Advanced Organocatalytic Synthesis of Chiral 3,4-Dihydroquinazolin-2-one Intermediates for Pharmaceutical Applications
The pharmaceutical industry continuously seeks robust and scalable methodologies for constructing complex chiral heterocycles, particularly those serving as privileged scaffolds in drug discovery. Patent CN116120253A introduces a groundbreaking approach to synthesizing chiral 3,4-dihydroquinazolin-2-one derivatives, a structural motif prevalent in numerous bioactive molecules and natural products. Unlike conventional strategies that depend heavily on transition metal catalysis, this invention leverages a novel organophosphine-catalyzed asymmetric [4+2] cycloaddition reaction. This metal-free paradigm shift not only addresses the critical issue of heavy metal residue in active pharmaceutical ingredients (APIs) but also offers a versatile platform for generating structural diversity. The core innovation lies in the design of a new Morita-Baylis-Hillman (MBH) adduct derivative, specifically a vinyl 4H-benzo[1,3]oxazine bearing an ester group, which acts as a highly reactive dipole in the presence of chiral nucleophilic catalysts. 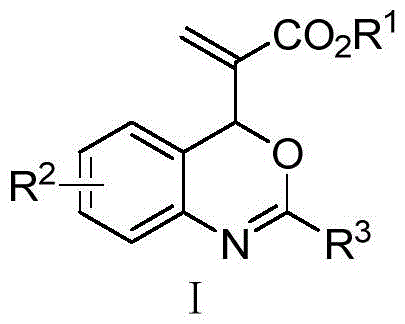
For procurement managers and supply chain directors, the implications of this technology are profound. By transitioning away from precious metal catalysts such as palladium or rhodium, manufacturers can achieve significant cost reduction in pharmaceutical intermediate manufacturing. The elimination of metal scavenging steps streamlines the production workflow, reducing both time and material costs associated with purification. Furthermore, the use of readily available organic catalysts enhances supply chain reliability, mitigating the risks associated with the volatility of rare earth metal markets. This process represents a reliable pharmaceutical intermediates supplier strategy, ensuring consistent quality and availability for downstream drug development projects.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of chiral dihydroquinazolinone cores has relied extensively on transition metal-catalyzed reactions or stoichiometric chiral auxiliaries. These traditional pathways often suffer from significant drawbacks that hinder their efficiency in a commercial setting. Firstly, the requirement for expensive metal-ligand complexes drastically increases the raw material cost, making the final API economically less viable. Secondly, and perhaps more critically, the removal of trace metal residues to meet stringent regulatory limits (often in the ppm range) necessitates additional purification steps, such as column chromatography or specialized filtration, which lowers overall yield and increases solvent waste. Additionally, many metal-catalyzed processes require harsh reaction conditions, including high temperatures or strictly anhydrous environments, which complicate scale-up and pose safety hazards in large reactors. The substrate scope in these older methods is frequently narrow, failing to accommodate diverse functional groups without compromising enantioselectivity.
The Novel Approach
The methodology disclosed in CN116120253A overcomes these barriers through an elegant organocatalytic design. The process begins with the synthesis of a specialized 4H-benzoxazine intermediate, achieved via a one-pot cyclization of an MBH alcohol with di-tert-butyl dicarbonate (Boc2O) using a simple Lewis base like DABCO. 
This intermediate then undergoes an enantioselective [4+2] cycloaddition with isocyanates catalyzed by chiral organophosphines. This approach operates under mild conditions, typically between room temperature and -5°C, utilizing common solvents like dichloromethane. The absence of transition metals simplifies the workup procedure significantly, often requiring only standard aqueous washes and silica gel chromatography. Moreover, the reaction exhibits excellent functional group tolerance, accommodating electron-withdrawing and electron-donating substituents on both the benzoxazine and isocyanate partners with high yields ranging from 63% to 98% for the intermediate and 71% to 81% for the final chiral product. This robustness makes it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Organophosphine-Catalyzed Asymmetric [4+2] Cycloaddition
The success of this synthetic route hinges on the unique reactivity of the ester-substituted vinyl 4H-benzoxazine. In the presence of a chiral nucleophilic organophosphine catalyst, such as the optimized catalyst P5, the electron-deficient double bond of the benzoxazine is activated. The phosphine attacks the beta-carbon of the vinyl group, generating a zwitterionic dipole intermediate characterized by a nitrogen anion nucleophilic site. This dipole is stabilized by the adjacent ester group, which enhances the electrophilicity of the system and directs the stereochemical outcome of the subsequent ring closure. ![Asymmetric [4+2] cycloaddition reaction scheme between benzoxazine I and isocyanate M2 catalyzed by chiral phosphine P](/insights/img/chiral-dihydroquinazolinone-intermediates-supplier-organocatalysis-20260305144446-04.webp)
The dipole then engages in a highly stereoselective cycloaddition with the isocyanate dipolarophile. The chiral environment provided by the bulky substituents on the phosphine catalyst ensures that the attack occurs from a specific face, leading to the formation of the new C-N and C-C bonds with high enantiomeric excess (ee). Experimental data indicates that optimizing the catalyst structure and reaction temperature is crucial; for instance, lowering the temperature to -5°C improved the ee value to 90% without sacrificing yield. This mechanistic pathway avoids the formation of racemic byproducts commonly seen in thermal cycloadditions, ensuring a clean impurity profile. The ability to fine-tune the electronic properties of the catalyst allows for precise control over the reaction kinetics, minimizing side reactions such as polymerization or hydrolysis of the sensitive isocyanate moiety.
How to Synthesize Chiral 3,4-Dihydroquinazolin-2-ones Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production. The process is divided into distinct stages: preparation of the MBH adduct precursor, cyclization to the benzoxazine core, and the final asymmetric coupling. Each step has been optimized for yield and purity, utilizing standard laboratory equipment and reagents. The detailed standardized synthesis steps see the guide below, which covers reagent ratios, solvent choices, and purification techniques essential for reproducing the high enantioselectivity reported in the examples.
- Preparation of MBH Alcohol Precursors: React substituted 2-aminobenzoic acids with LiAlH4 to form amino alcohols, followed by acylation and oxidation to generate the requisite aldehyde intermediates for MBH reaction.
- One-Pot Cyclization to 4H-Benzoxazines: Treat the MBH alcohol adducts with di-tert-butyl dicarbonate (Boc2O) and a Lewis base catalyst like DABCO in dichloromethane at room temperature to form the vinyl 4H-benzo[1,3]oxazine core.
- Asymmetric [4+2] Cycloaddition: React the 4H-benzoxazine derivative with an aryl isocyanate in the presence of a chiral organophosphine catalyst (e.g., P5) in DCM at -5°C to yield the target chiral 3,4-dihydroquinazolin-2-one with high enantiomeric excess.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this organocatalytic technology offers tangible benefits for the commercial production of high-purity pharmaceutical intermediates. From a cost perspective, the replacement of precious metal catalysts with organic phosphines results in substantial cost savings. Organic catalysts are generally more stable, easier to handle, and significantly cheaper than their metallic counterparts. Furthermore, the simplified purification process reduces solvent consumption and waste disposal costs, contributing to a greener and more economical manufacturing footprint. The use of common solvents like dichloromethane and ethyl acetate ensures that supply chains remain resilient, as these materials are globally available and not subject to the same geopolitical constraints as rare metals.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the associated ligand systems directly lowers the bill of materials. Additionally, the removal of metal scavenging units and specialized filtration steps reduces capital expenditure on equipment and operational expenses related to maintenance and validation. The high atom economy of the [4+2] cycloaddition further minimizes raw material waste, enhancing the overall process efficiency and profitability for large-scale production runs.
- Enhanced Supply Chain Reliability: By relying on organocatalysts and widely available starting materials such as anthranilic acid derivatives and isocyanates, manufacturers can mitigate supply chain disruptions. The robustness of the reaction conditions means that production is less susceptible to variations in raw material quality or environmental factors. This stability ensures consistent delivery schedules for downstream clients, reducing lead time for high-purity pharmaceutical intermediates and strengthening long-term supplier relationships.
- Scalability and Environmental Compliance: The mild reaction temperatures and ambient pressure conditions facilitate easy scale-up from gram to kilogram scales without the need for specialized high-pressure reactors. The reduced generation of hazardous metal waste aligns with increasingly strict environmental regulations, simplifying the permitting process for new manufacturing facilities. The one-pot nature of the benzoxazine formation step also reduces the number of unit operations, thereby lowering energy consumption and improving the overall sustainability profile of the synthesis.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis method. These answers are derived directly from the experimental data and scope defined in the patent documentation, providing clarity on the practical application of this technology for potential partners and licensees.
Q: How does this organocatalytic method improve upon traditional metal-catalyzed synthesis?
A: Traditional methods often rely on expensive transition metal complexes which require rigorous removal to meet pharmaceutical purity standards. This patented organophosphine-catalyzed route eliminates heavy metal contamination risks entirely, simplifies downstream purification, and operates under milder conditions while maintaining high enantioselectivity (up to 92% ee).
Q: What is the substrate scope for the benzoxazine and isocyanate components?
A: The methodology demonstrates exceptional versatility. The benzoxazine component tolerates various ester groups (methyl, ethyl, benzyl) and aromatic substituents including electron-withdrawing (F, Cl, Br) and electron-donating (Me, OMe) groups. Similarly, the isocyanate partner accepts diverse aryl substitutions without significant loss in yield or stereoselectivity.
Q: Is this synthetic route suitable for large-scale commercial production?
A: Yes, the process is designed for scalability. It utilizes commercially available starting materials, common organic solvents like dichloromethane, and avoids cryogenic conditions lower than -5°C. The one-pot nature of the benzoxazine formation step significantly reduces operational complexity and waste generation, facilitating cost reduction in pharmaceutical intermediate manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4-Dihydroquinazolin-2-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this organocatalytic technology in advancing drug discovery pipelines. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop to market. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to guarantee enantiomeric excess levels matching the patent's high standards. We are committed to delivering high-purity pharmaceutical intermediates that meet the exacting requirements of global regulatory bodies.
We invite you to collaborate with us to leverage this efficient and sustainable synthetic route for your next-generation therapeutics. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and target timelines. Please contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary molecules. Together, we can accelerate the development of life-saving medicines through innovative chemistry.
