Advanced Manufacturing of 4,4'-Difluorobenzil: Technical Breakthroughs and Commercial Scalability
The global demand for high-performance fluorinated building blocks continues to surge, driven by their critical role in the development of next-generation pharmaceuticals and advanced functional materials. Within this landscape, the efficient synthesis of 4,4'-difluorobenzil stands out as a pivotal challenge for process chemists aiming to balance yield, safety, and cost. Patent CN102363591A introduces a robust and industrially viable methodology for preparing this valuable diketone, utilizing a two-step sequence that begins with the condensation of p-fluorobenzaldehyde. This technical disclosure represents a significant departure from traditional, hazardous routes, offering a pathway that leverages benign catalytic systems such as Vitamin B1 or specialized thiazole salts. For R&D directors and procurement strategists, understanding the nuances of this patent is essential, as it outlines a protocol that not only enhances reaction safety but also streamlines the purification process, thereby reducing the overall cost of goods sold (COGS) in fine chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzil derivatives like 4,4'-difluorobenzil has been plagued by significant operational hurdles that hinder large-scale adoption. Traditional benzoin condensation reactions often rely on highly toxic cyanide salts as catalysts, posing severe risks to operator safety and creating complex wastewater treatment challenges that inflate operational expenditures. Furthermore, conventional oxidation methods to convert the intermediate benzoin to the target benzil frequently employ stoichiometric amounts of expensive or environmentally damaging oxidants, such as nitric acid or copper salts, which can lead to over-oxidation and difficult-to-remove metallic impurities. These legacy processes often suffer from inconsistent yields and require rigorous purification steps to meet the stringent purity specifications demanded by the pharmaceutical industry. The reliance on hazardous reagents also complicates regulatory compliance and increases the lead time for obtaining necessary environmental permits, creating bottlenecks in the supply chain for high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast, the methodology detailed in CN102363591A offers a streamlined alternative that addresses these critical pain points through the strategic application of green chemistry principles. By employing Vitamin B1 (thiamine) or 3-propyl-4-methyl-5-ethoxy thiazole bromide as the catalyst for the initial condensation, the process effectively mimics the enzymatic activity of thiamine pyrophosphate, enabling the coupling of p-fluorobenzaldehyde under mild alkaline conditions without the need for toxic cyanide. This is followed by a highly efficient oxidation step using ferric chloride (FeCl3) in an aqueous ethanol medium, which provides excellent selectivity for the formation of the 1,2-diketone motif. The integration of these specific reagents results in a reaction profile that is not only safer and more environmentally benign but also demonstrates superior reproducibility and scalability. This novel approach transforms the synthesis from a hazardous laboratory curiosity into a commercially viable process suitable for the production of complex organic intermediates.
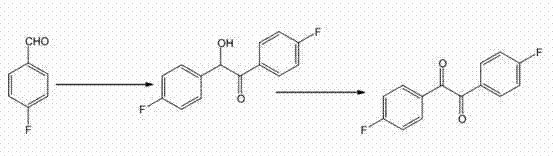
Mechanistic Insights into Vitamin B1 Catalyzed Benzoin Condensation
The core of this synthetic strategy lies in the sophisticated mechanism of Umpolung (polarity inversion) catalyzed by the thiazolium moiety present in both Vitamin B1 and the alternative thiazole bromide catalyst. In the first step, the catalyst deprotonates to form an active ylide species, which then attacks the carbonyl carbon of the p-fluorobenzaldehyde. This addition generates a Breslow intermediate, a crucial species where the natural electrophilic character of the aldehyde carbon is inverted to become nucleophilic. This nucleophilic carbon then attacks a second molecule of p-fluorobenzaldehyde, forming the carbon-carbon bond that constructs the 1,2-bis(4-fluorophenyl)-2-hydroxy ethanone skeleton. The presence of the fluorine substituent on the aromatic ring requires careful control of reaction conditions, specifically maintaining the temperature at 60°C in a saturated NaOH ethanol solution, to ensure optimal kinetics without promoting side reactions such as Cannizzaro disproportionation, which could otherwise degrade the yield of the desired hydroxy ketone intermediate.
Following the isolation of the benzoin intermediate, the subsequent oxidation to 4,4'-difluorobenzil is mediated by the redox properties of the ferric ion (Fe3+). In the presence of concentrated hydrochloric acid and heated to 70-75°C, the FeCl3 acts as a single-electron oxidant that facilitates the removal of hydrogen from the benzylic alcohol and the adjacent alpha-carbon. This mechanism proceeds through a radical or cationic pathway that efficiently converts the secondary alcohol into a ketone, completing the formation of the 1,2-dicarbonyl system. A key advantage of using FeCl3 in this context is its ability to function effectively in a mixed solvent system of water and ethanol, which simplifies the workup procedure. The resulting iron species are generally easier to separate from the organic product compared to heavy metal contaminants from other oxidation systems, thereby ensuring that the final 4,4'-difluorobenzil meets the rigorous impurity profiles required for downstream applications in luminescent materials and medicinal chemistry.
How to Synthesize 4,4'-Difluorobenzil Efficiently
To implement this synthesis effectively in a pilot or production setting, precise adherence to the reaction parameters outlined in the patent is critical for maximizing yield and purity. The process begins with the preparation of the condensation mixture, where the choice between Vitamin B1 and the thiazole derivative allows for flexibility based on raw material availability and cost considerations. Operators must carefully monitor the exotherm during the addition of p-fluorobenzaldehyde to the basic catalyst solution to prevent thermal runaway, ensuring the temperature remains stable at 60°C for the duration of the 4.5 to 7-hour reaction window. Following the crystallization and isolation of the intermediate, the oxidation step requires strict control of the acid concentration and temperature to drive the reaction to completion while minimizing the formation of oxidative byproducts. Detailed standardized operating procedures for each unit operation, from filtration to recrystallization, are essential to maintain batch-to-batch consistency.
- Condense p-fluorobenzaldehyde in saturated NaOH ethanol solution using Vitamin B1 or thiazole bromide catalyst at 60°C.
- Isolate the intermediate 1,2-bis(4-fluorophenyl)-2-hydroxy ethanone via crystallization and filtration.
- Oxidize the intermediate using FeCl3 in aqueous ethanol with HCl at 70-75°C to yield 4,4'-difluorobenzil.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the synthesis route described in CN102363591A presents a compelling value proposition centered on risk mitigation and cost optimization. The shift away from cyanide-based catalysis fundamentally alters the safety profile of the manufacturing process, eliminating the need for specialized containment infrastructure and expensive detoxification protocols associated with cyanide waste streams. This reduction in regulatory burden and safety overhead translates directly into lower operational costs and a more resilient supply chain, as the facility is less susceptible to shutdowns due to safety incidents or environmental compliance audits. Furthermore, the reliance on p-fluorobenzaldehyde as the sole carbon source simplifies the raw material inventory, leveraging a commodity chemical that is widely available from multiple global suppliers, thus reducing the risk of supply disruption.
- Cost Reduction in Manufacturing: The elimination of toxic cyanide catalysts and the use of inexpensive, abundant oxidants like ferric chloride drastically reduce the direct material costs associated with the synthesis. By avoiding the need for complex heavy metal scavenging resins or extensive purification trains required to remove trace cyanide or copper, the downstream processing costs are significantly lowered. Additionally, the high selectivity of the FeCl3 oxidation minimizes the loss of valuable starting materials to side products, improving the overall mass balance and atom economy of the process. These factors combine to create a leaner manufacturing model that delivers substantial cost savings without compromising the quality of the final API intermediate.
- Enhanced Supply Chain Reliability: Utilizing Vitamin B1 or simple thiazole salts as catalysts ensures that the critical reagents are sourced from stable, non-restricted supply chains, unlike specialized organometallic catalysts that may face geopolitical or logistical constraints. The robustness of the reaction conditions, which tolerate standard industrial-grade solvents like ethanol and water, further insulates the production schedule from fluctuations in the availability of high-purity anhydrous solvents. This operational flexibility allows manufacturers to maintain consistent production rates and meet tight delivery windows for high-purity pharmaceutical intermediates, even in volatile market conditions.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing common unit operations such as crystallization, filtration, and liquid-liquid extraction that are easily transferable from laboratory glassware to multi-ton reactor systems. The aqueous nature of the oxidation step and the use of ethanol as a primary solvent align well with modern green chemistry initiatives, facilitating easier permitting and reducing the environmental footprint of the facility. This alignment with sustainability goals not only future-proofs the manufacturing asset against tightening environmental regulations but also enhances the brand value of the end-product for eco-conscious pharmaceutical clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 4,4'-difluorobenzil, derived directly from the experimental data and beneficial effects reported in the patent literature. Understanding these specifics helps stakeholders evaluate the feasibility of integrating this technology into their existing portfolios. The answers reflect the practical realities of scaling this specific chemical transformation, focusing on yield optimization, impurity management, and operational safety.
Q: What are the primary advantages of using Vitamin B1 over cyanide catalysts in this synthesis?
A: Using Vitamin B1 eliminates the severe safety hazards and environmental toxicity associated with traditional cyanide-catalyzed benzoin condensations, significantly simplifying waste treatment and regulatory compliance.
Q: How does the FeCl3 oxidation step impact product purity?
A: The ferric chloride oxidation system operates under mild acidic conditions that minimize side reactions, allowing for the production of high-purity 4,4'-difluorobenzil with a sharp melting point range of 118-120°C.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly highlights the use of readily available raw materials and simple operational parameters, making it highly adaptable for commercial scale-up from pilot plants to multi-ton manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,4'-Difluorobenzil Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires more than just chemical knowledge; it demands deep engineering expertise and a commitment to quality excellence. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields reported in CN102363591A are realized consistently at an industrial level. We operate state-of-the-art rigorous QC labs equipped to verify stringent purity specifications, guaranteeing that every batch of 4,4'-difluorobenzil meets the exacting standards required for pharmaceutical and electronic material applications. Our capability to manage the entire lifecycle of the synthesis, from raw material sourcing to final packaging, positions us as a strategic partner for companies seeking to secure their supply of this critical intermediate.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can be tailored to your specific volume requirements and cost targets. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this greener, more efficient manufacturing process. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and innovation in your supply chain.
