Advanced Synthesis of Alkyl-Substituted Benzoxazinorifamycin Derivatives for Commercial Antibiotic Production
The pharmaceutical landscape continuously demands novel antibiotic scaffolds to combat evolving bacterial resistance, and patent CN87104870A presents a significant advancement in this domain by disclosing a new class of alkyl-substituted benzoxazinorifamycin derivatives. These compounds, characterized by their unique structural modifications at the rifamycin core, demonstrate potent antibacterial efficacy against Gram-positive and acid-fast bacteria. The invention provides not only the novel chemical entities defined by Formula (I) but also robust preparation methods that leverage oxidative coupling strategies. For industrial partners seeking a reliable pharmaceutical intermediate supplier, understanding the nuances of this synthetic route is critical, as it offers a pathway to high-purity active ingredients with manageable impurity profiles. The versatility of the substituents, ranging from C1-C6 alkyl groups to various cyclic amines, allows for fine-tuning of pharmacokinetic properties, addressing a key pain point in modern antibiotic development where bioavailability often limits clinical success.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for modifying the rifamycin skeleton often involve harsh reaction conditions that can compromise the stability of the sensitive ansa-bridge structure, leading to complex impurity spectra that are difficult to purge during downstream processing. Conventional alkylation or acylation strategies frequently require expensive protecting group manipulations or utilize heavy metal catalysts that necessitate rigorous removal steps to meet stringent regulatory limits for residual metals in final drug products. Furthermore, older synthetic routes may suffer from low regioselectivity, resulting in mixtures of isomers that significantly reduce overall yield and increase the cost of goods sold. The reliance on unstable intermediates in classical approaches often mandates cryogenic temperatures or inert atmospheres that complicate scale-up operations, creating bottlenecks for supply chain heads who require consistent, large-volume production capabilities without frequent batch failures.
The Novel Approach
The methodology outlined in the patent introduces a streamlined oxidative coupling process that directly reacts rifamycin derivatives of Formula (II) with specific amines of Formula (AH) in the presence of an oxidizing agent. This approach bypasses the need for complex multi-step protection-deprotection sequences, significantly simplifying the synthetic tree. By utilizing readily available oxidants such as manganese dioxide, silver oxide, or potassium ferricyanide, the process achieves the desired transformation under relatively mild thermal conditions, typically ranging from -20°C to the boiling point of the solvent. This novel route enables the efficient installation of diverse cyclic amine moieties, including 3-to 9-membered rings, directly onto the benzoxazine ring system. The result is a more convergent synthesis that enhances overall throughput and reduces the environmental footprint associated with solvent usage and waste generation, aligning perfectly with modern green chemistry principles valued by procurement teams focused on sustainability.
Mechanistic Insights into Oxidative Aminative Coupling
The core chemical transformation involves the nucleophilic attack of the amine nitrogen on the electrophilic centers of the rifamycin quinone system, followed by re-oxidation to restore aromaticity and stability. In the absence of an oxidizing agent, the reaction might stall at a reduced hydroquinone stage or lead to unstable adducts; however, the inclusion of agents like manganese dioxide ensures the rapid regeneration of the quinoid structure, driving the equilibrium towards the desired product. The reaction mechanism is highly dependent on the electronic nature of the substituents X1 and X2 on the benzoxazine ring, where alkyl groups provide steric bulk that can influence the stereochemical outcome and metabolic stability of the final molecule. Solvent selection plays a pivotal role, with polar aprotic solvents like DMSO and DMF facilitating the solvation of the ionic intermediates and stabilizing the transition state. Understanding this mechanistic pathway is essential for R&D directors aiming to optimize reaction parameters, as slight variations in pH or oxidant stoichiometry can drastically impact the ratio of the desired product versus over-oxidized byproducts.
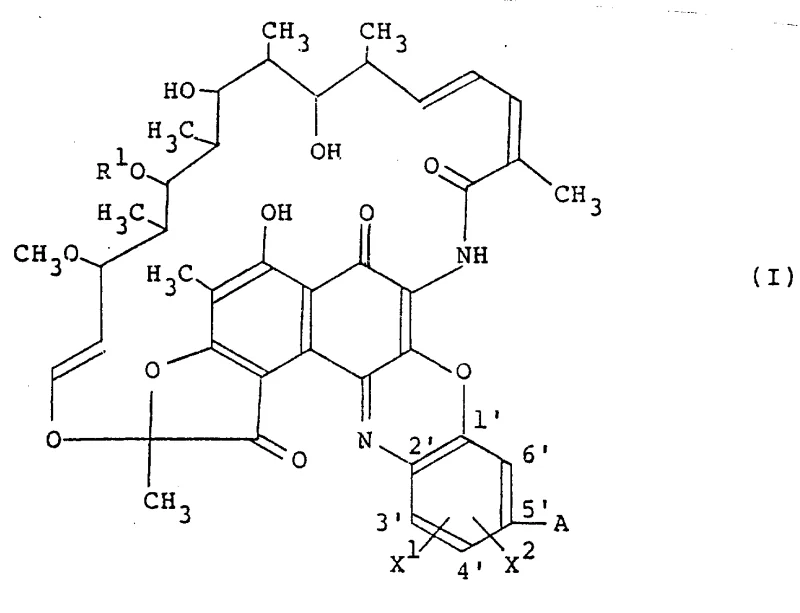
Impurity control is inherently built into this process through the careful selection of reaction temperature and time. The patent data indicates that reaction progress can be effectively monitored using thin-layer chromatography, allowing operators to quench the reaction precisely when the starting material is consumed but before degradation pathways become dominant. The use of specific cyclic amines, such as pyrrolidine, piperidine, or azetidine derivatives, introduces structural rigidity that can enhance the binding affinity to bacterial RNA polymerase while potentially reducing off-target toxicity. The ability to tune the lipophilicity of the molecule by varying the alkyl chain length at the X1 position provides a powerful tool for optimizing tissue penetration and serum half-life. This level of molecular precision ensures that the resulting active pharmaceutical ingredient meets the rigorous purity specifications required for clinical applications, minimizing the risk of adverse events associated with chemical impurities.
How to Synthesize Benzoxazinorifamycin Derivatives Efficiently
To successfully manufacture these high-value intermediates, operators must adhere to strict process controls regarding reagent quality and environmental conditions. The synthesis begins with the preparation of the rifamycin precursor, which is dissolved in a suitable organic solvent under an inert atmosphere to prevent premature oxidation. The amine component is then introduced, followed by the gradual addition of the oxidizing agent to manage the exotherm and ensure uniform mixing. Detailed standardized operating procedures for each step, including workup and purification via column chromatography or crystallization, are essential for reproducibility. The following guide outlines the critical operational phases derived from the patent examples, serving as a foundational reference for process engineers scaling this technology from the laboratory to the pilot plant.
- Dissolve the rifamycin derivative precursor (Formula II) in a polar organic solvent such as dimethyl sulfoxide (DMSO) or dimethylformamide (DMF).
- Add the specific cyclic amine (Formula AH) and an oxidizing agent like manganese dioxide (MnO2) to the reaction mixture.
- Stir the mixture at a controlled temperature between -5°C and 50°C for several hours to days, monitoring progress via TLC until completion.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the synthetic route described in CN87104870A offers substantial advantages that directly impact the bottom line and supply chain resilience. The elimination of precious metal catalysts in the key coupling step represents a significant cost reduction in pharmaceutical intermediate manufacturing, as it removes the need for expensive palladium or platinum reagents and the associated costly scavenging processes required to meet residual metal specifications. Furthermore, the use of common industrial solvents and oxidants like manganese dioxide ensures that raw material sourcing is robust and less susceptible to geopolitical supply disruptions. The simplicity of the workup procedure, which often involves mere filtration of the spent oxidant followed by solvent exchange, drastically reduces processing time and energy consumption. This efficiency translates into shorter production cycles, enabling suppliers to respond more agilely to market demand fluctuations and reducing the inventory carrying costs for downstream clients.
- Cost Reduction in Manufacturing: The process utilizes inexpensive and widely available oxidizing agents such as manganese dioxide instead of costly transition metal catalysts, which eliminates the need for complex metal removal steps and significantly lowers the cost of goods. By streamlining the synthetic sequence and reducing the number of unit operations, manufacturers can achieve substantial savings in both labor and utility expenses, making the final API more price-competitive in the global market.
- Enhanced Supply Chain Reliability: The starting materials, including Rifamycin S and various substituted phenols, are commodity chemicals with established global supply chains, ensuring consistent availability and mitigating the risk of production stoppages due to raw material shortages. The robustness of the reaction conditions, which tolerate a range of temperatures and do not require ultra-low temperature cryogenics, further enhances operational reliability, allowing for flexible scheduling and reduced dependency on specialized infrastructure.
- Scalability and Environmental Compliance: The synthetic method is inherently scalable, as demonstrated by the successful preparation of multi-gram quantities in the patent examples without loss of yield or purity. The reduced use of hazardous reagents and the ability to recycle solvents contribute to a lower environmental impact, facilitating easier regulatory approval and alignment with corporate sustainability goals. This scalability ensures that the technology can seamlessly transition from pilot-scale validation to multi-ton commercial production to meet global healthcare needs.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the synthesis and application of these novel derivatives, based on the detailed experimental data provided in the patent documentation. These insights are curated to assist technical teams in evaluating the feasibility of integrating this chemistry into their existing development pipelines. By clarifying the reaction scope and limitations, we aim to accelerate the decision-making process for partners considering this technology for their antibiotic portfolios.
Q: What is the primary advantage of the oxidative coupling method described in CN87104870A?
A: The method allows for the direct introduction of diverse cyclic amine groups onto the rifamycin scaffold under mild conditions, utilizing inexpensive oxidants like manganese dioxide instead of precious metal catalysts.
Q: Which solvents are preferred for the synthesis of these benzoxazinorifamycin derivatives?
A: The patent specifies that polar aprotic solvents such as dimethyl sulfoxide (DMSO), N,N-dimethylformamide (DMF), and pyridine yield excellent results, facilitating the dissolution of both the rifamycin precursor and the amine reactants.
Q: What is the antibacterial spectrum of the novel derivatives produced?
A: The synthesized derivatives exhibit strong antibacterial activity specifically against Gram-positive bacteria and acid-fast bacteria, making them highly relevant for tuberculosis and staphylococcal infection treatments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzoxazinorifamycin Derivatives Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis, leveraging deep expertise in complex antibiotic chemistry to deliver high-quality intermediates for the global pharmaceutical industry. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of benzoxazinorifamycin derivatives meets the highest international standards. Our commitment to quality and consistency makes us the preferred partner for companies seeking to secure their supply chain for next-generation antibacterial agents.
We invite you to engage with our technical procurement team to discuss your specific requirements and explore how our manufacturing capabilities can support your strategic goals. By requesting a Customized Cost-Saving Analysis, you can gain valuable insights into potential efficiencies and economic benefits tailored to your volume needs. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions with confidence and speed.
