Revolutionizing Isoindoline Production: A Scalable Catalytic Hydrogenation Route for High-Value Diabetes Intermediates
Revolutionizing Isoindoline Production: A Scalable Catalytic Hydrogenation Route for High-Value Diabetes Intermediates
The pharmaceutical industry constantly seeks robust, scalable synthetic routes for complex heterocyclic intermediates that serve as the backbone for next-generation therapeutics. A pivotal advancement in this domain is detailed in Chinese Patent CN1197848C, which discloses a highly efficient industrial process for the preparation of isoindoline. This specific heterocyclic amine is not merely a commodity chemical but a critical building block for the synthesis of potent insulin secretagogues, specifically the compound of Formula (I), known chemically as 2-(S)-benzyl-4-oxo-4-(cis-perhydroisoindol-2-yl)butyric acid. The significance of this patent lies in its departure from traditional, cumbersome methodologies, offering a streamlined catalytic hydrogenation pathway that utilizes commercially available phthalonitrile as the starting material. By eliminating the need for ammonia and leveraging specific platinum-based catalysis, this technology addresses long-standing challenges regarding yield, purity, and environmental safety, positioning it as a cornerstone for reliable pharmaceutical intermediate supplier networks aiming to support the production of non-insulin-dependent diabetes treatments.
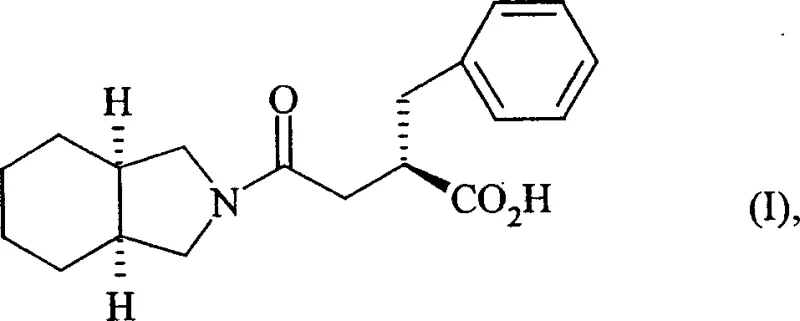
The strategic value of this synthesis cannot be overstated for R&D directors evaluating supply chain resilience. The compound of Formula (I) and its pharmaceutically acceptable salts exhibit valuable pharmacological properties as potent insulin secretors. However, the industrial viability of such active pharmaceutical ingredients (APIs) is entirely dependent on the availability of high-quality precursors. Historically, the synthesis of isoindoline has been plagued by low yields and hazardous reagents. The methodology outlined in CN1197848C resolves these bottlenecks by achieving yields greater than 75% with exceptional purity profiles. This breakthrough ensures that downstream manufacturers can secure a consistent supply of high-purity pharmaceutical intermediates, thereby reducing the risk of production delays and ensuring the continuity of essential diabetes medication supplies globally.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the chemical landscape for producing isoindoline was fraught with inefficiencies and safety hazards that made industrial scale-up economically unviable. Historical literature describes various approaches, such as the electrolytic or chemical reduction of phthalimides, which consistently failed to deliver yields exceeding 50%, resulting in substantial material waste and increased cost of goods sold. Another prevalent method involved the cyclization of α,α'-dibromo-xylene in the presence of p-toluenesulfonamide; however, this route suffered from similarly low yields and the distinct disadvantage of utilizing highly lacrimogenic starting materials, posing severe occupational health risks to plant personnel. Furthermore, processes relying on hexamethylenetetramine reactions were excessively lengthy and still failed to produce satisfactory yields. Perhaps most critically, earlier hydrogenation attempts using nickel or cobalt catalysts in ammonia-containing mixtures required extreme pressures (100-120 atm) and temperatures, yet proved irreproducible and environmentally burdensome due to the necessity of specialized equipment to handle toxic ammonia emissions.
The Novel Approach
In stark contrast to these legacy methods, the novel approach disclosed in the patent utilizes a direct catalytic hydrogenation of phthalonitrile that fundamentally simplifies the synthetic architecture. This method operates in a single step, bypassing the need for complex protection-deprotection sequences or hazardous alkylating agents. By employing a specific 5% Platinum on Carbon (Pt/C) catalyst system, the process achieves conversion rates that were previously unattainable, delivering isoindoline with yields surpassing 75% and purity levels that meet stringent pharmaceutical standards. Crucially, this route completely eliminates the use of ammonia, thereby removing the requirement for expensive, specialized containment infrastructure and significantly reducing the environmental footprint of the manufacturing process. The ability to use cheap, commercially available starting materials like phthalonitrile, combined with a simplified workup procedure involving distillation and salt precipitation, represents a paradigm shift in cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Pt/C-Catalyzed Hydrogenation
The success of this synthetic route hinges on the precise selection of the catalytic system and reaction medium, a detail of paramount importance for technical teams assessing process robustness. Extensive experimentation revealed that the choice of metal catalyst is not merely a variable of optimization but a binary determinant of success. As demonstrated in the patent data, conventional hydrogenation catalysts such as Palladium on Carbon (Pd/C), Raney Nickel, Rhodium on Carbon (Rh/C), and Ruthenium on Carbon (Ru/C) resulted in virtually zero conversion (0-1%) even after prolonged reaction times of 20 hours. In contrast, the 5% Pt/C catalyst facilitated rapid conversion, achieving nearly 90% yield within just 6 hours. This suggests a unique mechanistic interaction between the platinum surface and the nitrile groups of phthalonitrile that enables the selective reduction to the cyclic amine without excessive over-reduction or polymerization, a common side reaction in nitrile hydrogenations.
Furthermore, the solvent system plays a critical role in stabilizing the reaction intermediates and facilitating mass transfer of hydrogen. The patent explicitly identifies tetrahydrofuran (THF) and dimethoxyethane as the only viable solvents, with THF showing superior performance. The presence of water must be strictly controlled to less than 10%, preferably below 5%, to maintain catalyst activity. Alternative solvents like ethanol or dioxane led to poor conversions (40-49%), indicating that solvent polarity and coordination ability are key factors in the catalytic cycle. This deep understanding of the reaction parameters allows for precise impurity control; specifically, the process limits the formation of 2-methylbenzylamine, a structurally related impurity, to less than 1.5%, and preferably below 0.2%. Such tight control over the impurity profile is essential for meeting the rigorous specifications required for high-purity OLED material or pharmaceutical grade intermediates.
How to Synthesize Isoindoline Efficiently
Implementing this catalytic hydrogenation process requires adherence to specific operational parameters to ensure safety and reproducibility on a commercial scale. The synthesis involves charging an autoclave with phthalonitrile dissolved in anhydrous tetrahydrofuran, followed by the addition of the 5% Pt/C catalyst at a loading of 10-25% by weight. The reaction is conducted under a hydrogen pressure of 100-180 bar, preferably 150-180 bar, at temperatures ranging from 50-70°C. Following the reaction, the catalyst is removed by filtration, and the solvent is distilled off, allowing the isolation of isoindoline either by vacuum distillation or by conversion to its hydrochloride salt using ethyl acetate and HCl. For a detailed, step-by-step breakdown of the standardized operating procedures and safety protocols required for this synthesis, please refer to the technical guide below.
- Load an autoclave with commercial phthalonitrile dissolved in tetrahydrofuran (THF) and add 5% Pt/C catalyst at a loading of 10-25% by weight relative to the substrate.
- Purge the reactor with nitrogen, heat the mixture to 60°C, and pressurize with hydrogen gas to 180 bar for a reaction duration of approximately 6 hours.
- Filter off the catalyst, distill off the solvent under atmospheric pressure, and purify the residue by vacuum distillation or precipitation as the hydrochloride salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers tangible strategic benefits that extend beyond simple chemistry. The elimination of ammonia from the process flow removes a significant logistical and regulatory burden, as ammonia handling requires specialized storage, ventilation, and emergency response systems that drive up capital expenditure. By switching to a closed-loop hydrogenation system using solid catalysts and organic solvents, facilities can significantly reduce their environmental compliance costs and simplify their operational workflows. Additionally, the use of phthalonitrile, a widely available commodity chemical, ensures that the supply chain is not dependent on niche or custom-synthesized starting materials, thereby enhancing supply security and mitigating the risk of raw material shortages that often plague the fine chemical sector.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven primarily by the dramatic improvement in yield and the simplification of the purification train. Traditional methods yielding less than 50% effectively double the raw material cost per kilogram of product, whereas this new method delivers yields greater than 75%, representing a substantial reduction in material consumption. Furthermore, the high selectivity of the Pt/C catalyst minimizes the formation of difficult-to-remove byproducts, reducing the need for complex chromatographic purifications or multiple recrystallizations. This streamlined downstream processing translates directly into lower utility costs, reduced solvent consumption, and shorter batch cycle times, all of which contribute to a more competitive cost structure for the final intermediate.
- Enhanced Supply Chain Reliability: Reliability in the supply of critical intermediates is paramount for pharmaceutical manufacturers facing strict regulatory timelines. This process enhances reliability by utilizing robust, well-understood unit operations such as high-pressure hydrogenation and distillation, which are easily scalable from pilot plant to multi-ton production. The avoidance of unstable or hazardous reagents like α,α'-dibromo-xylene reduces the risk of unplanned shutdowns due to safety incidents or regulatory inspections. Moreover, the stability of the isoindoline hydrochloride salt form allows for easier storage and transportation, providing flexibility in inventory management and ensuring that just-in-time delivery schedules can be met without compromising product integrity.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces unforeseen challenges, but the linear nature of this hydrogenation reaction facilitates smooth technology transfer. The reaction conditions, while requiring high-pressure equipment, are standard in modern fine chemical manufacturing, meaning that existing infrastructure can often be utilized without major retrofitting. From an environmental perspective, the absence of ammonia emissions and the reduction in waste generation due to higher yields align perfectly with green chemistry principles. This not only simplifies waste treatment protocols but also positions the supply chain favorably against increasingly stringent global environmental regulations, ensuring long-term operational sustainability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this isoindoline synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these nuances is critical for technical teams evaluating the feasibility of adopting this route for their specific production needs.
Q: Why is Platinum on Carbon (Pt/C) preferred over other catalysts for this synthesis?
A: According to patent data, common catalysts like Pd/C, Raney Nickel, Rh/C, and Ru/C result in negligible conversion (0-1%) even after 20 hours. Only 5% Pt/C achieves high conversion (approx. 90%) within a reasonable 6-hour timeframe.
Q: What are the critical solvent requirements for this hydrogenation process?
A: The process requires tetrahydrofuran (THF), optionally with less than 10% water, or dimethoxyethane. Solvents like ethanol or dioxane result in significantly lower yields (40-49%) or complete failure.
Q: How does this method improve impurity profiles compared to prior art?
A: This ammonia-free route produces isoindoline hydrochloride with extremely high purity (>98.5%) and critically low levels of the 2-methylbenzylamine impurity (<1.5%), which is essential for downstream pharmaceutical quality.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isoindoline Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex pharmaceutical intermediates requires more than just a patent; it demands engineering excellence and unwavering commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We understand the critical nature of isoindoline in the synthesis of diabetes therapeutics and have established stringent purity specifications and rigorous QC labs to guarantee that every batch meets the exacting standards required by global regulatory bodies. Our facility is equipped to handle high-pressure hydrogenation safely and efficiently, making us an ideal partner for your long-term supply needs.
We invite you to collaborate with us to leverage this advanced synthetic technology for your projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this optimized route can improve your margins. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in catalytic hydrogenation can become a cornerstone of your supply chain strategy.
