Optimizing Iminostilbene Production: A Technical Breakthrough in Catalytic Dehydrogenation and Solvent Recovery
Optimizing Iminostilbene Production: A Technical Breakthrough in Catalytic Dehydrogenation and Solvent Recovery
The pharmaceutical industry constantly seeks more efficient pathways for synthesizing critical intermediates, and the recent advancements detailed in patent CN111217751B represent a significant leap forward in the production of iminostilbene. This compound serves as a pivotal building block for numerous active pharmaceutical ingredients (APIs), including carbamazepine and oxcarbazepine, necessitating a robust and scalable supply chain. The disclosed technology introduces a novel catalytic dehydrogenation process that fundamentally alters the mass balance of the reaction by integrating water vapor recycling and mother liquor reutilization. Unlike traditional methods that treat process water and solvent filtrates as waste streams requiring expensive treatment, this innovation transforms them into valuable resources, thereby aligning chemical synthesis with green chemistry principles. For R&D directors and process engineers, understanding the nuances of this water-mediated catalytic system is essential for evaluating its potential integration into existing manufacturing lines. The following analysis dissects the technical merits of this approach, highlighting its capacity to enhance yield and purity while simultaneously addressing the economic pressures faced by procurement and supply chain teams in the competitive landscape of fine chemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of iminostilbene from iminodibenzyl has relied heavily on high-temperature dehydrogenation processes that are energy-intensive and resource-inefficient. As illustrated in prior art such as Chinese patent CN101307021B, conventional techniques typically involve passing the raw material over a fixed catalyst bed at temperatures ranging from 400°C to higher extremes, often requiring substantial amounts of water vapor as a carrier gas. A critical drawback of these legacy methods is the single-pass nature of the utility streams; the vast quantities of steam injected to facilitate the reaction are subsequently condensed and discarded as wastewater, representing a significant loss of thermal energy and water resources. Furthermore, the organic solvents used to extract the product are often not optimized for recovery, leading to lower overall yields due to product loss in the mother liquor. This linear consumption model not only inflates operational expenditures through high utility bills but also imposes a heavy burden on environmental compliance teams who must manage large volumes of contaminated effluent. The inability to effectively recycle the heat and mass within the system creates a bottleneck for scaling up production without proportionally increasing the environmental footprint and cost base.
The Novel Approach
In stark contrast to these inefficient legacy systems, the methodology outlined in CN111217751B introduces a closed-loop strategy that maximizes resource utilization at every stage of the synthesis. The core innovation lies in the deliberate recovery and repurposing of the water vapor used during the catalytic dehydrogenation step. Instead of being vented or treated as waste, the steam is condensed alongside the reaction product, separated, and then directly reused as the wash water for acid and water washing of the organic layer. This circular approach drastically reduces the demand for fresh industrial water. Additionally, the process incorporates a sophisticated solvent management system where the refining mother liquor—the filtrate remaining after the initial crystallization—is not discarded. Instead, after undergoing a specific acid and water wash to remove impurities, this mother liquor is recycled back into the process as the primary organic solvent for absorbing reaction products in subsequent batches. This dual-recycling mechanism ensures that residual product trapped in the mother liquor is recovered in future cycles, thereby boosting the cumulative yield of the process while minimizing solvent purchase costs.
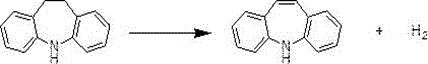
Mechanistic Insights into Catalytic Dehydrogenation and Phase Separation
The chemical transformation at the heart of this process is the catalytic dehydrogenation of iminodibenzyl, a reaction that requires precise thermal management and phase control to proceed efficiently. In this system, molten iminodibenzyl is metered into a vaporization mixer where it encounters superheated steam, creating a homogeneous gas-phase mixture that enters the catalytic bed. The presence of steam serves multiple mechanistic functions: it acts as a heat carrier to maintain the requisite reaction temperature, dilutes the reactant to prevent coking on the catalyst surface, and shifts the equilibrium towards the dehydrogenated product by Le Chatelier's principle through the continuous removal of hydrogen. The efficiency of this step is critical, as incomplete conversion leads to difficult-to-separate impurities that can compromise the purity of the final API intermediate. The subsequent quenching of this hot gas stream into an organic solvent, such as toluene or xylene, facilitates rapid condensation and absorption of the iminostilbene, preventing thermal degradation. The phase separation that follows relies on the immiscibility of the organic product layer and the condensed water layer, a physical property that is exploited to isolate the crude product before purification.
Impurity control is further refined through a rigorous two-stage crystallization protocol combined with the strategic washing of recycled streams. The organic layer, containing the crude iminostilbene, is subjected to acid washing to remove basic impurities and catalyst residues, followed by water washing to eliminate acidic traces. By utilizing the condensed process water for these washing steps, the system maintains a consistent ionic strength and temperature profile that aids in phase separation. The crystallization is performed by cooling the solution to below 10°C, a temperature threshold selected to maximize the precipitation of pure iminostilbene while keeping soluble impurities in the mother liquor. The patent emphasizes a second crystallization step for the coarse crystals, ensuring that the final lattice structure is free from occluded solvent or byproducts. This meticulous attention to thermodynamic control during the solid-liquid separation phases is what allows the process to consistently achieve purity levels exceeding 99.8%, a specification that is non-negotiable for downstream pharmaceutical synthesis.
How to Synthesize Iminostilbene Efficiently
Implementing this synthesis route requires a coordinated sequence of unit operations designed to handle high-temperature melts and sensitive catalytic reactions. The process begins with the melting of iminodibenzyl, followed by its precise injection into a steam stream to ensure uniform vaporization before contacting the catalyst. Operators must carefully monitor the temperature of the catalytic bed and the flow rates of both the melt and the steam to maintain optimal conversion rates. Following the reaction, the focus shifts to the separation train, where the efficiency of the organic solvent absorption and the subsequent layering determines the recovery rate. The crystallization steps demand strict temperature control to ensure the formation of well-defined crystals that are easy to filter and dry. For a comprehensive understanding of the specific parameters, equipment configurations, and safety protocols required to execute this synthesis at scale, please refer to the detailed technical guide provided below.
- Melt iminodibenzyl in a heating kettle and transfer the molten material to a mixing vessel.
- Mix the molten iminodibenzyl with superheated steam using a metering pump and vaporization mixer before entering the catalytic bed.
- Pass the mixture through a fixed catalytic bed for dehydrogenation reaction at elevated temperatures.
- Absorb the reaction product in an organic solvent (e.g., toluene), cool to separate layers, and recover the water layer for reuse.
- Wash the organic layer with acid and water, then perform two-stage cooling crystallization below 10°C to obtain high-purity crystals.
- Recycle the refining mother liquor by washing it with acid and water to use as the organic solvent for the next batch.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented synthesis method offers tangible benefits that extend beyond mere technical elegance, directly impacting the bottom line and supply chain resilience. For procurement managers tasked with managing the cost of goods sold (COGS) for key intermediates, the ability to recycle both water and organic solvents represents a significant opportunity for cost reduction in pharmaceutical intermediate manufacturing. By eliminating the need to purchase fresh solvent for every batch and reducing the volume of wastewater treatment, the operational expenditure associated with utilities and waste disposal is substantially lowered. Furthermore, the recovery of product from the mother liquor means that the effective yield per kilogram of starting material is increased, allowing manufacturers to produce more saleable product from the same amount of raw material input. These efficiencies translate into a more competitive pricing structure for the final iminostilbene, providing a buffer against volatility in raw material markets.
- Cost Reduction in Manufacturing: The elimination of single-use solvent streams and the recycling of process water create a leaner manufacturing model that minimizes variable costs. By reusing the refining mother liquor as a solvent, the process effectively captures value that would otherwise be lost in waste streams, leading to substantial cost savings over the lifecycle of the production campaign. Additionally, the reduced load on wastewater treatment facilities lowers the environmental compliance costs, which are increasingly becoming a major factor in the total cost of production for fine chemicals.
- Enhanced Supply Chain Reliability: Dependence on external utilities such as fresh water and bulk solvent supplies can be a vulnerability in the supply chain, particularly in regions facing resource constraints. This self-sustaining process design mitigates those risks by internalizing the supply of critical process aids. The robustness of the two-stage crystallization also ensures a consistent quality of output, reducing the likelihood of batch failures or rejections that could disrupt the supply of this critical API intermediate to downstream customers.
- Scalability and Environmental Compliance: The continuous nature of the catalytic dehydrogenation step, coupled with batch crystallization, offers a flexible platform for commercial scale-up of complex heterocyclic compounds. The process is inherently greener due to the recycling loops, making it easier to meet stringent environmental regulations regarding effluent discharge. This alignment with sustainability goals not only future-proofs the manufacturing site against tightening regulations but also enhances the brand value of the supplier in the eyes of environmentally conscious multinational pharmaceutical clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this iminostilbene synthesis technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding the specifics of solvent compatibility, water quality requirements, and yield expectations is crucial for technical teams evaluating the feasibility of adopting this route. The answers below clarify the operational boundaries and advantages of the method, helping stakeholders assess its fit within their current production capabilities.
Q: How does the new synthesis method reduce water consumption compared to traditional processes?
A: The patented process converts the large amount of water vapor used in the catalytic dehydrogenation step into liquid water during product recovery. Instead of discarding this condensate, it is reused as wash water for acid washing and water washing of the organic layer, significantly reducing fresh water intake.
Q: What is the role of refining mother liquor recycling in this synthesis?
A: The refining mother liquor, which contains dissolved product and solvent, is washed with acid and water and then reused as the organic solvent for absorbing reaction products in subsequent batches. This improves the overall yield by recovering residual iminostilbene and reduces solvent procurement costs.
Q: What purity levels can be achieved with this two-stage crystallization method?
A: Experimental data from the patent indicates that the two-stage crystallization process, involving cooling to below 10°C and filter pressing, consistently achieves purity levels exceeding 99.8% (GC), making it suitable for high-grade pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Iminostilbene Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to industrial reality requires a partner with deep technical expertise and robust infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of advanced synthesis routes like the one described in CN111217751B are fully realized in practice. We maintain stringent purity specifications through our rigorous QC labs, employing state-of-the-art analytical instrumentation to verify that every batch of iminostilbene meets the exacting standards required for pharmaceutical applications. Our commitment to quality assurance means that we do not just supply chemicals; we deliver validated solutions that integrate seamlessly into your drug development and manufacturing pipelines.
We invite you to engage with our technical procurement team to discuss how our capabilities align with your specific project needs. Whether you require a Customized Cost-Saving Analysis for your current supply chain or need to evaluate the feasibility of this specific catalytic route for your portfolio, we are ready to assist. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation and efficiency can drive value for your organization.
