to-remove phosphorus byproducts. These factors collectively increase the cost of goods sold (COGS) and complicate waste treatment protocols, making conventional methods less attractive for modern sustainable manufacturing.
The Novel Approach
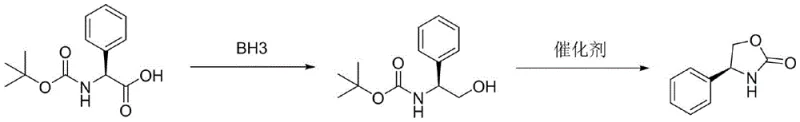
In stark contrast, the methodology disclosed in patent CN112500361A offers a remarkably cleaner and more controllable alternative. The process begins with the reduction of readily available N-Boc-L-phenylglycine using a borane reagent, such as borane-tetrahydrofuran complex, to generate N-Boc-L-phenylglycinol. This intermediate then undergoes a sophisticated ring-closing reaction catalyzed by alkoxides like potassium tert-butoxide or sodium tert-butoxide. This cascade effectively constructs the oxazolidinone core while eliminating the need for toxic carbon disulfide entirely. The reaction conditions are mild, typically proceeding at temperatures between 0°C and 50°C, which minimizes energy consumption and thermal stress on the equipment. By utilizing stable, commercially available starting materials and avoiding explosive or highly toxic reagents, this new approach drastically simplifies the operational workflow and enhances the overall safety profile of the manufacturing facility.
Mechanistic Insights into Borane Reduction and Base-Catalyzed Cyclization
The core of this synthetic innovation lies in the precise control of the reduction and subsequent cyclization steps. In the first stage, the carboxylic acid group of N-Boc-L-phenylglycine is selectively reduced to a primary alcohol by the borane reagent. Borane is a potent reducing agent that operates efficiently in ethers like tetrahydrofuran (THF) or 2-methyltetrahydrofuran (2-MeTHF), converting the acid to the corresponding amino alcohol derivative without affecting the stereochemistry at the chiral center. This preservation of chirality is paramount, as the resulting intermediate serves as the foundation for the optical purity of the final product. The reaction is typically quenched carefully with methanol to decompose excess borane species, ensuring a safe workup procedure before the isolation of the N-Boc-L-phenylglycinol intermediate.
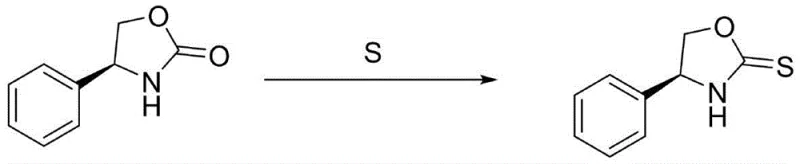
The second stage involves an elegant intramolecular cyclization driven by a strong base catalyst. When agents like potassium tert-butoxide are introduced, they facilitate the nucleophilic attack of the newly formed hydroxyl group onto the carbonyl carbon of the Boc protecting group. This transesterification-like process results in the formation of the five-membered oxazolidinone ring and the elimination of tert-butanol as a byproduct. The removal of tert-butanol, often achieved via distillation in optimized embodiments, drives the equilibrium towards the product, ensuring high conversion rates. This mechanism not only constructs the heterocyclic ring efficiently but also inherently purifies the system by volatilizing the byproduct. For the derivative (S)-4-phenyloxazolidine-2-thione, the process further employs sulfur powder combined with ammonium sulfide or polysulfide, providing a safe and effective thionation pathway that avoids the pitfalls of phosphorus-based reagents.
How to Synthesize (S)-4-Phenyl-2-Oxazolidinone Efficiently
Implementing this synthesis requires careful attention to stoichiometry and temperature control to maximize yield and purity. The process generally involves dissolving the starting amino acid derivative in a dry ether solvent and cooling the mixture before the slow addition of the borane complex. Following the reduction and workup, the crude amino alcohol is subjected to cyclization conditions using a catalytic amount of alkoxide base. Detailed standard operating procedures regarding specific molar ratios, solvent volumes, and crystallization techniques are essential for reproducibility. For a comprehensive guide on the exact experimental parameters and troubleshooting tips for this specific pathway, please refer to the standardized synthesis instructions below.
- Reduce N-Boc-L-phenylglycine using a borane reagent (such as BH3-THF) in an organic solvent like tetrahydrofuran at 0-25°C to obtain N-Boc-L-phenylglycinol.
- Add a catalyst such as potassium tert-butoxide or sodium tert-butoxide to the intermediate and heat the mixture to facilitate intramolecular cyclization.
- Isolate the final (S)-4-phenyl-2-oxazolidinone product through solvent evaporation, extraction, and recrystallization to achieve high purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented synthesis route offers compelling advantages for procurement managers and supply chain directors looking to optimize their sourcing strategies for chiral intermediates. The elimination of carbon disulfide and Lawesson's reagent removes significant cost drivers associated with hazardous material handling, specialized storage, and complex waste disposal. By switching to benign reagents like borane complexes and simple alkoxides, manufacturers can achieve substantial cost savings in both raw material procurement and environmental compliance. Furthermore, the use of common solvents such as THF and ethyl acetate ensures that the supply chain remains resilient, as these materials are widely available globally and less prone to logistical bottlenecks compared to specialty toxic reagents.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived primarily from the simplification of the reaction workflow and the avoidance of expensive proprietary reagents. Traditional methods relying on Lawesson's reagent incur high material costs and generate phosphorus waste that requires costly treatment. In contrast, the borane-mediated route utilizes cost-effective reagents and generates tert-butanol as a manageable byproduct. Additionally, the high purity achieved directly from recrystallization reduces the need for extensive chromatographic purification, thereby lowering solvent consumption and labor hours. This streamlined approach translates directly into a more competitive price point for the final API intermediate without compromising on quality standards.
- Enhanced Supply Chain Reliability: Supply chain stability is significantly improved by removing dependencies on highly regulated and dangerous chemicals. Carbon disulfide is subject to strict transportation and storage regulations due to its flammability and toxicity, which can lead to delays and increased logistics costs. By substituting this with stable borane solutions and solid sulfur powders, the manufacturing process becomes far more robust against regulatory changes and supply disruptions. The ability to source raw materials from multiple global suppliers ensures continuity of supply, allowing pharmaceutical companies to maintain consistent production schedules for their downstream drug candidates.
- Scalability and Environmental Compliance: The scalability of this process is a key asset for industrial expansion. The reaction conditions are mild and do not involve violent exotherms or high-pressure requirements, making the transition from pilot plant to commercial scale straightforward and safe. From an environmental standpoint, the process adheres to green chemistry principles by minimizing the use of toxic substances and reducing the generation of hazardous waste. This alignment with sustainability goals is increasingly important for multinational corporations aiming to reduce their carbon footprint and meet stringent environmental, social, and governance (ESG) criteria. The ease of waste treatment further lowers the operational burden on facility management teams.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of (S)-4-phenyl-2-oxazolidinone based on the disclosed patent technology. These insights are derived from the specific reaction mechanisms and advantageous effects detailed in the intellectual property documentation. Understanding these nuances helps stakeholders make informed decisions about integrating this intermediate into their development pipelines.
Q: Why is this new synthesis route safer than conventional methods?
A: Conventional methods often rely on carbon disulfide (CS2), which is highly toxic, volatile, and flammable, or expensive reagents like Lawesson's reagent. This patented process utilizes borane reagents and simple alkoxide catalysts, significantly reducing workplace hazards and eliminating the need for specialized containment for toxic gases.
Q: What is the expected purity of the final oxazolidinone product?
A: According to the patent data, the described method consistently yields products with purity exceeding 99.0% after recrystallization. This high level of optical and chemical purity is critical for its application as a chiral auxiliary in asymmetric synthesis.
Q: Can this process be scaled for industrial production?
A: Yes, the process is designed for scalability. It avoids violent exothermic reactions associated with hydrogen peroxide additions found in older methods and uses common solvents like THF and 2-MeTHF, making it suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-4-Phenyl-2-Oxazolidinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality chiral auxiliaries play in the development of next-generation therapeutics. Our technical team has extensively analyzed the route disclosed in patent CN112500361A and possesses the expertise to execute this green synthesis with precision. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with reliability and speed. Our facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications for every batch of (S)-4-phenyl-2-oxazolidinone we deliver, supporting your R&D and commercial manufacturing goals.
We invite you to collaborate with us to leverage this advanced technology for your specific applications. Whether you require custom synthesis or bulk supply, our team is ready to provide a Customized Cost-Saving Analysis tailored to your project volume. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a stable, cost-effective, and high-quality supply of this essential chiral building block for your pharmaceutical or agrochemical programs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
