Advanced Biocatalytic Synthesis of Enantiomeric Cyclopentene Derivatives for Commercial Scale-Up
Advanced Biocatalytic Synthesis of Enantiomeric Cyclopentene Derivatives for Commercial Scale-Up
The pharmaceutical industry continuously seeks more efficient and sustainable pathways for producing chiral intermediates, particularly for antiviral agents and nucleoside analogs. Patent CN1302116C discloses a groundbreaking method for preparing enantiomers of (1R, 4S)- or (1S, 4R)-1-amino-4-(hydroxymethyl)-2-cyclopentene derivatives, which serve as critical building blocks for complex therapeutic molecules. This technology represents a significant leap forward by replacing traditional chemical resolution methods with a highly selective biocatalytic process. By leveraging specific microorganisms and enzymes, manufacturers can achieve superior stereochemical control while minimizing the environmental footprint associated with heavy metal catalysts and harsh solvents. For R&D directors and procurement specialists, understanding this patented route is essential for securing a reliable supply of high-purity pharmaceutical intermediates.
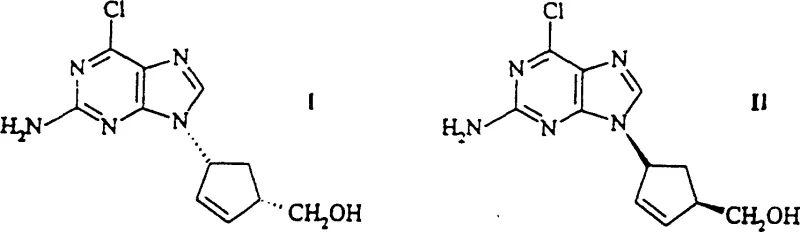
The core innovation lies in the ability to transform readily available racemic precursors into optically active compounds with exceptional fidelity. The process begins with the acylation of a bicyclic ketone, followed by reduction to form a racemic N-acyl amino alcohol. The pivotal step involves the use of microorganisms capable of utilizing these derivatives as a sole nitrogen or carbon source, effectively performing a kinetic resolution. This biological specificity allows for the isolation of the desired (1R, 4S) or (1S, 4R) isomers with enantiomeric excess values reaching up to 98%. Such high purity is paramount for downstream coupling reactions in API synthesis, ensuring that the final drug substance meets stringent regulatory specifications without the need for extensive recrystallization.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral cyclopentene derivatives relied heavily on chemical resolution techniques that were both costly and operationally complex. As noted in the background art, previous methods often required the use of valuable protecting groups, such as tert-butyloxycarbonyl (BOC), on expensive precursors like (±)-2-azabicyclo[2.2.1]heptan-5-ene-3-one. These chemical routes frequently involved multiple protection and deprotection steps, each adding to the overall production time and waste generation. Furthermore, separating enantiomers using chiral chromatography or diastereomeric salt formation often resulted in a maximum theoretical yield of 50% for the desired isomer, unless dynamic kinetic resolution was employed, which introduces additional catalyst costs. The reliance on harsh chemical reagents also posed challenges for waste treatment and operator safety, making scale-up less attractive for cost-sensitive manufacturing environments.
The Novel Approach
In contrast, the novel approach detailed in the patent utilizes a chemo-enzymatic strategy that streamlines the synthesis into fewer, more efficient steps. Instead of relying on stoichiometric chiral auxiliaries, the process employs catalytic amounts of highly specific enzymes or whole-cell microorganisms. The method starts with the acylation of the ketone precursor using simple reagents like acetyl chloride or butyryl chloride, followed by a straightforward reduction with sodium borohydride. The resulting racemic mixture is then subjected to bio-transformation, where the enzyme selectively hydrolyzes one enantiomer, leaving the other intact or converting it into a separable form. This approach not only improves the atom economy but also operates under mild aqueous conditions, typically at temperatures between 20-40°C and neutral pH. The result is a robust process that significantly reduces the consumption of organic solvents and eliminates the need for expensive chiral separation columns.
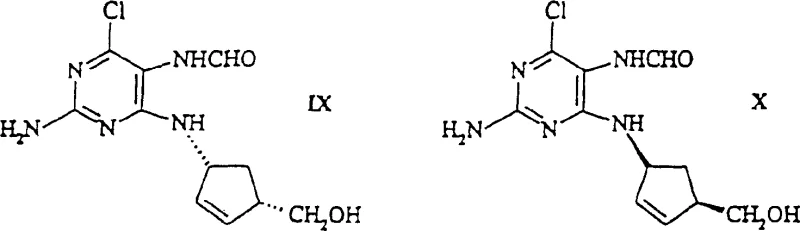
Mechanistic Insights into Enzymatic Kinetic Resolution
The heart of this technology is the enzymatic hydrolysis of N-acylaminoalcohols, a reaction mediated by N-acylaminoalcohol hydrolases or penicillin G acylases. The patent describes a diverse range of microorganisms capable of catalyzing this transformation, including strains from the genera Rhodococcus, Alcaligenes, and Gordona. Mechanistically, these enzymes recognize the specific stereochemistry of the N-acyl group attached to the cyclopentene ring. For instance, Rhodococcus CB 101 (DSM 10686) exhibits high activity towards N-acetyl and N-butyryl derivatives, selectively cleaving the amide bond of the (1R, 4S) isomer while leaving the (1S, 4R) isomer untouched, or vice versa depending on the specific enzyme variant. This selectivity is driven by the precise fit of the substrate into the enzyme's active site, where steric hindrance prevents the binding of the unwanted enantiomer. The reaction proceeds efficiently in buffered aqueous media, often requiring only minimal co-solvents to maintain substrate solubility.
Controlling impurities in this biocatalytic process is inherently easier compared to chemical synthesis due to the high specificity of the biological catalyst. Side reactions such as over-reduction or non-specific hydrolysis are minimized because the enzyme is evolved to target a specific bond. The patent data indicates that by optimizing parameters such as pH (preferably 6-8) and temperature (25-30°C), the formation of by-products can be kept to negligible levels. Furthermore, the use of resting cells or immobilized enzymes allows for the reuse of the biocatalyst, further enhancing process economics. The final products, such as (1R, 4S)-1-amino-4-(hydroxymethyl)-2-cyclopentene, are obtained with high optical purity, often exceeding 98% ee, which simplifies the downstream purification to basic extraction and crystallization steps. This level of control is critical for ensuring the quality of the final API intermediate.

How to Synthesize (1R, 4S)-1-amino-4-(hydroxymethyl)-2-cyclopentene Efficiently
The synthesis of these high-value chiral intermediates follows a logical sequence designed to maximize yield and optical purity while minimizing operational complexity. The process begins with the preparation of the racemic N-acyl precursor, which serves as the substrate for the biological resolution. Following the enzymatic step, the resulting free amine is isolated and can be further derivatized or coupled with heterocyclic bases to form nucleoside analogs. The patent provides extensive experimental data supporting the scalability of each step, from gram-scale laboratory preparations to potential multi-kilogram production runs. For process chemists looking to implement this route, the key lies in selecting the appropriate microbial strain and optimizing the fermentation conditions to induce the necessary enzymatic activity. Detailed standard operating procedures for the cultivation of strains like DSM 10686 and the subsequent bio-transformation are essential for reproducible results.
- Acylation of (±)-2-azabicyclo[2.2.1]heptan-5-ene-3-one using carbonyl halides or anhydrides to form N-acyl derivatives.
- Reduction of the N-acyl ketone derivatives using alkali metal borohydrides to generate racemic N-acyl amino alcohols.
- Enzymatic hydrolysis using specific microorganisms or penicillin G acylase to resolve the racemate into optically active (1R, 4S) or (1S, 4R) isomers.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this biocatalytic pathway offers substantial strategic benefits beyond mere technical feasibility. The shift from chemical to enzymatic resolution fundamentally alters the cost structure of manufacturing these intermediates. By eliminating the need for expensive chiral resolving agents and reducing the number of synthetic steps, the overall cost of goods sold (COGS) is significantly lowered. Additionally, the use of robust microbial strains ensures a consistent and reliable supply of the catalyst, mitigating the risks associated with the availability of precious metal catalysts or specialized chemical reagents. This reliability is crucial for maintaining continuous production schedules and meeting the demanding delivery timelines of global pharmaceutical clients.
- Cost Reduction in Manufacturing: The primary economic driver of this technology is the drastic simplification of the purification process. Traditional chemical resolutions often suffer from low yields due to the 50% theoretical limit of separating racemates, whereas enzymatic kinetic resolution can be tuned to favor the desired isomer with high efficiency. Moreover, the reaction conditions are mild, typically requiring ambient pressure and moderate temperatures, which translates to lower energy consumption and reduced wear on processing equipment. The elimination of toxic solvents and heavy metal catalysts also reduces waste disposal costs, contributing to a leaner and more sustainable manufacturing budget.
- Enhanced Supply Chain Reliability: Sourcing high-purity chiral intermediates can often be a bottleneck in the supply chain, especially when relying on single-source suppliers of specialized reagents. This patented process utilizes widely available starting materials and biocatalysts that can be produced in-house or sourced from multiple vendors. The robustness of the microbial strains, such as those deposited under the Budapest Treaty, ensures that the biological component of the supply chain is secure. This diversification of supply sources reduces the risk of disruption and allows for greater flexibility in responding to fluctuations in market demand.
- Scalability and Environmental Compliance: Scaling up biocatalytic processes is generally more straightforward than scaling complex chemical syntheses involving hazardous reagents. The aqueous nature of the bio-transformation step simplifies reactor design and heat management, facilitating the transition from pilot plant to commercial production. Furthermore, the environmental profile of this process aligns well with increasingly strict global regulations on chemical emissions and waste. By generating less hazardous waste and consuming fewer resources, manufacturers can achieve compliance with environmental standards more easily, avoiding potential fines and reputational damage associated with non-compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this biocatalytic synthesis route. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the technology's capabilities and limitations. Understanding these details is vital for stakeholders evaluating the feasibility of integrating this process into their existing manufacturing portfolios.
Q: What are the primary advantages of this biocatalytic method over conventional chemical resolution?
A: The patented biocatalytic method eliminates the need for expensive chiral protecting groups like BOC on early precursors and avoids complex chromatographic separations. By utilizing specific microbial strains such as Rhodococcus CB 101 or enzymes like penicillin G acylase, the process achieves high enantiomeric excess (up to 98% ee) directly from racemic substrates, significantly simplifying the purification workflow and reducing raw material costs.
Q: Which specific microorganisms are identified as effective catalysts in this patent?
A: The patent explicitly identifies several robust microbial strains capable of acting as sole nitrogen or carbon sources for the bio-transformation. Key strains include Alcaligenes faecalis FB 188 (DSM 11172), Rhodococcus erythropolis CB 101 (DSM 10686), and Gordona sp. CB 100 (DSM 10687). These organisms exhibit high specificity for N-acyl amino alcohol substrates, enabling efficient kinetic resolution under mild aqueous conditions.
Q: What purity levels and yields can be expected from this synthesis route?
A: Experimental data within the patent demonstrates that the final optically active compounds, such as (1R, 4S)-1-amino-4-(hydroxymethyl)-2-cyclopentene hydrochloride, can be achieved with an enantiomeric excess of 98%. Isolated yields for key intermediates vary by step, with acylation steps reaching up to 85% and subsequent reduction steps achieving around 64-80%, ensuring a commercially viable overall throughput for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (1R, 4S)-1-amino-4-(hydroxymethyl)-2-cyclopentene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of next-generation therapeutics. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and efficiency. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether you require custom synthesis of complex chiral building blocks or large-scale supply of established intermediates, our infrastructure is designed to support your growth from clinical trials to market launch.
We invite you to collaborate with us to explore how this advanced biocatalytic technology can optimize your supply chain and reduce manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can become a cornerstone of your successful drug development program.
