Advanced Enzymatic Synthesis of D-N-Carbamoyl-Alpha-Amino Acids for Commercial Scale
The pharmaceutical industry continuously seeks robust methodologies for producing optically active amino acids, which serve as critical building blocks for semi-synthetic penicillins and cephalosporins. Patent CN1916164A discloses a groundbreaking method for the production of D-N-carbamoyl-alpha-amino acids from 5-substituted hydantoins using a highly efficient hydantoinase enzyme. This enzyme is produced by a novel transformant microorganism generated through recombinant DNA technology, specifically utilizing genes derived from Bacillus, Agrobacterium, or Pseudomonas species. The significance of this innovation lies in its ability to overcome the historical limitations of low enzyme yields and expensive culture media associated with earlier microbial enzymatic reactions. By leveraging specific strains such as Bacillus KNK245, the process ensures a consistent and high-volume supply of the necessary biocatalyst, fundamentally shifting the economic feasibility of large-scale D-amino acid manufacturing for global supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of D-N-carbamoyl-alpha-amino acids relied on microbial enzymatic reactions that suffered from significant inefficiencies and economic drawbacks. Prior art methods, such as those disclosed in various Japanese patent applications, utilized microorganisms that demonstrated insufficient enzyme production capabilities, necessitating large volumes of costly culture media to achieve marginal yields. Furthermore, the enzymatic activity of these conventional spore-forming thermophilic microorganisms was often only marginally higher than baseline levels, requiring extended reaction times and complex downstream processing to isolate the desired chiral intermediates. These inefficiencies created bottlenecks in the supply chain, leading to increased lead times and higher operational expenditures for manufacturers attempting to scale production to meet the demands of the antibiotic market. The reliance on wild-type strains also introduced variability in batch consistency, posing risks to quality control protocols essential for pharmaceutical grade intermediates.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a transformant microorganism engineered to possess extremely high hydantoinase production abilities, effectively resolving the yield and cost issues plaguing previous methodologies. By inserting specific DNA fragments encoding the hydantoinase gene into host microorganisms like Escherichia coli or Bacillus subtilis, the process achieves a dramatic amplification of enzymatic activity without the need for exotic or expensive growth substrates. This recombinant strategy allows for the asymmetric cleavage hydrolysis of 5-substituted hydantoins to proceed with high specificity and efficiency, accumulating substantial quantities of the target D-N-carbamoyl-alpha-amino acids in the aqueous medium. The ability to use standard nutrient media combined with the high turnover rate of the recombinant enzyme translates directly into a more streamlined manufacturing process that is both economically viable and technically superior for industrial applications seeking reliable pharmaceutical intermediate supplier partnerships.
Mechanistic Insights into Hydantoinase-Catalyzed Hydrolysis
The core of this technological advancement rests on the precise mechanistic action of the hydantoinase enzyme, which facilitates the D-specific hydrolysis of the hydantoin ring structure. The enzyme operates by recognizing the 5-substituted hydantoin substrate and catalyzing the asymmetric cleavage of the ring to form the corresponding D-N-carbamoyl-alpha-amino acid while leaving the L-isomer untouched or allowing it to racemize chemically in the reaction medium. This dynamic kinetic resolution ensures that theoretically all starting material, whether D, L, or DL-5-substituted hydantoin, can be converted into the desired D-product, maximizing atom economy and minimizing waste generation. The reaction proceeds optimally in an aqueous medium where parameters such as pH and temperature are tightly controlled to maintain enzyme stability and activity over extended periods, often ranging from 5 to 100 hours depending on the substrate load and specific strain characteristics employed in the bioreactor.
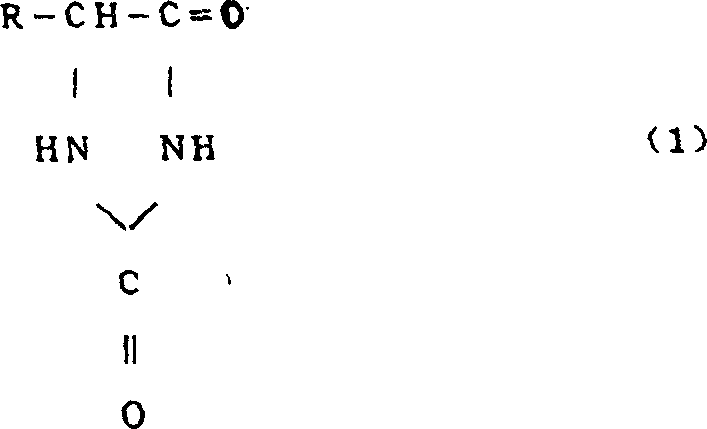
Controlling the impurity profile is paramount in the synthesis of pharmaceutical intermediates, and this enzymatic route offers inherent advantages in selectivity that chemical synthesis often struggles to match. The hydantoinase derived from strains like Agrobacterium KNK712 exhibits high stereoselectivity, ensuring that the resulting amino acid derivatives possess the correct optical rotation required for downstream drug synthesis, such as the production of D-phenylglycine. By avoiding harsh chemical reagents and extreme conditions, the process minimizes the formation of side products and degradation impurities that would otherwise require rigorous and costly purification steps. The use of immobilized enzyme preparations, as further exemplified in the patent examples, enhances this purity profile by allowing for easy separation of the biocatalyst from the product stream, thereby reducing the risk of protein contamination in the final high-purity D-amino acids intended for sensitive therapeutic applications.
How to Synthesize D-N-Carbamoyl-Alpha-Amino Acids Efficiently
Implementing this synthesis route requires a systematic approach to fermentation and biocatalysis that leverages the genetic potential of the recombinant strains. The process begins with the cultivation of the transformant in a nutrient-rich medium under aerobic conditions to induce maximum enzyme expression, followed by the harvesting of cells which may be used directly or processed into immobilized forms for repeated batch operations. Operators must carefully manage the reaction environment, maintaining specific pH ranges between 4 and 9.5 and temperatures from 10 to 80 degrees Celsius to optimize the hydrolysis rate while preserving enzyme integrity throughout the conversion cycle. Detailed standardized synthesis steps see the guide below for specific operational parameters regarding substrate feeding strategies and product isolation techniques that ensure consistent quality across large-scale production runs.
- Cultivate the transformed microorganism, such as Bacillus KNK245 or Agrobacterium KNK712, in a conventional nutrient medium containing carbon and nitrogen sources under aerobic conditions to express the hydantoinase enzyme.
- React the harvested microbial cells or enzyme extract with 5-substituted hydantoin substrates in an aqueous medium at a controlled pH and temperature to facilitate asymmetric cleavage hydrolysis.
- Isolate and purify the resulting D-N-carbamoyl-alpha-amino acids from the reaction mixture using standard separation techniques such as crystallization or ion exchange chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this recombinant enzymatic technology presents a compelling value proposition centered around cost efficiency and operational reliability. The elimination of expensive culture media components and the significant increase in enzyme yield per fermentation batch directly contribute to a reduction in the overall cost of goods sold for these critical intermediates. Furthermore, the robustness of the recombinant strains ensures a stable supply of biocatalyst, mitigating the risks associated with biological variability that often disrupt production schedules in traditional fermentation processes. This stability allows for better forecasting and inventory management, ensuring that downstream pharmaceutical manufacturers receive their materials on time without unexpected delays caused by upstream biological failures or low conversion efficiencies.
- Cost Reduction in Manufacturing: The transition to high-yield recombinant strains eliminates the need for costly media supplements and reduces the volume of bioreactor capacity required to produce a given amount of enzyme, leading to substantial cost savings in the manufacturing process. By achieving higher conversion rates and minimizing waste, the overall resource consumption per unit of product is drastically lowered, enhancing the economic viability of producing complex pharmaceutical intermediates. This efficiency gain allows suppliers to offer more competitive pricing structures without compromising on the stringent quality standards required for regulatory compliance in the global pharmaceutical market.
- Enhanced Supply Chain Reliability: The use of well-characterized recombinant microorganisms provides a consistent and reliable source of enzymatic activity, reducing the variability that can lead to batch failures and supply disruptions. This reliability is crucial for maintaining continuous production lines in the fast-paced pharmaceutical industry, where delays can have cascading effects on drug development timelines and market availability. The ability to scale the fermentation process predictably ensures that supply chain heads can secure long-term contracts with confidence, knowing that the production capacity can be ramped up to meet surging demand without significant re-engineering of the biological process.
- Scalability and Environmental Compliance: The enzymatic process operates under mild conditions and generates less hazardous waste compared to traditional chemical synthesis routes, aligning with increasingly strict environmental regulations and sustainability goals. The scalability of the fermentation and biocatalysis steps allows for seamless transition from pilot scale to commercial production, facilitating rapid market entry for new drug candidates requiring these specific chiral intermediates. Additionally, the potential for enzyme immobilization and reuse further reduces the environmental footprint by minimizing solid waste generation and lowering the consumption of fresh biocatalyst materials over the lifecycle of the production campaign.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this hydantoinase technology in industrial settings. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits and technical feasibility for potential partners evaluating this synthesis route for their supply chains. Understanding these details is essential for making informed decisions about integrating this biocatalytic process into existing manufacturing frameworks to achieve optimal efficiency and product quality.
Q: What are the advantages of using recombinant hydantoinase over conventional microbial sources?
A: Recombinant hydantoinase transformants offer significantly higher enzyme production yields compared to wild-type strains, reducing the overall cost of the biocatalyst and enabling more efficient substrate conversion rates in industrial settings.
Q: Can this process handle diverse 5-substituted hydantoin substrates?
A: Yes, the enzymatic method described supports a wide range of substituents including phenyl, hydroxyphenyl, and alkyl groups, making it versatile for producing various D-amino acid intermediates required for semi-synthetic antibiotics.
Q: How does this technology impact supply chain stability for amino acid intermediates?
A: By utilizing robust recombinant strains with high enzymatic activity, manufacturers can achieve consistent batch-to-batch quality and reduce dependency on expensive culture media, thereby enhancing supply chain reliability and scalability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable D-N-Carbamoyl-Alpha-Amino Acids Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of life-saving medications, and we are equipped to leverage advanced technologies like the one described in CN1916164A to serve your needs. Our facility boasts extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with precision and consistency. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of D-N-carbamoyl-alpha-amino acids meets the highest international standards for pharmaceutical use, providing you with a secure foundation for your drug synthesis projects.
We invite you to engage with our technical procurement team to discuss how our capabilities can align with your specific project goals and timeline requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our enzymatic processes can optimize your budget while maintaining superior quality. We encourage you to contact us today to索取 specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to being your trusted partner in the complex landscape of fine chemical manufacturing and supply chain solutions.
