Scalable Synthesis of Chiral Azepine Intermediates for High-Value Kinase Inhibitors
Scalable Synthesis of Chiral Azepine Intermediates for High-Value Kinase Inhibitors
The pharmaceutical industry's relentless pursuit of potent protein kinase C (PKC) inhibitors has placed a premium on the efficient synthesis of complex heterocyclic scaffolds. Patent CN1491951A discloses a groundbreaking methodology for the preparation of azepine intermediates, specifically targeting the core structure of Balanol and related bioactive molecules. This intellectual property represents a significant leap forward in process chemistry, offering two distinct yet complementary pathways: a highly stereoselective transition metal-catalyzed asymmetric hydrogenation and a specialized microbial reduction route. For R&D directors and procurement strategists alike, understanding the nuances of this patent is critical, as it provides a roadmap for producing high-purity pharmaceutical intermediates with superior cost-efficiency and supply chain reliability compared to legacy resolution methods.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral azepine derivatives relied heavily on the resolution of racemic mixtures, a process inherently plagued by a maximum theoretical yield of 50%. Traditional approaches often involved cumbersome chromatographic separations or multiple recrystallization steps to achieve acceptable enantiomeric purity, leading to substantial material loss and increased waste generation. Furthermore, the construction of the seven-membered azepine ring with precise stereochemical control at the 3 and 4 positions posed significant synthetic challenges, often requiring harsh conditions that compromised functional group tolerance. These inefficiencies not only inflated the cost of goods sold (COGS) but also introduced variability in supply continuity, making it difficult for manufacturers to meet the rigorous demands of global drug development pipelines.
The Novel Approach
In stark contrast, the methodology outlined in CN1491951A introduces a streamlined strategy that bypasses the limitations of racemic resolution through direct asymmetric synthesis. The patent details a robust catalytic system utilizing optically active Group VIII metal complexes, particularly rhodium and ruthenium coordinated with atropisomeric diphosphine ligands such as MeOBIPHEP and BINAP. This approach allows for the direct conversion of prochiral keto-esters into chiral hydroxy-esters with exceptional stereocontrol. Additionally, the inclusion of a biocatalytic alternative using Hanseniaspora uvarum offers a green chemistry perspective, leveraging enzymatic specificity to achieve similar outcomes under mild conditions. This dual-pathway flexibility empowers manufacturers to select the most economically viable route based on available infrastructure and scale requirements.
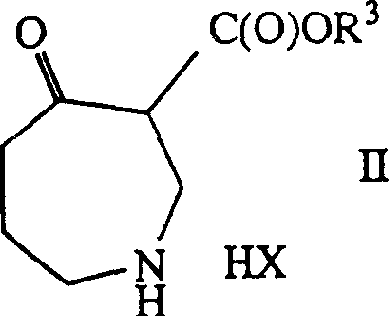
Mechanistic Insights into Rh-Catalyzed Asymmetric Hydrogenation
The core of the chemical innovation lies in the precise orchestration of the catalytic cycle during the hydrogenation of the 4-oxo-azepine-3-carboxylate substrate. The patent specifies the use of cationic rhodium or ruthenium complexes, such as [Ru(OAc)2((R)-MeOBIPHEP)], which facilitate the transfer of hydrogen to the ketone functionality with high facial selectivity. The mechanism involves the coordination of the substrate to the metal center, followed by oxidative addition of hydrogen and migratory insertion, ultimately yielding the (3R, 4R) configured product with enantiomeric excess values frequently exceeding 99%. This level of precision is achieved by tuning the steric and electronic properties of the ligand, where substituents like methoxy groups on the biphenyl backbone play a crucial role in defining the chiral pocket around the metal center.
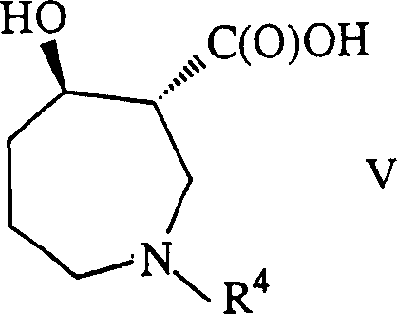
Beyond the primary reduction step, the patent elucidates a sophisticated sequence of protective group manipulations designed to preserve and verify stereochemical integrity throughout the synthesis. Following hydrogenation, the resulting amine is protected, typically with a tert-butoxycarbonyl (Boc) group, and the ester is saponified to the corresponding carboxylic acid. This acid is then subjected to a Curtius rearrangement or cyclization to form an oxazolidinone intermediate (Formula VI), effectively locking the relative stereochemistry of the adjacent chiral centers. This strategic cyclization not only prevents epimerization during subsequent processing but also serves as a robust handle for further functionalization, ensuring that the final API intermediate meets the stringent purity specifications required for clinical applications.
How to Synthesize Chiral Azepine Derivatives Efficiently
The synthesis of these high-value intermediates requires a meticulous adherence to reaction parameters to maximize yield and optical purity. The process begins with the preparation of the keto-ester substrate, followed by the critical asymmetric hydrogenation step performed under controlled pressure and temperature. Subsequent steps involve careful pH management during saponification and precise thermal control during the cyclization to the oxazolidinone. For a comprehensive breakdown of the operational parameters, reagent stoichiometry, and workup procedures necessary to replicate these results in a pilot plant setting, please refer to the standardized protocol below.
- Prepare the substrate by synthesizing 4-oxo-azepine-3-carboxylate salts (Formula II/III) via ring expansion of piperidine derivatives.
- Perform asymmetric hydrogenation using a Rhodium or Ruthenium catalyst complexed with chiral diphosphine ligands like (R)-MeOBIPHEP under 40-100 bar hydrogen pressure.
- Isolate the chiral hydroxy-ester intermediate (Formula IV), protect the amine, saponify the ester, and cyclize to form the oxazolidinone (Formula VI) to lock stereochemistry.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of the technologies described in CN1491951A offers transformative benefits for supply chain optimization and cost management. By shifting from resolution-based processes to direct asymmetric synthesis, manufacturers can theoretically double their output from the same amount of starting material, fundamentally altering the economics of production. This efficiency gain translates directly into reduced raw material consumption and lower waste disposal costs, aligning with modern sustainability goals while enhancing profit margins. Furthermore, the robustness of the catalytic systems described ensures consistent batch-to-batch quality, reducing the risk of production delays caused by out-of-specification results.
- Cost Reduction in Manufacturing: The elimination of resolution steps removes the inherent 50% yield ceiling associated with separating racemates, thereby drastically reducing the cost per kilogram of the final intermediate. Additionally, the use of recyclable catalyst systems and the avoidance of expensive chiral resolving agents contribute to significant operational expenditure savings. The streamlined process flow reduces the number of unit operations required, lowering energy consumption and labor costs associated with extended processing times and complex purification sequences.
- Enhanced Supply Chain Reliability: The versatility of the disclosed methods, offering both chemical and biological routes, provides a strategic buffer against supply chain disruptions. If catalyst availability becomes constrained, the microbial route using commercially available yeast strains offers a viable backup, ensuring continuous production capability. This redundancy is crucial for maintaining uninterrupted supply to downstream API manufacturers, mitigating the risks associated with single-source dependencies and volatile raw material markets.
- Scalability and Environmental Compliance: The reactions described operate under conditions that are readily scalable from laboratory to industrial volumes, utilizing standard high-pressure hydrogenation equipment found in most multipurpose chemical plants. The microbial route, in particular, operates under mild aqueous conditions, significantly reducing the reliance on hazardous organic solvents and minimizing the environmental footprint. This alignment with green chemistry principles facilitates easier regulatory approval and reduces the burden of environmental compliance, making the process more attractive for long-term commercial investment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on the feasibility and advantages of the described synthetic routes for potential licensees and manufacturing partners.
Q: What is the primary advantage of the catalytic route over traditional resolution?
A: The patented asymmetric hydrogenation route achieves enantiomeric excess (ee) values greater than 99% directly, eliminating the need for multiple recrystallizations and significantly improving overall yield compared to racemic resolution.
Q: Can this process be scaled for commercial production?
A: Yes, the process utilizes standard high-pressure hydrogenation equipment and robust catalysts like Rh-MeOBIPHEP complexes, which are well-suited for kilogram-to-ton scale manufacturing in GMP facilities.
Q: What are the key intermediates generated in this pathway?
A: Key intermediates include the chiral 4-hydroxy-azepine-3-carboxylate (Formula IV), the oxazolidinone derivative (Formula VI), and the final amino-alcohol scaffold (Formula VII), which are critical for Balanol synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Azepine Intermediates Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated chemistry described in CN1491951A can be translated into reliable manufacturing reality. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced chiral HPLC and NMR capabilities to guarantee the stereochemical integrity of every batch, providing our partners with the confidence they need to advance their drug candidates.
We invite you to collaborate with us to leverage these advanced synthetic technologies for your specific project needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our expertise in azepine chemistry can accelerate your timeline and optimize your budget.
