Stereoselective Manufacturing of Phencyclonate Optical Isomers for Advanced Pharmaceutical Applications
The pharmaceutical landscape for anticholinergic agents is increasingly demanding higher purity standards, particularly for active isomers that exhibit superior therapeutic efficacy. Patent CN100420674C introduces a groundbreaking stereoselective synthesis method for preparing optical isomers of 2′-phenyl-2′-cyclopentyl-2′-hydroxyacetic acid-9α-[N-methyl-3-azabicyclo(3,3,1)nonyl]ester, commonly known as Phencyclonate. This technology represents a significant leap forward from traditional racemic synthesis, utilizing optically pure R or S mandelic acid as a chiral template to directly construct the complex molecular architecture. By leveraging this chiral pool strategy, the process achieves an optical purity of greater than 98%, addressing the critical need for high-quality pharmaceutical intermediates in the treatment of motion sickness and Parkinson's disease. The innovation lies not just in the final product quality, but in the elegant efficiency of the pathway, which minimizes waste and maximizes stereochemical integrity throughout the multi-step sequence.
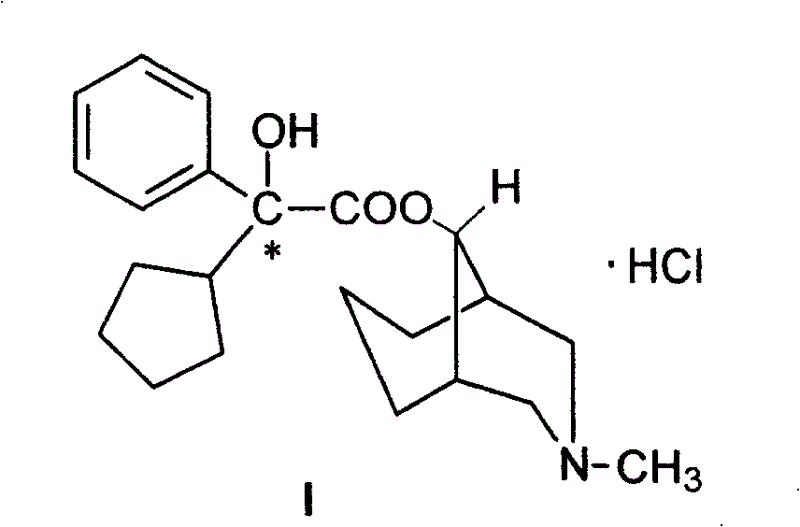
Furthermore, the clinical implications of accessing single enantiomers cannot be overstated, as preliminary studies indicate that the R-isomer possesses significantly higher affinity for brain M receptors compared to its S-counterpart. This patent provides the robust chemical foundation required to isolate and produce these high-value isomers reliably. For R&D directors and procurement specialists, understanding this pathway is essential for securing a supply chain that can meet the rigorous specifications of modern regulatory bodies. The method described offers a clear route to reducing impurity profiles associated with racemic mixtures, thereby simplifying downstream purification and enhancing the overall safety profile of the final drug substance. As we delve deeper into the technical specifics, the strategic advantages of this chiral template approach become evident for any organization aiming to lead in the production of specialized neurological therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of Phencyclonate and similar anticholinergic agents relied heavily on the synthesis of racemic mixtures followed by resolution, a process fraught with inherent inefficiencies and economic drawbacks. Traditional methods often involve reacting non-optically active raw materials with Grignard reagents, resulting in a 50:50 mixture of enantiomers that requires costly and yield-limiting separation techniques. This resolution step typically discards half of the synthesized material, effectively doubling the raw material consumption and waste generation for the desired active isomer. Moreover, the use of Grignard reagents introduces safety hazards and sensitivity to moisture, complicating the operational parameters for large-scale manufacturing. The resulting impurity profiles from racemic synthesis are often complex, requiring extensive chromatographic purification which further erodes profit margins and extends production lead times. For supply chain managers, these factors translate into higher costs of goods sold and increased vulnerability to raw material price fluctuations, making the conventional route less sustainable in a competitive market environment.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a chiral pool strategy starting from commercially available and inexpensive optically pure mandelic acid. This method bypasses the need for resolution entirely by embedding the chirality at the very beginning of the synthesis through the formation of a protected lactone intermediate. The process employs mild reaction conditions, such as acid-catalyzed condensation with pivalaldehyde at moderate temperatures, which are far more manageable than the cryogenic conditions often required for asymmetric catalysis. By locking the stereochemistry early in the sequence, the subsequent steps, including the critical Michael addition, proceed with high diastereoselectivity, preserving the optical integrity of the molecule. This direct asymmetric synthesis not only improves the overall atom economy but also significantly simplifies the purification workflow, as the byproducts are chemically distinct rather than enantiomeric. For manufacturers, this translates to a streamlined process that is easier to validate and control, ensuring consistent batch-to-batch quality while drastically reducing the environmental footprint associated with discarded isomers.
Mechanistic Insights into Stereoselective Lactone Formation and Michael Addition
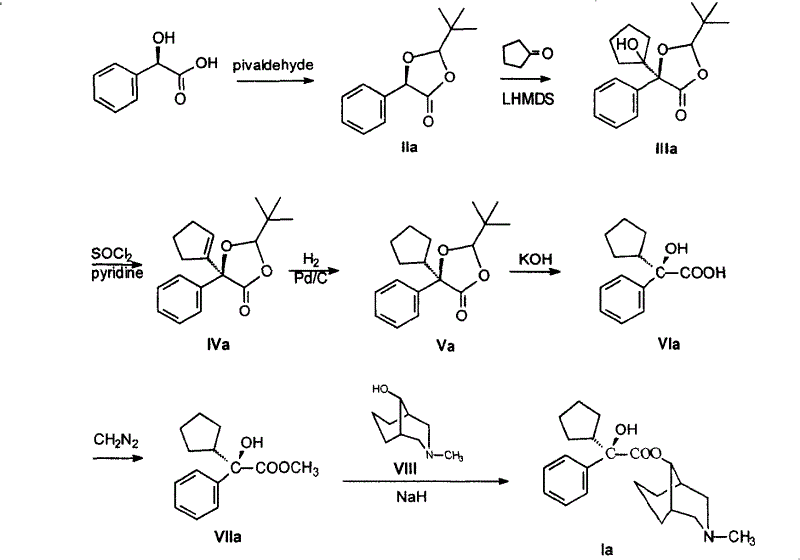
The core of this synthetic breakthrough lies in the ingenious protection and activation of the chiral center derived from mandelic acid. Initially, the hydroxyl group of the mandelic acid undergoes an aldol condensation with pivalaldehyde, followed by intramolecular cyclization to form a rigid 2-tert-butyl-5-phenyl-[1,3]dioxolan-4-one lactone structure. This cyclic acetal serves a dual purpose: it protects the sensitive hydroxyl and carboxyl functionalities while imposing a steric environment that directs the facial selectivity of subsequent nucleophilic attacks. The bulky tert-butyl group acts as a chiral auxiliary, shielding one face of the molecule and forcing incoming reagents to approach from the less hindered side. This mechanistic feature is crucial for maintaining high optical purity, as it prevents racemization during the harsh conditions of subsequent transformations. The stability of this lactone intermediate allows for isolation and characterization, providing a critical quality control checkpoint before proceeding to the more complex bond-forming steps.
Following the formation of the chiral lactone, the synthesis proceeds via a stereocontrolled Michael addition with cyclopentanone, a step that constructs the quaternary carbon center essential for the drug's activity. In the presence of strong lithium bases such as LDA or lithium bis(trimethylsilyl)amide at temperatures ranging from -80 to -60°C, the lactone is converted into its enol form. This enolate then attacks the cyclopentanone with high precision, guided by the existing chiral information in the dioxolanone ring. The resulting addition product retains the stereochemical configuration established in the first step, demonstrating the robustness of the chiral template. Subsequent dehydration, hydrogenation, and deprotection steps reveal the free acid functionality while preserving the newly formed stereocenters. This sequence exemplifies a classic yet highly effective application of substrate-controlled asymmetry, where the molecular architecture itself dictates the outcome of the reaction, minimizing the need for expensive external chiral catalysts.
How to Synthesize Phencyclonate Optical Isomers Efficiently
The synthesis of these high-value optical isomers requires precise control over reaction parameters to ensure the preservation of chirality throughout the multi-step sequence. The process begins with the condensation of mandelic acid and pivalaldehyde, followed by a series of transformations including Michael addition, dehydration, and hydrogenation to generate the key hydroxy acid intermediate. Detailed standard operating procedures for each step, including specific solvent choices like n-pentane or tetrahydrofuran and precise temperature controls, are critical for reproducibility. The final coupling with 9α-[N-methyl-3-azabicyclo(3,3,1)nonyl] alcohol completes the assembly of the target molecule.
- Condense optically pure R or S mandelic acid with pivalaldehyde to form a chiral lactone intermediate.
- Perform a stereocontrolled Michael addition with cyclopentanone using lithium reagents to introduce the cyclopentyl group.
- Execute dehydration, hydrogenation, and deprotection to yield the key hydroxy acid intermediate, followed by esterification and coupling.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this stereoselective synthesis route offers substantial strategic benefits that extend beyond mere technical superiority. The reliance on mandelic acid, a commodity chemical produced on a massive scale for various industries, ensures a stable and cost-effective supply of the primary chiral source. Unlike processes dependent on rare earth metals or proprietary chiral ligands, this method utilizes common organic reagents and bases, mitigating the risk of supply chain disruptions caused by geopolitical factors or mining constraints. The elimination of the resolution step inherently reduces the volume of raw materials required per kilogram of active product, leading to significant cost reductions in manufacturing without compromising on quality. Furthermore, the simplified purification profile reduces the consumption of solvents and chromatography media, contributing to lower operational expenditures and a greener manufacturing footprint. These factors combined create a resilient supply chain capable of meeting high-volume demands with consistent reliability.
- Cost Reduction in Manufacturing: The elimination of the racemic resolution step effectively doubles the theoretical yield of the desired isomer compared to traditional methods, as no material is discarded as the unwanted enantiomer. This improvement in atom economy directly translates to lower raw material costs and reduced waste disposal fees, which are significant components of the total cost of ownership for pharmaceutical intermediates. Additionally, the use of standard reagents like pivalaldehyde and cyclopentanone avoids the premium pricing associated with specialized chiral catalysts, further driving down the cost basis. The streamlined workflow also reduces labor hours and equipment occupancy time, allowing for higher throughput in existing facilities. Consequently, manufacturers can achieve a more competitive pricing structure while maintaining healthy margins, a critical advantage in the generic and specialty pharmaceutical markets.
- Enhanced Supply Chain Reliability: By anchoring the synthesis on widely available starting materials, the process minimizes dependency on single-source suppliers for exotic reagents. Mandelic acid and cyclopentanone are produced by multiple global vendors, ensuring that procurement teams can negotiate favorable terms and maintain backup sources to prevent production stoppages. The robustness of the reaction conditions, which do not require extreme cryogenic temperatures beyond standard industrial cooling capabilities, further enhances operational reliability. This stability allows for better production planning and inventory management, reducing the need for excessive safety stocks. For long-term contracts, this reliability is paramount, as it guarantees the continuity of supply for critical medications treating motion sickness and neurological disorders, thereby strengthening the partnership between suppliers and pharmaceutical companies.
- Scalability and Environmental Compliance: The synthetic route is designed with scalability in mind, utilizing unit operations such as distillation, crystallization, and filtration that are easily transferred from pilot plant to commercial scale. The avoidance of heavy metal catalysts simplifies the regulatory burden regarding residual metal limits in the final API, facilitating faster approval processes and reducing the need for complex scavenging steps. Moreover, the reduced solvent usage and higher overall yield contribute to a lower E-factor (environmental factor), aligning with modern green chemistry principles and corporate sustainability goals. This environmental compliance is increasingly becoming a deciding factor for procurement decisions, as pharmaceutical companies strive to reduce their carbon footprint. The process thus offers a future-proof solution that meets both economic and ecological criteria for modern chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Phencyclonate optical isomers, derived directly from the patented methodology. Understanding these details helps stakeholders evaluate the feasibility and benefits of integrating this technology into their supply chains. The answers reflect the specific advantages of the chiral template approach over conventional racemic synthesis.
Q: What is the primary advantage of this synthesis route over traditional racemic resolution?
A: The primary advantage is the direct utilization of optically pure mandelic acid as a chiral template, which eliminates the need for inefficient resolution steps and ensures optical purity exceeding 98% from the outset.
Q: Which specific reagents are critical for maintaining stereochemistry during the Michael addition?
A: Lithium reagents such as LDA or lithium bis(trimethylsilyl)amide are critical, as they facilitate the formation of the enol form of the lactone intermediate under low temperatures (-80 to -60°C) for precise stereocontrol.
Q: Is this process suitable for large-scale commercial production of anticholinergic intermediates?
A: Yes, the process utilizes readily available raw materials like mandelic acid and cyclopentanone, and avoids complex transition metal catalysts, making it highly amenable to scale-up for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phencyclonate Optical Isomer Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity intermediates in the development of next-generation anticholinergic therapies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated stereoselective chemistry described in CN100420674C can be successfully translated from the laboratory to the manufacturing floor. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced chiral HPLC and NMR capabilities to verify the optical purity of every batch. Our commitment to quality assurance means that we can consistently deliver Phencyclonate isomers with optical purity exceeding 98%, meeting the exacting standards required for clinical and commercial applications. By partnering with us, you gain access to a supply chain that is not only reliable but also technically proficient in handling complex chiral molecules.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your specific project requirements. We are prepared to provide a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this stereoselective method for your production needs. Please contact us to request specific COA data and route feasibility assessments tailored to your volume requirements. Our goal is to be more than just a vendor; we aim to be a strategic partner in your success, offering the technical expertise and manufacturing capacity needed to bring life-saving medications to market faster and more efficiently.
