Advanced Catalytic Hydrogenation for High-Purity 2-Amino-2-Arylethanols Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to synthesize complex amino alcohol scaffolds, which serve as critical building blocks for numerous active pharmaceutical ingredients. A pivotal advancement in this domain is detailed in patent CN1066711C, which discloses a novel process for the preparation of 2-amino-2-arylethanols through the catalytic hydrogenation of alpha-hydroxyketoximes. This technology represents a significant departure from traditional reduction methods, offering a robust alternative that utilizes Raney nickel or Raney cobalt catalysts in the presence of a diluent and a base. The strategic shift towards base-metal catalysis not only addresses the economic limitations of precious metal systems but also resolves critical selectivity issues associated with halogenated substrates. By leveraging this methodology, manufacturers can achieve high-purity outputs with exceptional yields, thereby establishing a reliable foundation for the commercial scale-up of complex pharmaceutical intermediates.
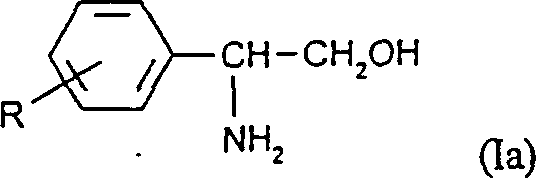
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-amino-2-aryl alcohols and their derivatives has relied heavily on stoichiometric reduction agents such as sodium borohydride combined with boron trifluoride or lithium aluminum hydride. While effective on a laboratory scale, these metal hydride reagents present severe drawbacks when translated to technical or industrial scales, primarily due to their exorbitant cost and the substantial security costs associated with handling such reactive species. Furthermore, alternative catalytic methods employing noble metals like rhodium or palladium have demonstrated significant functional group intolerance; for instance, palladium catalysts frequently induce the unwanted reductive removal of halogen atoms from halogenated aromatic compounds, leading to compromised product integrity. Additionally, certain substrates, such as 2-indanone oxime, have proven resistant to reduction using rhodium catalysts, highlighting the restricted applicability of these conventional noble metal systems in diverse synthetic contexts.
The Novel Approach
In stark contrast to these legacy methods, the novel approach outlined in the patent utilizes Raney nickel or Raney cobalt to catalyze the hydrogenation of alpha-hydroxyketoximes under basic conditions. This methodology effectively circumvents the dehalogenation issues plaguing palladium-catalyzed routes, allowing for the successful processing of halogen-substituted aryl groups without loss of the halogen functionality. The process operates efficiently in common organic solvents such as methanol or ethanol, often in the presence of ammonia, which serves as a crucial base to facilitate the reaction mechanism. By shifting from expensive stoichiometric reagents or sensitive noble metals to robust, non-noble metal catalysts, this approach drastically simplifies the operational complexity and enhances the economic viability of producing high-purity 2-amino-2-arylethanols for large-scale applications.
Mechanistic Insights into Raney Nickel-Catalyzed Hydrogenation
The core of this technological breakthrough lies in the specific interaction between the alpha-hydroxyketoxime substrate and the Raney nickel surface under hydrogen pressure. The reaction proceeds through the activation of molecular hydrogen on the catalyst surface, followed by the sequential reduction of the oxime functionality to the corresponding amine while preserving the adjacent hydroxyl group. The presence of a base, typically ammonia, is mechanistically significant as it likely assists in stabilizing intermediate species and preventing the formation of undesirable byproducts such as hydroxyoximes, which are known to form when using platinum or Raney nickel catalysts under neutral or acidic conditions. This precise control over the reaction environment ensures that the reduction stops selectively at the amino alcohol stage rather than proceeding to further degradation or side reactions.
From an impurity control perspective, the choice of Raney nickel over palladium is paramount for maintaining the purity profile of halogenated derivatives. In conventional palladium-catalyzed hydrogenations, the carbon-halogen bond is often susceptible to hydrogenolysis, resulting in dehalogenated impurities that are difficult to separate and can compromise the safety profile of the final drug substance. The Raney nickel system described herein exhibits superior chemoselectivity, tolerating various substituents including fluorine, chlorine, and bromine on the aromatic ring. This selectivity mechanism ensures that the final product retains the intended substitution pattern, thereby minimizing the burden on downstream purification processes and ensuring that the impurity spectrum remains well within the stringent specifications required for pharmaceutical grade materials.
How to Synthesize 2-Amino-2-Arylethanols Efficiently
The practical implementation of this synthesis involves a straightforward yet highly controlled hydrogenation protocol that is amenable to standard industrial reactor setups. The process begins with the preparation of the alpha-hydroxyketoxime intermediate, which can be synthesized from corresponding omega-hydroxyketones via reaction with hydroxylamine hydrochloride. Once the oxime is prepared, it is suspended in a suitable alcohol solvent, and liquid ammonia is introduced to establish the necessary basic environment before the addition of the Raney nickel catalyst. The reaction is then pressurized with hydrogen gas and heated to moderate temperatures, typically around 50°C, until hydrogen uptake indicates completion. For a comprehensive understanding of the specific operational parameters and safety protocols required for this transformation, please refer to the detailed standardized synthesis steps provided in the guide below.
- Prepare the reaction mixture by suspending the alpha-hydroxyketoxime intermediate in a suitable organic diluent such as methanol or ethanol, ensuring the presence of a base like liquid ammonia.
- Add the heterogeneous catalyst, specifically Raney nickel or Raney cobalt, to the reaction vessel under an inert atmosphere before introducing hydrogen gas.
- Conduct the hydrogenation at elevated pressures between 60 to 70 bar and temperatures around 50°C until hydrogen uptake ceases, followed by filtration and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this Raney nickel-catalyzed process offers compelling advantages rooted in both cost efficiency and supply reliability. By eliminating the dependency on precious metal catalysts like rhodium and palladium, which are subject to volatile market pricing and geopolitical supply risks, manufacturers can secure a more stable and predictable cost structure for their raw materials. Furthermore, the avoidance of expensive stoichiometric reducing agents like lithium aluminum hydride removes significant safety overheads and waste disposal costs associated with hazardous chemical handling. This transition to a catalytic hydrogenation process using abundant base metals translates directly into substantial cost savings in pharmaceutical intermediate manufacturing, allowing for more competitive pricing strategies without compromising on quality or yield.
- Cost Reduction in Manufacturing: The replacement of noble metal catalysts with Raney nickel significantly lowers the direct material costs associated with the catalytic system, as nickel is vastly more abundant and less expensive than rhodium or palladium. Additionally, the process avoids the use of costly metal hydrides, which require specialized handling and generate significant amounts of inorganic waste, thereby reducing both raw material expenditure and waste treatment expenses. The high yields reported, such as the 84.5% theoretical yield observed in specific embodiments, further enhance the overall process economics by maximizing the output per unit of input material. These factors combine to create a highly cost-effective production route that optimizes the total cost of ownership for the final amino alcohol product.
- Enhanced Supply Chain Reliability: Utilizing Raney nickel, a widely available and commercially mature catalyst, mitigates the supply chain risks associated with scarce precious metals that often face bottlenecks and long lead times. The robustness of the catalyst allows for consistent batch-to-batch performance, reducing the likelihood of production delays caused by catalyst failure or variability. Moreover, the ability to process a wide range of substituted aryl groups, including those with halogen functionalities, means that a single versatile platform can be used to produce multiple different intermediates, simplifying inventory management and enhancing the agility of the supply chain to respond to changing market demands.
- Scalability and Environmental Compliance: The process is inherently scalable, operating effectively at hydrogen pressures between 60 to 70 bar and temperatures around 50°C, conditions that are well within the capabilities of standard industrial hydrogenation reactors. The use of common solvents like methanol and ethanol simplifies solvent recovery and recycling, contributing to a greener manufacturing footprint. By avoiding the generation of heavy metal waste streams associated with stoichiometric reductions, this method aligns better with increasingly stringent environmental regulations, facilitating easier permitting and long-term operational sustainability for large-scale commercial production facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic hydrogenation technology. These answers are derived directly from the experimental data and technical disclosures found within the patent literature, providing a factual basis for evaluating the feasibility of this route for your specific project needs. Understanding these nuances is essential for R&D teams planning process validation and for procurement teams assessing the long-term viability of this supply source.
Q: Why is Raney Nickel preferred over noble metals for this hydrogenation?
A: Raney Nickel is preferred because it avoids the dehalogenation side reactions often observed with palladium catalysts when processing halogenated aromatic compounds, ensuring higher structural integrity of the final product.
Q: What are the typical reaction conditions for this process?
A: The process typically operates at temperatures between 30°C and 120°C, with hydrogen pressures ranging from 15 to 100 bar, utilizing alcoholic solvents and ammonia as a base.
Q: How does this method improve upon traditional metal hydride reductions?
A: Unlike expensive and hazardous metal hydrides like LiAlH4, this catalytic method uses molecular hydrogen and reusable catalysts, significantly reducing safety risks and raw material costs on an industrial scale.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Amino-2-Arylethanols Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in the development of next-generation pharmaceuticals. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering high-purity 2-amino-2-arylethanols that meet stringent purity specifications, supported by our rigorous QC labs which employ advanced analytical techniques to verify every batch. Our expertise in catalytic hydrogenation allows us to optimize these processes for maximum yield and minimal impurity formation, providing our partners with a secure and high-quality supply of essential intermediates.
We invite you to collaborate with us to leverage this advanced technology for your upcoming projects. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can support your supply chain goals and drive value for your organization.
