Advanced Synthetic Route for Heteroaryl Thiosemicarbazone Antineoplastic Intermediates
Advanced Synthetic Route for Heteroaryl Thiosemicarbazone Antineoplastic Intermediates
The pharmaceutical industry is constantly seeking robust, scalable, and safe methodologies for the production of potent antineoplastic agents. Patent CN1907970A introduces a transformative synthetic strategy for heteroaryl thiosemicarbazone compounds, a class of molecules demonstrating significant tumor-suppression activity. This technology addresses critical bottlenecks in traditional manufacturing, specifically the reliance on hazardous reagents and multi-step purification processes that erode margins. By leveraging a novel condensation pathway between organometallic-derived imines and thiosemicarbazides, this method offers a streamlined approach to generating high-value pharmaceutical intermediates. For R&D directors and procurement specialists, understanding this shift is vital for securing supply chains for next-generation cancer therapeutics.
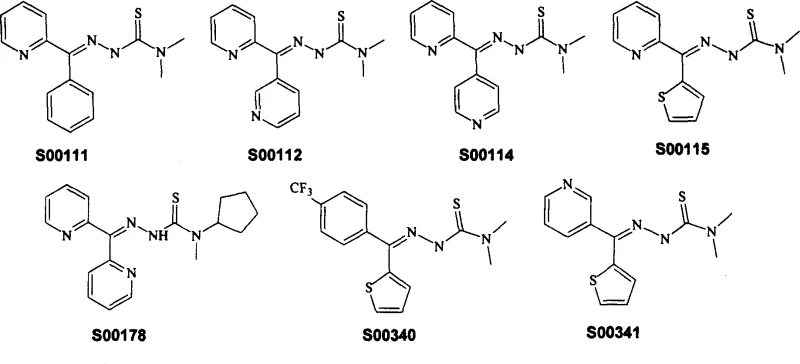
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of thiosemicarbazone derivatives has been plagued by significant operational and safety challenges. Traditional routes, often depicted in earlier literature and prior patent filings, frequently rely on the direct reaction of ketones or aldehydes with thiosemicarbazides derived from toxic isothiocyanates or methylmercaptan precursors. These conventional pathways suffer from low atom economy and generate substantial quantities of hazardous waste, particularly pungent and toxic sulfur-containing byproducts that require expensive abatement systems. Furthermore, the purification of the final product is often difficult due to the formation of closely related impurities, necessitating multiple recrystallization steps that drastically reduce the overall yield. The cumulative effect of these inefficiencies is a high cost of goods sold (COGS) and a supply chain vulnerable to regulatory scrutiny regarding environmental emissions.
The Novel Approach
In stark contrast, the methodology disclosed in CN1907970A circumvents these issues by constructing the carbon-nitrogen framework through a Grignard or lithium-mediated addition to nitriles. This innovative route allows for the generation of the key imine intermediate under controlled conditions, which is then condensed with a pre-formed thiosemicarbazide. A major advantage of this approach is the ability to utilize the crude imine directly in the subsequent condensation step without rigorous isolation, effectively telescoping the synthesis. This not only reduces solvent consumption and processing time but also minimizes material loss associated with intermediate workups. By eliminating the need for methylmercaptan and simplifying the purification train, this method presents a compelling case for cost reduction in pharmaceutical intermediate manufacturing while adhering to stricter environmental standards.
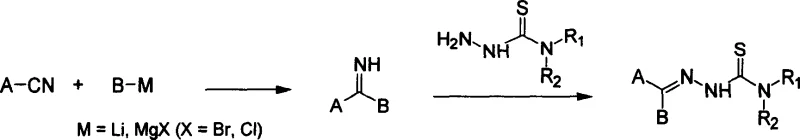
Mechanistic Insights into Grignard-Mediated Imine Formation and Condensation
The core of this technological advancement lies in the precise orchestration of organometallic chemistry to build the heteroaryl scaffold. The process initiates with the nucleophilic attack of a Grignard reagent (R-MgX) or an organolithium species on a nitrile group (R'-CN). This addition forms a metallo-imine intermediate which, upon acidic hydrolysis or quenching with ammonium salts, yields the desired ketimine structure. The versatility of this step is remarkable, as it accommodates a wide range of heterocyclic substituents, including pyridyl and thienyl groups, which are essential for the biological activity of the final antineoplastic agent. The reaction is typically conducted in ethereal solvents such as tetrahydrofuran (THF) or diethyl ether at temperatures ranging from -50°C to 60°C, allowing for fine-tuned control over regioselectivity and side reactions.
Following the formation of the imine, the second critical mechanistic phase involves the acid-catalyzed condensation with the thiosemicarbazide moiety. The thiosemicarbazide itself is synthesized via a safe, aqueous pathway involving the reaction of amines with dithiocarbonic anhydride and sodium chloroacetate, followed by hydrazinolysis. When the imine and thiosemicarbazide are mixed in the presence of a catalytic amount of acid, such as dry hydrogen chloride or acetic acid, a dehydration condensation occurs to form the final C=N bond of the thiosemicarbazone. This mechanism ensures high fidelity in the product structure, minimizing the formation of geometric isomers or hydrolysis byproducts. The resulting crude product can be purified to exceptional standards (>99.5% purity) through simple recrystallization, validating the robustness of the chemical design.
How to Synthesize Heteroaryl Thiosemicarbazone Efficiently
The implementation of this synthetic route requires strict adherence to moisture-free conditions during the organometallic step, followed by controlled acidification for the condensation. The patent outlines a clear four-stage protocol that begins with the preparation of the thiosemicarbazide core, followed by the generation of the heteroaryl imine, and concludes with the coupling reaction. Operators must ensure that the Grignard reagent is freshly prepared or titrated to ensure stoichiometric accuracy, as this directly impacts the yield of the imine intermediate. The subsequent condensation is remarkably tolerant, allowing for the use of crude imine solutions which streamlines the workflow significantly. For a detailed breakdown of the specific molar ratios, solvent choices, and temperature profiles required to replicate this high-yielding process, please refer to the standardized synthesis guide below.
- Prepare the thiosemicarbazide core by reacting organic amines with dithiocarbonic anhydride and sodium chloroacetate in aqueous sodium hydroxide, followed by hydrazine hydrate treatment.
- Synthesize the heteroaryl-substituted imine intermediate by reacting the corresponding nitrile compound with a Grignard or lithium reagent in ether or THF solvents at controlled temperatures.
- Execute the final condensation by mixing the crude imine with the thiosemicarbazide derivative in the presence of a catalytic amount of acid, followed by recrystallization to achieve high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic methodology offers tangible strategic benefits beyond mere technical elegance. The primary value driver is the substantial simplification of the manufacturing process, which directly correlates to reduced operational expenditures. By eliminating the need for hazardous methylmercaptan handling, facilities can avoid the capital investment and ongoing maintenance costs associated with specialized scrubbing systems and personal protective equipment required for toxic gas management. Furthermore, the telescoping of the imine formation and condensation steps reduces the number of unit operations, leading to faster batch cycle times and increased throughput capacity without the need for additional reactor volume. This efficiency gain is crucial for meeting the demanding delivery schedules of global pharmaceutical clients.
- Cost Reduction in Manufacturing: The economic impact of this route is driven by the elimination of expensive purification stages and the use of readily available commodity chemicals. Since the crude imine can be used directly, there is a significant reduction in solvent usage and energy consumption associated with drying and isolating intermediates. Additionally, the avoidance of transition metal catalysts or exotic reagents means that raw material sourcing is stable and less susceptible to market volatility. The overall process intensity is lower, allowing for competitive pricing structures that enhance the margin potential for downstream API manufacturers.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the reliance on fundamental chemical building blocks such as nitriles and amines, which are produced at scale by the global petrochemical industry. Unlike specialized intermediates that may have single-source suppliers, the starting materials for this route are widely accessible, mitigating the risk of supply disruptions. The robustness of the reaction conditions also means that production is less sensitive to minor variations in utility supplies or environmental conditions, ensuring consistent output quality and reliable delivery timelines for long-term contracts.
- Scalability and Environmental Compliance: From an EHS (Environment, Health, and Safety) perspective, this process represents a significant leap forward in sustainability. The aqueous synthesis of the thiosemicarbazide precursor and the absence of volatile sulfur byproducts simplify waste stream management, reducing the burden on wastewater treatment facilities. This alignment with green chemistry principles facilitates easier regulatory approval for new manufacturing sites and supports the corporate sustainability goals of multinational partners. The process is inherently scalable from kilogram to multi-ton production, making it suitable for both clinical trial material and commercial launch volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this heteroaryl thiosemicarbazone synthesis. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aiming to clarify the operational feasibility and quality advantages of this novel approach. Understanding these nuances is essential for technical teams evaluating the transfer of this technology to pilot or production scales.
Q: How does this synthetic method improve safety compared to conventional thiosemicarbazone production?
A: Traditional methods often generate methylmercaptan, a highly toxic and pungent byproduct that requires complex scrubbing and safety protocols. This novel route completely avoids the use of methylmercaptan precursors, significantly reducing hazardous waste handling costs and improving operator safety profiles.
Q: What purity levels can be achieved with this Grignard-based condensation route?
A: The patent data indicates that through optimized recrystallization protocols using solvents like ethanol or ethyl acetate, the final heteroaryl thiosemicarbazone products can achieve purity specifications exceeding 99.5%, which is critical for downstream pharmaceutical applications.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process utilizes commercially available starting materials such as nitriles and standard Grignard reagents. The reaction conditions are moderate (ranging from -50°C to 60°C), and the ability to use crude imine intermediates directly in the condensation step simplifies the workflow, enhancing scalability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Heteroaryl Thiosemicarbazone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex antineoplastic intermediates requires more than just a patent; it demands deep process engineering expertise. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, utilizing our rigorous QC labs to verify every batch against the highest international standards. Our infrastructure is designed to handle sensitive organometallic chemistries safely and efficiently, guaranteeing supply continuity for your critical drug development programs.
We invite you to collaborate with us to leverage this advanced synthetic technology for your pipeline. By partnering with our technical procurement team, you can obtain a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential partners to contact us directly to request specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized manufacturing capabilities can drive value and accelerate your time to market.
