Advanced Synthesis of 4-(N,N-Dimethyl)aminopyridine Derivatives for Commercial Scale-up
Introduction to Novel DMAP Derivative Synthesis
The landscape of asymmetric organocatalysis is continuously evolving, driven by the urgent need for cost-effective and scalable chiral auxiliaries in the pharmaceutical industry. Patent CN101891678A introduces a significant advancement in the synthesis of 4-(N,N-dimethyl)aminopyridine (DMAP) derivatives, specifically targeting the limitations of previous generation catalysts. This technology leverages a palladium-catalyzed Heck coupling strategy to attach unsaturated side chains directly to the pyridine core, bypassing the laborious multi-step syntheses associated with ferrocene-based analogues. By utilizing 3-bromo-4-(N,N-dimethyl)aminopyridine as a key starting material, the inventors have established a versatile platform capable of generating a diverse library of eleven distinct derivatives under relatively mild thermal conditions or rapid microwave irradiation. This approach not only streamlines the manufacturing workflow but also opens new avenues for tuning the steric and electronic properties of organocatalysts used in the production of high-purity pharmaceutical intermediates.
Furthermore, the patent highlights the successful application of these novel derivatives in the asymmetric kinetic resolution of secondary alcohols, such as 1-phenylethanol. While the enantioselectivity observed is moderate, the trade-off between synthetic accessibility and catalytic performance presents a compelling value proposition for process chemists. The ability to access these functionalized pyridine scaffolds without relying on expensive transition metal complexes or harsh cryogenic environments aligns perfectly with the industry's shift towards greener, more sustainable chemical manufacturing. For procurement and supply chain leaders, this represents a tangible opportunity to secure reliable sources of chiral catalysts that do not carry the heavy logistical burden of specialized organometallic reagents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of high-performance chiral DMAP catalysts has been dominated by the incorporation of bulky, rigid scaffolds such as ferrocene, as pioneered by researchers like Gregory C. Fu. While these ferrocene-based derivatives (e.g., structures 1a-1d in the background art) exhibit exceptional stereoselectivity in certain kinetic resolutions, their synthesis is fraught with significant industrial challenges. The construction of these molecules typically requires multiple synthetic steps involving sensitive organolithium reagents, strict anhydrous and oxygen-free conditions, and cryogenic temperatures often dropping to -78°C. Such demanding parameters severely limit the scalability of these catalysts, making them prohibitively expensive for large-scale API manufacturing. Additionally, the reliance on stoichiometric amounts of hazardous reagents increases waste generation and complicates safety protocols, creating bottlenecks for supply chain continuity and environmental compliance in commercial settings.
The Novel Approach
In stark contrast, the methodology disclosed in CN101891678A offers a paradigm shift by employing a direct C-C bond formation strategy via Heck coupling. This novel approach utilizes 3-bromo-4-(N,N-dimethyl)aminopyridine, a commercially accessible building block, which reacts efficiently with various vinyl compounds in the presence of a palladium catalyst and a base. The reaction conditions are remarkably forgiving, operating effectively at temperatures between 80°C and 160°C, or alternatively, utilizing microwave energy to complete the transformation in mere seconds. This drastic reduction in reaction time and the elimination of cryogenic requirements fundamentally alter the economic model of catalyst production. By simplifying the synthetic route to a single coupling step followed by straightforward purification, this method significantly lowers the barrier to entry for producing functionalized DMAP derivatives, thereby enhancing the feasibility of cost reduction in pharmaceutical intermediates manufacturing.
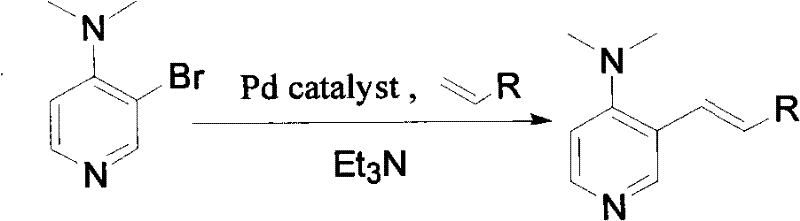
Mechanistic Insights into Pd-Catalyzed Heck Coupling
The core of this innovation lies in the palladium-catalyzed cross-coupling mechanism, which facilitates the attachment of diverse functional groups to the electron-deficient pyridine ring. The reaction initiates with the oxidative addition of the palladium(0) species into the carbon-bromine bond of the 3-bromo-DMAP substrate, forming a reactive organopalladium intermediate. Subsequent coordination and insertion of the vinyl compound (such as ethyl acrylate or acrylonitrile) into the palladium-carbon bond extends the carbon skeleton, introducing the desired unsaturated side chain. The presence of a base, typically triethylamine or pyridine, is crucial for the final reductive elimination step, which regenerates the active palladium catalyst and releases the coupled product. This mechanistic pathway is highly robust, tolerating a variety of electron-withdrawing and electron-donating groups on the vinyl partner, as evidenced by the successful synthesis of esters, amides, and nitriles described in the patent examples.
Beyond the initial coupling, the patent further explores the derivatization of these unsaturated intermediates through Corey-Chaykovsky cyclopropanation. This secondary transformation involves the reaction of the alpha,beta-unsaturated carbonyl system with a sulfur ylide, generated in situ from trimethylsulfoxonium iodide and sodium hydride. This step introduces a strained cyclopropane ring adjacent to the pyridine core, adding a new dimension of steric bulk that can influence the catalyst's chiral environment. The ability to sequentially build molecular complexity from a common intermediate underscores the modularity of this synthetic platform. For R&D directors, understanding this mechanistic flexibility is key, as it allows for the rapid iteration of catalyst structures to optimize enantioselectivity for specific substrate classes without reinventing the entire synthetic wheel.
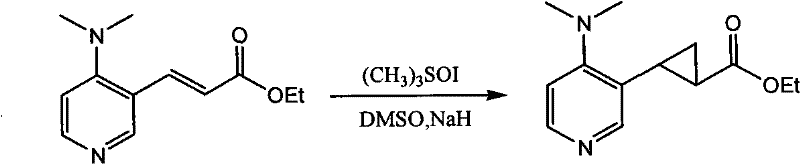
How to Synthesize 4-(N,N-Dimethyl)aminopyridine Derivatives Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing operational simplicity and reproducibility. The process begins with the precise mixing of 3-bromo-4-(N,N-dimethyl)aminopyridine, the chosen vinyl compound, and an acid binding agent in a molar ratio of approximately 1:5:10, dissolved in a polar aprotic solvent such as THF or acetonitrile. A catalytic amount of a palladium complex, such as Pd(OAc)2 paired with triphenylphosphine, is then introduced to initiate the coupling. The reaction mixture is subjected to thermal heating or microwave irradiation, driving the conversion to completion with high efficiency. Following the reaction, standard workup procedures involving aqueous extraction and drying allow for the isolation of the crude product, which can be further purified via column chromatography to achieve the desired purity specifications required for catalytic applications. Detailed standardized synthesis steps are provided in the guide below.
- Mix 3-bromo-4-(N,N-dimethyl)aminopyridine, vinyl compound, and acid binding agent (triethylamine) in a molar ratio of 1: 5:10 with a palladium catalyst in solvent.
- Heat the mixture to 80°C–160°C for 12–36 hours, or apply 750W microwave radiation for 20–40 seconds to accelerate the Heck condensation.
- Cool to room temperature, separate organic layer with methylene dichloride and water, dry over anhydrous sodium sulfate, and purify via chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic methodology offers profound advantages for organizations seeking to optimize their supply chains and reduce manufacturing overheads. The primary driver of value is the substantial simplification of the production process, which directly translates to lower operational expenditures. By replacing multi-step, cryogenic syntheses with a single-pot thermal or microwave-assisted coupling, manufacturers can significantly reduce energy consumption and equipment wear. Furthermore, the use of commodity chemicals like 3-bromo-DMAP and simple acrylates ensures a stable and predictable supply of raw materials, mitigating the risks associated with sourcing specialized organometallic precursors. This stability is critical for maintaining continuous production schedules and avoiding costly delays in the downstream synthesis of active pharmaceutical ingredients.
- Cost Reduction in Manufacturing: The elimination of expensive ferrocene scaffolds and the avoidance of stoichiometric organolithium reagents result in a drastic decrease in raw material costs. Additionally, the ability to utilize microwave heating reduces reaction times from days to seconds, dramatically increasing reactor throughput and lowering utility costs per kilogram of product. This efficiency gain allows for a more competitive pricing structure for the final chiral catalysts, enabling cost reduction in pharmaceutical intermediates manufacturing without compromising on quality.
- Enhanced Supply Chain Reliability: The reliance on robust, shelf-stable starting materials enhances the resilience of the supply chain against market fluctuations. Unlike air- and moisture-sensitive reagents that require specialized storage and handling, the inputs for this process are widely available from multiple global suppliers. This diversification of supply sources reduces dependency on single vendors and minimizes the risk of production stoppages due to raw material shortages, ensuring a consistent flow of high-purity intermediates to the production line.
- Scalability and Environmental Compliance: The transition away from hazardous organometallic reagents and cryogenic solvents simplifies waste management and improves the overall environmental footprint of the process. The milder reaction conditions facilitate easier scale-up from gram to ton quantities, as heat transfer and mixing become less challenging compared to exothermic low-temperature reactions. This scalability supports the commercial expansion of complex pharmaceutical additives while adhering to increasingly stringent environmental regulations regarding solvent usage and heavy metal discharge.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this DMAP derivative synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, aiming to clarify the practical implications for industrial adoption. Understanding these nuances is essential for stakeholders evaluating the feasibility of integrating these novel catalysts into existing process workflows.
Q: What are the primary advantages of this Heck coupling method over traditional ferrocene-based DMAP synthesis?
A: This method eliminates the need for complex multi-step organometallic sequences and cryogenic conditions (-78°C) often required for ferrocene scaffold modifications. It utilizes readily available 3-bromo-DMAP and vinyl compounds under milder thermal or rapid microwave conditions, significantly simplifying the supply chain and reducing operational complexity.
Q: Can these DMAP derivatives be used for high-selectivity kinetic resolution?
A: Yes, the synthesized chiral N,N-dimethylaminopyridine analogues have been successfully applied to the asymmetric kinetic resolution of secondary alcohols like 1-phenylethanol. While the patent reports moderate enantiomeric excess (ee) values compared to some highly specialized prior art, the ease of synthesis makes them viable candidates for process optimization where catalyst cost is a critical factor.
Q: Is microwave irradiation a viable alternative for scaling this reaction?
A: The patent data indicates that microwave promotion (750W for 20–40 seconds) yields results comparable to conventional heating (12–36 hours). For industrial scale-up, this suggests potential for continuous flow microwave reactors to drastically reduce residence time and increase throughput, although conventional thermal heating remains a robust option for large batch processing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable DMAP Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced organocatalysts play in the efficient production of modern therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are committed to delivering high-purity DMAP derivatives and related pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our expertise in process optimization allows us to adapt the Heck coupling methodology described in CN101891678A to meet your specific volume and quality requirements.
We invite you to collaborate with us to explore how these innovative synthetic routes can enhance your production efficiency. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your current catalyst needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our solutions can drive value and reliability in your supply chain.
