Advanced Synthesis of Amisulpride Impurity H: Technical Breakthroughs for Commercial Scale-Up
Advanced Synthesis of Amisulpride Impurity H: Technical Breakthroughs for Commercial Scale-Up
In the highly regulated landscape of pharmaceutical manufacturing, the precise characterization and quantification of impurities are non-negotiable requirements for regulatory approval. Patent CN114507174A introduces a significant technological advancement in the synthesis of Amisulpride Impurity H, a critical reference standard required for the quality control of the antipsychotic drug Amisulpride. This patent details a novel, streamlined methodology that overcomes the inefficiencies of prior art by utilizing a one-step preparation of the N-methylated pyrrolidine intermediate followed by a robust mixed anhydride coupling strategy. For R&D directors and procurement specialists, this innovation represents a pivotal shift towards more efficient, cost-effective, and high-purity production of complex pharmaceutical intermediates. The technical depth of this approach ensures that supply chains can rely on consistent quality while minimizing the operational burdens associated with multi-step synthetic routes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Amisulpride Impurity H has been plagued by inefficient multi-step sequences that drive up costs and complicate supply chain logistics. As illustrated in prior art such as Patent CN113061108A, the conventional route requires a tedious three-step process to synthesize the key amine intermediate, N-ethyl-2-methylaminomethylpyrrolidine. This typically involves protecting the primary amine with a Boc group, performing a methylation substitution using reagents like potassium iodide, and finally executing an acidic deprotection step. Each additional unit operation introduces potential yield losses, increases solvent consumption, and generates more waste, thereby inflating the environmental footprint and production costs. Furthermore, the final coupling step in older methods often relies on expensive activating agents like carbonylbisimidazole (CDI), which not only adds to the raw material cost but also generates imidazole by-products that can be difficult to remove, compromising the final purity of the impurity standard.
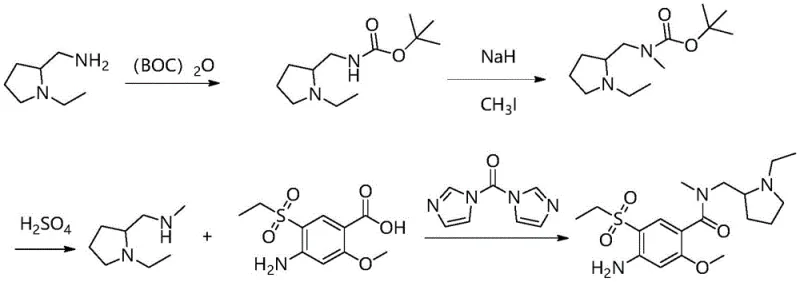
The Novel Approach
The methodology disclosed in CN114507174A fundamentally reengineers this workflow by collapsing the amine synthesis into a highly efficient protection-reduction sequence. Instead of a disjointed three-step protection-substitution-deprotection cycle, the new process employs di-tert-butyl dicarbonate (Boc2O) followed immediately by reduction with lithium aluminum hydride (LiAlH4). This elegant transformation directly converts the protected carbamate into the desired secondary amine in a single operational flow, drastically reducing processing time and equipment usage. Additionally, the coupling strategy has been optimized to use ethyl chloroformate and a tertiary amine base to generate a mixed anhydride in situ. This activation method is not only significantly cheaper than CDI but also proceeds under mild conditions, typically between -5°C and 10°C, ensuring minimal degradation of the sensitive amino acid substrate. The result is a robust process capable of delivering the target impurity with exceptional purity profiles, as evidenced by the reported 99.1% purity in experimental examples.
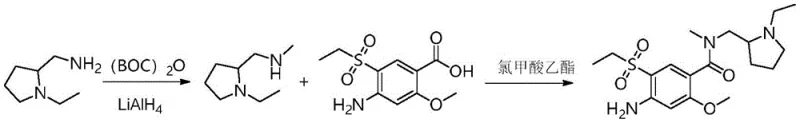
Mechanistic Insights into Mixed Anhydride Activation and Reductive Transformation
The core chemical innovation lies in the dual optimization of the amine synthesis and the amide bond formation. In the first stage, the reaction of N-ethyl-2-aminomethylpyrrolidine with Boc2O forms a stable carbamate intermediate. The subsequent addition of LiAlH4 acts as a powerful hydride donor, reducing the carbamate carbonyl group directly to a methylene group. This reductive cleavage is highly selective, preserving the pyrrolidine ring integrity while successfully installing the requisite N-methyl group. The mechanism avoids the harsh conditions often associated with direct alkylation, thereby suppressing the formation of quaternary ammonium salts or over-alkylated by-products that frequently plague traditional methylation routes. The use of anhydrous solvents like tetrahydrofuran (THF) is critical here to maintain the activity of the hydride reagent and ensure complete conversion.
In the second stage, the formation of the amide bond between Amic acid and the synthesized amine is achieved via a mixed anhydride mechanism. At controlled low temperatures (0°C to 5°C), ethyl chloroformate reacts with the carboxylate anion of Amic acid (generated by triethylamine) to form a reactive mixed anhydride species. This intermediate is highly electrophilic yet sufficiently stable to allow for the controlled addition of the nucleophilic amine. This kinetic control is vital for minimizing racemization (though less relevant for this specific achiral acid) and preventing the hydrolysis of the activated species. The subsequent nucleophilic attack by the secondary amine displaces the ethyl carbonate leaving group, forming the stable amide linkage found in Impurity H. This pathway is superior because the by-products (ethanol, CO2, and triethylamine salts) are easily removed during the aqueous workup, contributing to the high purity observed in the final crystallized product.
How to Synthesize Amisulpride Impurity H Efficiently
The execution of this synthesis requires precise adherence to temperature controls and stoichiometric ratios to maximize yield and purity. The process begins with the protection of the starting amine in dichloromethane at 0°C, followed by the critical reduction step using LiAlH4 in THF, which must be handled under inert atmosphere due to the pyrophoric nature of the reagent. The final coupling reaction demands careful thermal management, initiating at sub-zero temperatures to form the mixed anhydride before allowing the reaction to warm slightly to drive completion. For a detailed, step-by-step breakdown of the exact molar ratios, solvent volumes, and work-up procedures validated in the patent examples, please refer to the standardized protocol below.
- Protect N-ethyl-2-aminomethylpyrrolidine with di-tert-butyl dicarbonate (Boc2O) and subsequently reduce the carbamate intermediate using Lithium Aluminum Hydride (LiAlH4) to obtain N-ethyl-2-methylaminomethylpyrrolidine.
- Activate 4-amino-5-ethylsulfonyl-2-methoxybenzoic acid (Amic acid) using ethyl chloroformate and triethylamine at low temperature (-5 to 10°C) to form a mixed anhydride.
- React the activated acid intermediate with the synthesized N-ethyl-2-methylaminomethylpyrrolidine, followed by pH adjustment and crystallization to isolate high-purity Impurity H.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers tangible strategic benefits that extend beyond simple chemical curiosity. The primary advantage is the drastic simplification of the manufacturing workflow, which directly translates to reduced operational expenditures. By eliminating two full synthetic steps in the preparation of the amine intermediate, manufacturers can significantly lower labor costs, energy consumption, and solvent waste disposal fees. The replacement of expensive coupling reagents like CDI with commodity chemicals such as ethyl chloroformate further drives down the Bill of Materials (BOM) cost, making the production of this high-value impurity standard much more economically viable. These efficiencies allow suppliers to offer more competitive pricing without compromising on the stringent quality specifications required by global pharmaceutical regulators.
- Cost Reduction in Manufacturing: The streamlined process eliminates the need for multiple isolation and purification stages that are typical in the conventional three-step amine synthesis. By combining protection and reduction into a seamless sequence, the process minimizes material loss between steps, leading to a higher overall yield per batch. Furthermore, the use of ethyl chloroformate as an activating agent represents a substantial cost saving compared to heterocyclic coupling agents, as it is a bulk commodity chemical with a stable global supply chain. This reduction in raw material complexity and processing time results in a leaner manufacturing model that is less susceptible to price volatility in the specialty chemical market.
- Enhanced Supply Chain Reliability: Simplifying the synthetic route inherently reduces the risk of supply chain disruptions. Fewer steps mean fewer opportunities for batch failures, equipment bottlenecks, or quality deviations that could delay shipments. The reliance on widely available reagents like Boc2O, LiAlH4, and ethyl chloroformate ensures that production is not dependent on niche suppliers with long lead times. This robustness allows for more accurate demand forecasting and inventory planning, ensuring that critical reference standards are available exactly when R&D and QC teams need them for method validation and stability testing.
- Scalability and Environmental Compliance: The reaction conditions described in the patent are mild and utilize standard organic solvents like dichloromethane, acetone, and THF, which are well-understood in terms of safety and waste management. The absence of heavy metal catalysts or exotic reagents simplifies the environmental impact assessment and wastewater treatment processes. This makes the technology highly scalable from kilogram-level laboratory synthesis to multi-ton commercial production without requiring significant capital investment in specialized reactor infrastructure. The ability to scale efficiently ensures that the supply of Amisulpride Impurity H can grow in tandem with the global demand for the parent drug, securing long-term supply continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of Amisulpride Impurity H. These insights are derived directly from the experimental data and claims presented in patent CN114507174A, providing a clear understanding of the process capabilities and limitations for potential partners.
Q: How does the new synthesis method improve upon previous patents like CN113061108A?
A: The novel method described in CN114507174A reduces the synthesis of the key amine intermediate from three steps to a streamlined one-step protection-reduction sequence. Furthermore, it replaces the expensive carbonylbisimidazole (CDI) coupling agent with a cost-effective chloroformate/acid-binding agent system, significantly simplifying the process and improving overall yield.
Q: What represents the critical quality attribute for this impurity standard?
A: High purity is paramount for reference standards used in HPLC quantification. The patented process achieves a purity of 99.1% through precise temperature control during the coupling reaction (0°C to 28°C) and optimized crystallization conditions (pH 9-11, 0-5°C), ensuring accurate impurity profiling for regulatory submissions.
Q: Is this synthesis route scalable for industrial production?
A: Yes, the route utilizes common industrial reagents such as ethyl chloroformate, triethylamine, and lithium aluminum hydride. The reaction conditions are mild (-5°C to 28°C), and the work-up involves standard aqueous washes and crystallization, making it highly suitable for commercial scale-up without requiring specialized exotic catalysts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Amisulpride Impurity H Supplier
At NINGBO INNO PHARMCHEM, we recognize that the availability of high-quality impurity standards is the cornerstone of robust pharmaceutical quality control. Our technical team has extensively analyzed the pathways described in CN114507174A and possesses the expertise to implement this advanced synthesis at scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of Amisulpride Impurity H meets the exacting standards required for regulatory filings and analytical method validation.
We invite you to collaborate with us to optimize your supply chain for this critical intermediate. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our implementation of this novel synthetic technology can enhance your operational efficiency and reduce your overall cost of goods sold.
