Scalable Production of High-Purity Hydrazone Compounds for Optoelectronic Applications
The landscape of electronic chemical manufacturing is constantly evolving, driven by the need for safer, more efficient, and higher-purity synthetic routes. Patent CN1976896A introduces a transformative methodology for the production of hydrazone compounds, which serve as critical photoconductive substances in electrophotographic photoreceptors. This technology addresses long-standing challenges in the industry, specifically the handling of toxic and unstable intermediates that have historically plagued the synthesis of these vital materials. By shifting from a multi-step isolation process to a streamlined one-pot synthesis, this innovation not only enhances the quality of the final product but also significantly mitigates occupational health risks associated with mutagenic hydrazine compounds. For stakeholders in the display and optoelectronic sectors, understanding this process is key to securing a reliable supply chain for high-performance imaging materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of hydrazone compounds has relied on methods that involve the isolation of hydrazine intermediates, often as hydrochloride salts, following the reduction of N-nitrosamine precursors. Traditional reduction techniques frequently employ zinc powder in acetic acid or other harsh reducing agents like sodium-ethanol, which introduce significant operational inefficiencies. A major drawback of these conventional pathways is the necessity of filtering out metal residues post-reduction, a step that inadvertently exposes the unstable hydrazine intermediate to air, leading to oxidative decomposition. This degradation results in substantially lower yields, often hovering around 60%, and compromises the purity of the final photoconductive material. Furthermore, the isolation and handling of hydrazine compounds pose severe safety hazards due to their known toxicity and mutagenicity, creating regulatory and liability burdens for manufacturers.
The Novel Approach
In stark contrast, the novel approach detailed in patent CN1976896A utilizes a sophisticated one-pot synthesis strategy that completely eliminates the isolation of the hydrazine intermediate. By employing thiourea dioxide and a base within a carefully controlled aqueous-organic solvent system, the N-nitrosamine precursor is reduced directly in the reaction mixture. This method bypasses the filtration steps that cause oxidative loss, thereby preserving the integrity of the reactive hydrazine species until it immediately reacts with the carbonyl compound. The result is a dramatic improvement in process efficiency, with yields consistently exceeding 80% and product purity reaching levels above 99%. This seamless transition from intermediate to final product not only optimizes resource utilization but also creates a closed system that effectively shields workers from exposure to hazardous substances.
Mechanistic Insights into Thiourea Dioxide Reduction and Condensation
The core of this technological advancement lies in the precise control of the reduction and condensation phases within a single reactor environment. The process begins with the nitrosation of an amine compound, represented by general formula (1), using sodium nitrite and an acid in a mixed solvent system. 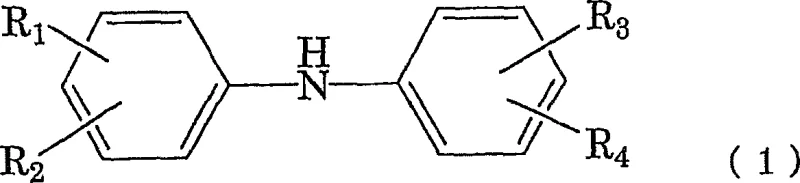 This initial step generates the N-nitrosamine intermediate, depicted as general formula (2), which serves as the substrate for the subsequent reduction.
This initial step generates the N-nitrosamine intermediate, depicted as general formula (2), which serves as the substrate for the subsequent reduction. 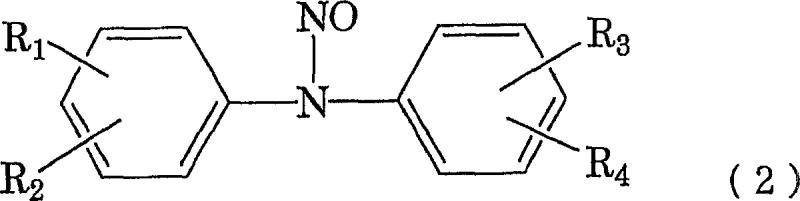 Unlike traditional metal-based reductions, the use of thiourea dioxide allows for a cleaner conversion to the hydrazine derivative, shown as general formula (3), without generating solid metal waste that requires removal.
Unlike traditional metal-based reductions, the use of thiourea dioxide allows for a cleaner conversion to the hydrazine derivative, shown as general formula (3), without generating solid metal waste that requires removal. 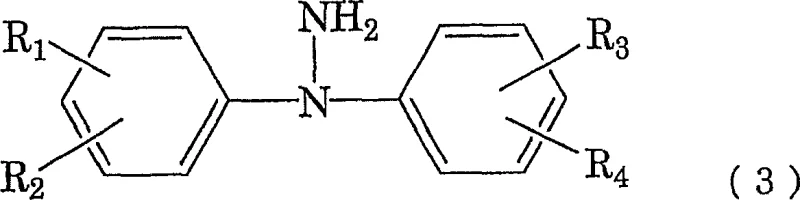 Critical to this mechanism is the maintenance of the reaction temperature between 38°C and 43°C; deviations outside this narrow window can lead to the cleavage of the N-N bond or the regeneration of the starting amine, both of which would detrimentally impact the overall yield and purity of the synthesis.
Critical to this mechanism is the maintenance of the reaction temperature between 38°C and 43°C; deviations outside this narrow window can lead to the cleavage of the N-N bond or the regeneration of the starting amine, both of which would detrimentally impact the overall yield and purity of the synthesis.
Following the formation of the hydrazine species, the process leverages its inherent reactivity by introducing a carbonyl compound, defined by general formula (4), directly into the mixture. 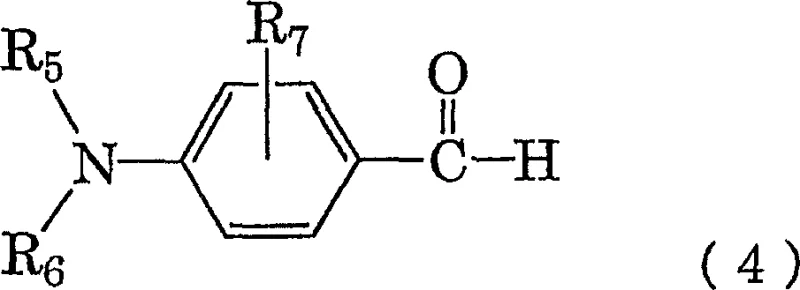 This immediate condensation reaction capitalizes on the high concentration of the freshly generated hydrazine, driving the equilibrium towards the formation of the target hydrazone compound, general formula (5). The absence of isolation steps ensures that the hydrazine does not undergo auto-oxidation, a common failure mode in batch processes involving unstable intermediates. Furthermore, the choice of solvents, typically a blend of water and methanol followed by extraction with toluene or ethers, is optimized to facilitate phase separation and product crystallization without the need for complex chromatographic purification. This mechanistic elegance translates directly into a robust manufacturing protocol capable of delivering electronic-grade chemicals with minimal impurity profiles.
This immediate condensation reaction capitalizes on the high concentration of the freshly generated hydrazine, driving the equilibrium towards the formation of the target hydrazone compound, general formula (5). The absence of isolation steps ensures that the hydrazine does not undergo auto-oxidation, a common failure mode in batch processes involving unstable intermediates. Furthermore, the choice of solvents, typically a blend of water and methanol followed by extraction with toluene or ethers, is optimized to facilitate phase separation and product crystallization without the need for complex chromatographic purification. This mechanistic elegance translates directly into a robust manufacturing protocol capable of delivering electronic-grade chemicals with minimal impurity profiles.
How to Synthesize Hydrazone Compounds Efficiently
The implementation of this synthesis route requires strict adherence to the sequential addition of reagents and temperature controls to maximize the benefits of the one-pot design. The detailed standardized synthesis steps, including specific molar ratios and workup procedures derived from the patent examples, are outlined below to guide technical teams in replicating this high-efficiency process.
- Dissolve the amine compound in a mixed solvent of water and a water-miscible organic solvent, then react with acid and sodium nitrite to form the N-nitrosamine intermediate.
- Add thiourea dioxide and a base directly to the reaction solution to reduce the N-nitrosamine to the hydrazine compound without isolating it.
- Condense the resulting hydrazine solution directly with a carbonyl compound in the same reactor to obtain the final high-purity hydrazone product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented process offers tangible strategic advantages that extend beyond mere technical specifications. The elimination of metal-based reducing agents like zinc powder removes the logistical burden of handling and disposing of heavy metal waste, which is both costly and environmentally regulated. By simplifying the workflow to a continuous liquid-phase operation, manufacturers can significantly reduce cycle times and increase throughput capacity without expanding physical infrastructure. This efficiency gain is crucial for meeting the demanding delivery schedules of the global electronics market, where just-in-time delivery of high-purity intermediates is often a prerequisite for maintaining production lines.
- Cost Reduction in Manufacturing: The shift to a one-pot synthesis fundamentally alters the cost structure of hydrazone production by removing multiple unit operations such as filtration, drying, and re-dissolution of intermediates. The substantial increase in yield from approximately 60% in conventional methods to over 80% in this new process means that less raw material is required to produce the same amount of final product, directly lowering the cost of goods sold. Additionally, the avoidance of expensive purification steps like column chromatography, which are often necessary to remove metal traces and oxidation byproducts in older methods, further drives down operational expenditures. These cumulative efficiencies result in a more competitive pricing model for high-purity electronic chemicals without compromising on quality standards.
- Enhanced Supply Chain Reliability: Safety is a critical component of supply chain continuity, and the reduced toxicity profile of this process minimizes the risk of shutdowns due to occupational health incidents. By keeping mutagenic hydrazine intermediates contained within the reactor, the facility operates with a lower risk profile, ensuring uninterrupted production even under stringent regulatory scrutiny. The use of common, readily available solvents such as methanol, toluene, and acetic acid ensures that raw material sourcing remains stable and resilient against market fluctuations. This reliability makes the manufacturer a more dependable partner for long-term contracts, providing assurance to downstream clients that their supply of photoconductive materials will remain consistent.
- Scalability and Environmental Compliance: The simplicity of the workup procedure, which involves distillation and crystallization rather than complex solid-liquid separations, makes this process highly amenable to scale-up from pilot plants to multi-ton commercial production. The absence of heavy metal sludge simplifies wastewater treatment and waste disposal, aligning the manufacturing process with increasingly rigorous environmental compliance standards globally. This green chemistry aspect not only reduces disposal costs but also enhances the corporate sustainability profile of the supplier, a factor that is becoming increasingly important for multinational corporations when selecting vendors. The ability to scale this clean technology ensures that supply can grow in tandem with market demand for advanced optoelectronic devices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this hydrazone synthesis technology. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making.
Q: Why is the one-pot synthesis method safer than conventional methods for hydrazone production?
A: Conventional methods require isolating the hydrazine intermediate, which is often toxic and mutagenic. The one-pot method keeps this unstable intermediate in the reactor, preventing worker exposure and minimizing oxidation risks.
Q: What are the yield improvements of using thiourea dioxide compared to zinc reduction?
A: Traditional zinc powder reduction methods typically achieve yields around 60% due to oxidative decomposition during filtration. The thiourea dioxide method described in patent CN1976896A achieves yields exceeding 80% with purity above 99%.
Q: Can this process be scaled for commercial electronic chemical manufacturing?
A: Yes, the process eliminates complex filtration steps associated with metal residues and uses common solvents like methanol and toluene, making it highly suitable for commercial scale-up in the production of photoconductive materials.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Hydrazone Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity hydrazone compounds play in the performance of next-generation electrophotographic photoreceptors. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN1976896A are fully realized in an industrial setting. Our rigorous QC labs and stringent purity specifications guarantee that every batch of photoconductive substance meets the exacting standards required by the display and imaging industries. We are committed to delivering not just a chemical product, but a comprehensive solution that enhances your manufacturing efficiency and product quality.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain specific insights into how switching to this one-pot method can optimize your budget and reduce lead times. We encourage potential partners to contact us for specific COA data and route feasibility assessments tailored to your unique project requirements. Let us help you engineer a more robust and cost-effective supply chain for your electronic chemical needs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
