Advanced Zinc-Catalyzed Synthesis of 1,2,3,4-Tetrazole Intermediates for Global Pharma Supply Chains
The landscape of fine chemical manufacturing is undergoing a significant transformation driven by the urgent need for greener, more efficient synthetic pathways, particularly for high-value heterocyclic scaffolds like 1,2,3,4-tetrazoles. Patent CN1304381C introduces a groundbreaking chemical synthesis method that leverages zinc trifluoromethanesulfonate as a Lewis acid catalyst in an aqueous medium, marking a departure from traditional organic solvent-based systems. This innovation addresses critical pain points for R&D Directors and Supply Chain Heads by offering a route that combines high reaction yields, generally exceeding 80%, with exceptional environmental compatibility. The strategic shift to water as a solvent not only mitigates the risks associated with volatile organic compounds but also simplifies the downstream processing infrastructure required for commercial scale-up. For global procurement teams, this patent represents a viable pathway to secure a reliable pharma intermediates supplier capable of delivering consistent quality while adhering to increasingly stringent regulatory frameworks regarding industrial emissions and waste management.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,2,3,4-tetrazolium compounds has relied heavily on the use of amine or amine salt catalysts dissolved in organic solvents, a methodology exemplified by prior art such as US4,097,479. These conventional processes typically require equimolar amounts of amine salts, which not only drives up the raw material costs but also complicates the purification process due to the difficulty in separating the catalyst from the final product. Furthermore, these reactions often necessitate elevated temperatures and extended reaction times, leading to energy inefficiencies and potential thermal degradation of sensitive functional groups on the nitrile substrate. The reliance on organic solvents introduces significant safety hazards related to flammability and toxicity, imposing heavy burdens on facility safety protocols and waste treatment systems. Consequently, the overall production cost is inflated by the need for extensive solvent recovery units and the disposal of hazardous chemical waste, making cost reduction in pharmaceutical manufacturing a challenging objective under legacy protocols.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a catalytic amount of zinc trifluoromethanesulfonate dissolved in water, creating a homogeneous reaction system that operates under significantly milder conditions. This method allows for the reaction between nitriles and sodium azide to proceed efficiently at temperatures ranging from 50°C to 100°C, drastically reducing the energy input required compared to high-temperature organic syntheses. The use of water as the primary solvent eliminates the need for expensive and hazardous organic media, thereby streamlining the workup procedure to a simple acid neutralization and filtration step. 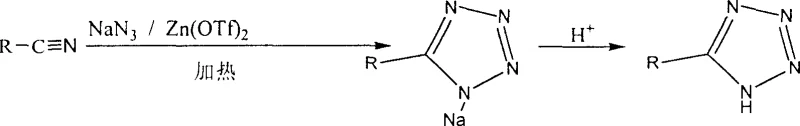 This streamlined workflow not only enhances operational safety but also facilitates easier commercial scale-up of complex polymer additives and pharmaceutical intermediates by removing solvent exchange bottlenecks. The ability to achieve high conversion rates with minimal catalyst loading demonstrates a robust process design that is inherently more sustainable and economically viable for large-volume production.
This streamlined workflow not only enhances operational safety but also facilitates easier commercial scale-up of complex polymer additives and pharmaceutical intermediates by removing solvent exchange bottlenecks. The ability to achieve high conversion rates with minimal catalyst loading demonstrates a robust process design that is inherently more sustainable and economically viable for large-volume production.
Mechanistic Insights into Zn(OTf)2-Catalyzed Cyclization
From a mechanistic perspective, the role of zinc trifluoromethanesulfonate is pivotal in activating the nitrile group towards nucleophilic attack by the azide ion, facilitating the formation of the tetrazole ring through a coordinated transition state. The zinc center acts as a Lewis acid, coordinating with the nitrogen atom of the nitrile to increase its electrophilicity, which lowers the activation energy barrier for the cycloaddition reaction. This catalytic cycle is highly efficient, allowing for the turnover of a large number of substrate molecules per unit of catalyst, which is essential for maintaining low production costs in an industrial setting. The resulting tetrazole products exist in tautomeric equilibrium, as illustrated in the structural diagrams, where the proton can migrate between the nitrogen atoms within the ring system depending on the electronic nature of the substituent R1. 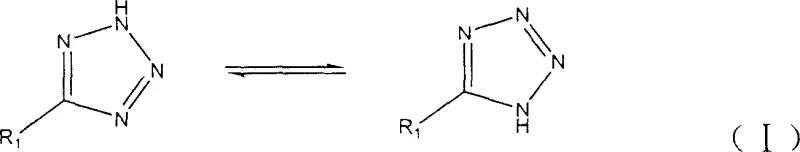 Understanding this tautomerism is crucial for R&D teams focusing on purity and impurity profiles, as the stability of specific tautomers can influence the crystallization behavior and final physical properties of the active pharmaceutical ingredient. The aqueous environment further stabilizes the ionic intermediates involved in the reaction, ensuring a clean transformation with minimal side reactions.
Understanding this tautomerism is crucial for R&D teams focusing on purity and impurity profiles, as the stability of specific tautomers can influence the crystallization behavior and final physical properties of the active pharmaceutical ingredient. The aqueous environment further stabilizes the ionic intermediates involved in the reaction, ensuring a clean transformation with minimal side reactions.
Impurity control is another critical aspect where this mechanism offers distinct advantages, particularly regarding the removal of unreacted starting materials which can compromise the quality of high-purity OLED material or drug intermediates. The patent describes a specific purification strategy where, if the reactant nitrile is a solid, the reaction mixture can be filtered prior to acidification to remove any unreacted nitrile that might otherwise co-precipitate with the product. This pre-filtration step ensures that the final filter cake consists almost exclusively of the desired tetrazole derivative, significantly enhancing the crude purity before any recrystallization steps are applied. By preventing the entrapment of starting materials in the product lattice, the process reduces the burden on downstream purification units and ensures that the stringent purity specifications required by top-tier pharmaceutical clients are met consistently. This level of control over the impurity profile is a key differentiator for any reliable agrochemical intermediate supplier aiming to serve regulated markets.
How to Synthesize 5-Phenyl-1H-Tetrazole Efficiently
To implement this synthesis effectively, operators must adhere to precise stoichiometric ratios and temperature controls to maximize yield and catalyst efficiency. The process begins with the preparation of the catalyst solution, followed by the controlled addition of reactants to maintain a safe exotherm profile during the azide coupling. Detailed standard operating procedures regarding mixing speeds, heating ramps, and filtration techniques are essential to replicate the laboratory success on a multi-ton scale.
- Prepare an aqueous solution of zinc trifluoromethanesulfonate and mix with nitrile and sodium azide at a molar ratio of 1: 1.0-4.0:0.02-1.0.
- Heat the reaction mixture to 50-100°C and maintain for 1-24 hours to ensure complete conversion of the nitrile starting material.
- Neutralize the reaction liquid with acid to pH 5-6, filter to collect the solid product, and retain the filtrate for catalyst recycling.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this zinc-catalyzed aqueous synthesis translates into tangible strategic benefits that extend beyond simple unit price considerations. The elimination of organic solvents removes a major variable from the supply chain, reducing exposure to volatile petrochemical pricing and the logistical complexities associated with transporting and storing hazardous fluids. This shift contributes to substantial cost savings by simplifying the infrastructure requirements for the manufacturing plant, as there is no longer a need for explosion-proof reactors or complex solvent recovery distillation columns. Furthermore, the ability to recycle the catalyst solution directly from the filtrate back into the next batch creates a closed-loop system that minimizes raw material consumption and waste disposal fees. These factors combined result in a more resilient supply chain that is less susceptible to external market shocks and regulatory changes regarding environmental compliance.
- Cost Reduction in Manufacturing: The economic model of this process is fundamentally superior due to the drastic reduction in auxiliary material costs and waste treatment expenses. By replacing expensive organic solvents with water and utilizing a recyclable zinc catalyst, the variable cost per kilogram of product is significantly lowered without compromising on quality. The high reaction yields generally above 80% mean that less raw material is wasted, improving the overall atom economy of the process. Additionally, the simplified workup procedure reduces labor hours and utility consumption, as there is no need for energy-intensive solvent stripping or drying operations. These cumulative efficiencies allow for a more competitive pricing structure while maintaining healthy margins, addressing the core mandate for cost reduction in electronic chemical manufacturing and related sectors.
- Enhanced Supply Chain Reliability: Operational reliability is greatly enhanced by the inherent safety and simplicity of the aqueous reaction system. The use of water as a solvent eliminates fire hazards associated with organic volatiles, reducing the risk of unplanned shutdowns due to safety incidents or regulatory inspections. The mild reaction conditions (50-100°C) place less stress on reactor equipment, extending the lifespan of capital assets and reducing maintenance downtime. Moreover, the robustness of the catalyst system ensures consistent batch-to-batch performance, which is critical for reducing lead time for high-purity pharmaceutical intermediates. Suppliers can guarantee delivery schedules with greater confidence, knowing that the process is not dependent on hard-to-source specialty solvents or sensitive anhydrous conditions that could disrupt production flow.
- Scalability and Environmental Compliance: Scaling this technology from pilot plant to full commercial production is straightforward due to the absence of mass transfer limitations often encountered in heterogeneous organic systems. The homogeneous aqueous phase ensures efficient heat and mass transfer, allowing for larger reactor volumes without the need for complex engineering modifications. From an environmental standpoint, the process generates basically no three wastes, aligning perfectly with global sustainability goals and corporate ESG mandates. The aqueous effluent is easier to treat than organic waste streams, and the recycling of the catalyst minimizes the discharge of heavy metals. This environmental stewardship not only protects the company's reputation but also future-proofs the supply chain against tightening environmental regulations that could otherwise render older technologies obsolete.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the patent specifications and practical application scenarios. Understanding these details helps stakeholders evaluate the feasibility of integrating this method into their existing supply networks.
Q: Can the zinc catalyst be recovered in this tetrazole synthesis process?
A: Yes, the patent specifies that the filtrate (Solution B) obtained after product filtration can be directly reused as the catalyst solution (Solution A) for subsequent batches, significantly reducing waste.
Q: What are the typical reaction yields for this aqueous method?
A: The documented examples demonstrate reaction yields generally above 80%, with specific embodiments achieving up to 98% purity and high conversion rates under optimized conditions.
Q: How does this method improve environmental compliance compared to traditional routes?
A: By using water as the sole solvent instead of organic solvents and enabling catalyst recycling, the process minimizes hazardous waste generation and eliminates the need for complex solvent recovery systems.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2,3,4-Tetrazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this zinc-catalyzed technology and have integrated similar green chemistry principles into our CDMO operations to serve the global market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory patent to industrial reality is seamless and efficient. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to verify that every batch of 1,2,3,4-tetrazole derivatives meets the exacting standards required by the pharmaceutical and agrochemical industries. Our infrastructure is designed to handle the specific nuances of aqueous catalytic systems, providing a secure and compliant manufacturing environment for our partners.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this greener methodology for your supply chain. We encourage you to contact us to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data and expert analysis. Partnering with us ensures access to cutting-edge chemical technologies backed by a commitment to quality, sustainability, and long-term supply security.
