Advanced Copper Complex Catalyst Enables Efficient Room Temperature Mannich Reactions for Pharmaceutical Intermediates
The landscape of industrial organic synthesis is constantly evolving, driven by the urgent need for greener, more efficient, and cost-effective catalytic systems. A significant breakthrough in this domain is documented in patent CN110357907B, which discloses a novel preparation method and application for a 4,6-bis(pyridin-2-yl)pyrimidin-2-amine copper complex. This specific coordination compound represents a paradigm shift in how Mannich reactions are approached, particularly for the synthesis of valuable pharmaceutical and agrochemical intermediates. Unlike conventional catalysts that often require harsh thermal conditions or generate substantial waste, this innovation leverages the unique redox properties and structural diversity of metallic copper salts to achieve high-performance catalysis under mild, ambient conditions. For R&D directors and process engineers, the implications are profound: a pathway to cleaner synthesis with simplified downstream processing. The patent details a robust methodology where copper chloride, the specific pyrimidine-amine ligand, and dimethyl sulfoxide react to form a stable, blocky crystal structure that serves as a highly active heterogeneous catalyst.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the Mannich reaction has been a cornerstone in the production of bioactive molecules, yet its industrial implementation has been plagued by significant inefficiencies. Traditional catalytic systems relying on weak acids, strong alkalis, or generic transition metal salts often fail to meet modern sustainability standards. These legacy methods frequently necessitate elevated temperatures to drive reaction kinetics, leading to excessive energy consumption and increased safety risks in large-scale reactors. Furthermore, a critical bottleneck in conventional processes is the inability to effectively recover and recycle the catalyst, resulting in high operational costs due to continuous fresh catalyst loading and complicated waste treatment protocols. The environmental footprint is further exacerbated by the generation of toxic byproducts and the difficulty in separating the catalyst from the final product matrix, which often requires extensive purification steps that erode overall yield and profitability.
The Novel Approach
In stark contrast, the technology described in patent CN110357907B introduces a sophisticated copper coordination complex that overcomes these historical barriers through precise molecular engineering. This novel approach utilizes a specifically designed ligand system that stabilizes the copper center, enabling the reaction to proceed efficiently at room temperature, thereby eliminating the need for energy-intensive heating. The catalyst exhibits remarkable heterogeneity, allowing for straightforward separation via simple filtration after the reaction is complete. Crucially, the system maintains its catalytic integrity over multiple cycles, addressing the recyclability issue that plagues traditional methods. The scope of this new methodology is impressive, demonstrating high conversion rates and perfect selectivity across a diverse range of substituted phenylacetylenes, as illustrated in the reaction scope below.
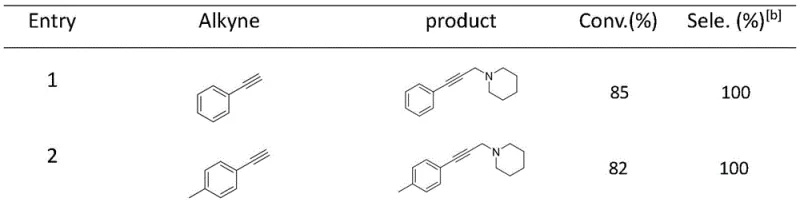
This visual data underscores the versatility of the catalyst, showing consistent performance whether the substrate contains electron-donating groups like methyl and methoxy or electron-withdrawing groups like chlorine. Such broad substrate tolerance is essential for a reliable pharmaceutical intermediate supplier aiming to streamline the production of diverse drug candidates without needing to re-optimize conditions for every new molecule.
Mechanistic Insights into Cu-Complex Catalyzed Mannich Reaction
To fully appreciate the value of this innovation for research and development teams, one must delve into the structural nuances of the catalyst itself. The complex is formed through a coordination interaction between the Cu2+ cation, chloride anions, and the nitrogen-rich 4,6-bis(pyridin-2-yl)pyrimidin-2-amine ligand, solvated by dimethyl sulfoxide molecules. The definitive molecular structure, confirmed by single-crystal X-ray diffraction, reveals a stable architecture that prevents the leaching of copper ions into the reaction medium, a common failure mode in homogeneous catalysis. This structural rigidity is key to its function; it creates a specific electronic environment around the copper center that activates the alkyne and iminium intermediates simultaneously, facilitating the three-component coupling with high precision. The presence of the pyrimidine and pyridine rings provides a rigid scaffold that likely enforces a specific geometry favorable for the transition state of the Mannich reaction.
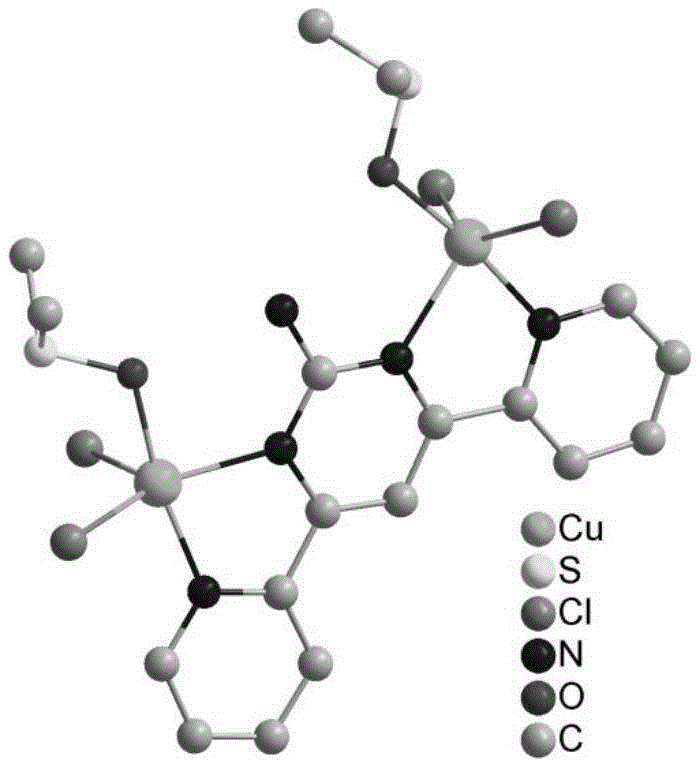
From an impurity control perspective, this mechanism offers distinct advantages. The high selectivity observed—often reaching 100% in the patent examples—suggests that the catalyst effectively suppresses side reactions such as polymerization of the alkyne or over-alkylation, which are common pitfalls in acid-catalyzed variants. By minimizing the formation of structurally similar byproducts, the burden on downstream purification is drastically reduced. For a cost reduction in pharmaceutical intermediate manufacturing strategy, this is a critical factor; fewer impurities mean less solvent usage for recrystallization or chromatography, directly translating to lower production costs and higher throughput. The ability to operate at room temperature further mitigates the risk of thermal degradation of sensitive functional groups, ensuring the integrity of complex molecular architectures often found in late-stage intermediates.
How to Synthesize 4,6-bis(pyridin-2-yl)pyrimidin-2-amine Copper Complex Efficiently
The preparation of this high-value catalyst is remarkably straightforward, designed to be accessible for both laboratory scale-up and industrial production. The process involves a simple solution reaction where the organic ligand and copper salt are mixed in DMSO at ambient temperature, followed by a period of stirring and slow evaporation to induce crystallization. This simplicity is a major asset for supply chain stability, as it does not require specialized high-pressure equipment or extreme thermal controls. The detailed standardized synthesis steps, including precise molar ratios and timing for optimal crystal growth, are outlined in the guide below to ensure reproducibility and quality consistency.
- Sequentially add the organic ligand 4,6-di(pyridine-2-yl)pyrimidine-2-amine and dimethyl sulfoxide (DMSO) into a clean beaker.
- Add CuCl2·2H2O to the mixture and stir electromagnetically at room temperature for 2.0 to 5.0 hours.
- Filter the reaction mixture, allow the filtrate to stand at room temperature, and slowly evaporate to obtain blocky crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this copper complex technology translates into tangible strategic benefits beyond mere chemical efficiency. The primary advantage lies in the drastic simplification of the manufacturing workflow. By enabling reactions at room temperature, facilities can utilize existing infrastructure without the need for expensive heating jackets or energy-intensive thermal regulation systems, leading to substantial operational expenditure savings. Furthermore, the recyclability of the catalyst means that the effective cost per kilogram of the final product is significantly lowered over time, as the initial investment in the catalyst is amortized over multiple batches rather than being a single-use expense.
- Cost Reduction in Manufacturing: The elimination of harsh reaction conditions and the ability to reuse the catalyst multiple times without significant loss of activity creates a compelling economic case. Traditional processes often incur hidden costs related to catalyst disposal and the neutralization of acidic or basic waste streams; this novel method circumvents those expenses entirely. Additionally, the high selectivity reduces the volume of raw materials wasted on byproduct formation, optimizing the atom economy of the process and ensuring that every gram of starting material contributes to the final revenue-generating product.
- Enhanced Supply Chain Reliability: The synthesis of the catalyst itself relies on readily available starting materials such as copper chloride and commercially available nitrogen heterocycles, reducing the risk of supply bottlenecks associated with exotic or proprietary reagents. The robustness of the catalyst storage and handling—being a stable solid crystal—simplifies logistics and inventory management. For a reliable pharmaceutical intermediate supplier, this stability ensures that production schedules can be maintained consistently without interruptions caused by catalyst degradation or complex storage requirements.
- Scalability and Environmental Compliance: As regulatory pressures regarding heavy metal residues and chemical waste intensify globally, this technology offers a proactive solution for compliance. The heterogeneous nature of the catalyst facilitates easy removal, minimizing the risk of copper contamination in the final API or intermediate, which is a critical quality attribute. The process generates less hazardous waste compared to traditional acid/base catalysis, aligning with green chemistry principles and reducing the environmental liability for manufacturing partners seeking to enhance their sustainability profiles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this copper complex technology. These insights are derived directly from the experimental data and structural analysis provided in the patent literature, aimed at clarifying the operational parameters and potential applications for industrial partners. Understanding these details is crucial for evaluating the feasibility of integrating this catalyst into existing production lines.
Q: What are the primary advantages of this copper complex catalyst over traditional Mannich reaction catalysts?
A: Unlike traditional weak acid, alkali, or transition metal salt catalysts that often suffer from poor recyclability and environmental pollution, this novel copper complex operates efficiently at room temperature, offers high selectivity up to 100%, and can be easily separated and reused multiple times without significant loss of activity.
Q: What is the structural composition of the synthesized catalyst?
A: The catalyst is identified as a 4,6-bis(pyridin-2-yl)pyrimidin-2-amine copper complex with the molecular formula [(CuCl)2)2(C14H11N5)(DMSO)2]·2DMSO. It features a definite molecular structure characterized by single crystal X-ray diffraction, containing Cu2+ cations and Cl- anions coordinated with the organic ligand and DMSO solvent molecules.
Q: Can this catalyst be applied to a wide range of substrates in industrial synthesis?
A: Yes, the catalyst demonstrates excellent versatility in the three-component coupling reaction of substituted phenylacetylene, formaldehyde, and piperidine. It maintains high conversion rates (over 80%) and perfect selectivity across various substrates including those with methyl, ethyl, propyl, chloro, and methoxy substituents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,6-bis(pyridin-2-yl)pyrimidin-2-amine Copper Complex Supplier
The technological potential of the 4,6-bis(pyridin-2-yl)pyrimidin-2-amine copper complex is clear, but realizing its value requires a partner with the capability to execute complex syntheses at scale. NINGBO INNO PHARMCHEM stands ready to support your development goals, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, ensuring that the catalysts and intermediates we deliver meet the exacting standards required for global pharmaceutical and agrochemical supply chains. We understand that consistency is key, and our process engineering team is dedicated to maintaining the high selectivity and conversion rates demonstrated in the patent data.
We invite you to engage with our technical procurement team to discuss how this innovative catalytic system can be tailored to your specific synthesis needs. By requesting a Customized Cost-Saving Analysis, you can quantify the potential efficiencies this room-temperature process brings to your operations. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your next project benefits from the highest standards of chemical innovation and supply chain reliability.
