Advanced Semi-Synthetic Route for Corosolic Acid: Scaling High-Purity Production for Global Pharma
Advanced Semi-Synthetic Route for Corosolic Acid: Scaling High-Purity Production for Global Pharma
The global demand for effective anti-diabetic and anti-obesity agents has placed Corosolic Acid (CA) at the forefront of pharmaceutical research, driven by its potent "glucokinin" activity that mimics insulin. However, the commercial viability of this triterpenoid has historically been constrained by the limitations of natural extraction, where content in source plants like Banaba leaves is notoriously low, often ranging between 0.01% and 1%. Addressing this critical supply bottleneck, patent CN101805389A discloses a robust semi-synthetic methodology that transforms readily available Ursolic Acid into high-purity Corosolic Acid through a streamlined three-step sequence. This technological breakthrough not only circumvents the geographical and seasonal dependencies of plant harvesting but also establishes a reproducible chemical pathway capable of delivering purity levels exceeding 98%. For R&D directors and procurement strategists, this patent represents a pivotal shift from unreliable agricultural sourcing to precise, scalable chemical manufacturing, ensuring a consistent supply of this high-value bioactive compound for the global nutraceutical and pharmaceutical markets.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the industry has relied heavily on the extraction of Corosolic Acid from specific botanical sources, primarily the leaves of Lagerstroemia speciosa (Banaba), which presents severe logistical and economic challenges. The natural abundance of the target molecule is exceptionally scarce, with hot water extracts containing merely 0.01% of the active ingredient and even concentrated alcohol extracts rarely surpassing 1% content. This inefficiency necessitates the processing of massive quantities of raw plant material, leading to exorbitant production costs and a heavy environmental footprint associated with biomass waste. Furthermore, previous semi-synthetic attempts, such as those described in patent CN1634971A, have struggled with excessive procedural complexity, involving intricate sequences of esterification, enolization, hydroboration-oxidation, and catalytic hydrogenolysis. These multi-stage processes not only suffer from low overall recovery rates but also require sophisticated equipment and rigorous control measures, rendering them economically unfeasible for large-scale commercial adoption and creating significant vulnerabilities in the supply chain for high-purity intermediates.
The Novel Approach
In stark contrast to these cumbersome legacy methods, the methodology outlined in patent CN101805389A introduces a remarkably concise and efficient synthetic strategy that leverages the structural similarity between Ursolic Acid and Corosolic Acid. By utilizing Ursolic Acid—a triterpenoid widely distributed in over 62 plant species and available in substantial quantities—as the starting feedstock, the process bypasses the need for rare botanical resources entirely. The core innovation lies in a direct functionalization strategy that introduces the critical 2-alpha-hydroxyl group through a targeted acetoxylation-reduction sequence, effectively shortening the technical route and minimizing intermediate treatment steps. This approach not only drastically simplifies the operational workflow but also enhances the overall economic viability by reducing solvent consumption, energy usage, and labor hours associated with complex purification protocols. The result is a streamlined manufacturing process that achieves an overall reaction yield of over 34%, providing a reliable and cost-effective alternative for the mass production of Corosolic Acid.
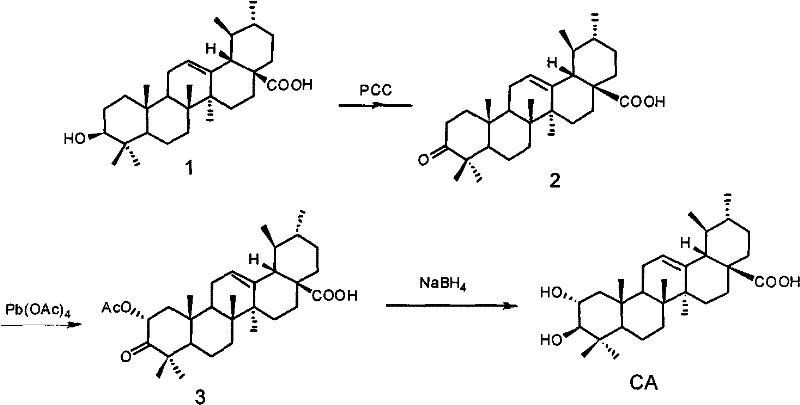
Mechanistic Insights into the Three-Step Oxidation-Acetoxylation-Reduction Sequence
The chemical elegance of this synthesis is rooted in a precise three-step transformation that meticulously modifies the ursane skeleton to install the requisite hydroxyl functionality at the C-2 position. The initial phase involves the selective oxidation of the C-3 hydroxyl group of Ursolic Acid to a ketone, typically employing pyridinium chlorochromate (PCC) or similar chromium-based oxidants in solvents like methylene dichloride at controlled temperatures ranging from -5°C to 5°C. This step generates 3-carbonyl ursolic acid with high efficiency, as evidenced by exemplary yields reaching 88%, setting the stage for the subsequent regioselective functionalization. The second and most critical step utilizes lead tetraacetate to introduce an acetoxy group at the C-2 position alpha to the newly formed carbonyl, a reaction that proceeds effectively in solvents such as benzene or acetic acid at temperatures between 50°C and 80°C. This acetoxylation is the key differentiator, as it installs the oxygen handle necessary for the final stereochemical configuration without requiring protecting group manipulations that would add unnecessary steps to the synthesis.
The final stage of the synthesis involves the stereoselective reduction of the C-2 acetoxy group back to a hydroxyl group, thereby completing the conversion to Corosolic Acid. This reduction is typically carried out using mild reducing agents such as sodium borohydride in ethanol at low temperatures (around 0°C), which ensures the preservation of other sensitive functional groups within the complex triterpenoid framework while achieving the desired stereochemistry. Following the reduction, the crude product undergoes purification via column chromatography or recrystallization, utilizing solvent systems like petroleum ether and ethyl acetate to isolate the final product with a purity exceeding 98%. This rigorous purification protocol is essential for meeting the stringent quality standards required for pharmaceutical applications, ensuring that the final API intermediate is free from residual heavy metals or organic impurities that could compromise safety or efficacy in downstream drug formulation.
How to Synthesize Corosolic Acid Efficiently
The execution of this synthesis requires careful attention to reaction conditions and reagent stoichiometry to maximize yield and minimize byproduct formation. The process begins with the oxidation of Ursolic Acid, followed by the crucial acetoxylation with lead tetraacetate, and concludes with the reduction of the acetoxy intermediate. Detailed operational parameters, including specific solvent ratios, temperature gradients, and workup procedures, are critical for reproducing the high purity and yield reported in the patent literature. For process chemists looking to implement this route, adherence to the standardized protocols ensures consistent quality and operational safety.
- Oxidize Ursolic Acid using PCC or similar oxidants to form 3-carbonyl ursolic acid.
- React the 3-carbonyl intermediate with lead tetraacetate to introduce an acetoxy group at the C-2 position.
- Reduce the acetoxy group using sodium borohydride to yield Corosolic Acid, followed by purification to >98% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this semi-synthetic route offers transformative advantages that directly address the volatility and cost inefficiencies inherent in botanical extraction. By shifting the source of raw materials from geographically constrained plant species to widely available chemical feedstocks like Ursolic Acid, manufacturers can decouple their production schedules from agricultural harvest cycles and climate variability. This strategic pivot not only stabilizes the supply chain against external shocks but also opens up opportunities for significant cost reduction in pharmaceutical intermediates manufacturing by eliminating the extensive logistics and processing costs associated with tons of plant biomass. Furthermore, the simplified three-step process reduces the capital expenditure required for specialized extraction equipment, allowing facilities to utilize standard organic synthesis reactors and thereby lowering the barrier to entry for high-volume production.
- Cost Reduction in Manufacturing: The elimination of complex multi-step sequences found in prior art, such as hydroboration and catalytic hydrogenolysis, results in a drastic simplification of the manufacturing workflow. By reducing the number of unit operations and intermediate isolation steps, the process minimizes solvent usage, energy consumption, and labor costs, leading to substantial overall savings in the cost of goods sold. Additionally, the use of commercially available and relatively inexpensive reagents like PCC and sodium borohydride avoids the need for exotic catalysts or high-pressure hydrogenation equipment, further driving down the operational expenditure required to produce high-purity Corosolic Acid.
- Enhanced Supply Chain Reliability: Sourcing Ursolic Acid as a starting material provides a robust and diversified supply base, as this compound is abundant in numerous common plant sources such as apple peels and basil, unlike the limited availability of Banaba leaves. This abundance ensures a continuous and reliable flow of raw materials, mitigating the risk of supply disruptions that frequently plague botanical supply chains due to seasonal variations or geopolitical factors. Consequently, manufacturers can maintain consistent inventory levels and meet fluctuating market demands with greater agility, securing their position as a dependable partner for downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The synthetic route is designed with scalability in mind, utilizing standard reaction conditions and solvents that are easily managed in large-scale industrial reactors. The reduction in process steps inherently lowers the generation of chemical waste and effluent, simplifying waste treatment protocols and enhancing compliance with increasingly stringent environmental regulations. This eco-friendly profile not only reduces the environmental footprint of production but also aligns with the sustainability goals of modern pharmaceutical companies, making the process attractive for long-term commercial investment and regulatory approval.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of Corosolic Acid synthesized via this patented method. These insights are derived directly from the experimental data and process descriptions provided in the patent documentation, offering clarity on yield expectations, purity profiles, and scalability potential. Understanding these fundamentals is crucial for stakeholders evaluating the feasibility of integrating this intermediate into their broader supply chain strategies.
Q: What are the advantages of this semi-synthetic route over plant extraction?
A: Plant extraction yields are extremely low (0.01-1%) and resource-intensive. This semi-synthetic method uses abundant Ursolic Acid as a starting material, achieving over 34% overall yield and >98% purity, significantly lowering production costs and ensuring supply stability.
Q: How does this method improve upon previous semi-synthetic patents?
A: Previous methods involved complex multi-step sequences including esterification and hydroboration. This patented approach streamlines the process into three direct steps (oxidation, acetoxylation, reduction), reducing intermediate handling and operational complexity.
Q: Is this process scalable for industrial manufacturing?
A: Yes, the process utilizes standard organic reagents like PCC, lead tetraacetate, and sodium borohydride under controllable temperatures (-5°C to 80°C), making it highly suitable for scale-up from laboratory to commercial tonnage production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Corosolic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a stable and high-quality supply of advanced pharmaceutical intermediates like Corosolic Acid to support the development of next-generation metabolic therapies. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent batches that meet stringent purity specifications. Our state-of-the-art facilities are equipped with rigorous QC labs capable of performing comprehensive analytical testing, guaranteeing that every shipment of Corosolic Acid adheres to the highest international standards for identity, potency, and impurity control.
We invite forward-thinking pharmaceutical and nutraceutical companies to collaborate with us to leverage this efficient synthetic technology for their product pipelines. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements, along with specific COA data and route feasibility assessments to validate the integration of our material into your manufacturing processes. Let us help you optimize your supply chain and accelerate your time-to-market with our reliable, high-performance Corosolic Acid solutions.
