Scalable Synthesis of Novel Benzothiadiazoloimidazole Fluorescent Dyes for Advanced Optoelectronic Applications
Scalable Synthesis of Novel Benzothiadiazoloimidazole Fluorescent Dyes for Advanced Optoelectronic Applications
The rapid evolution of the optoelectronic industry demands materials that offer superior photostability, tunable emission profiles, and enhanced solubility characteristics. Addressing these critical needs, the technical disclosure found in patent CN113999254B introduces a groundbreaking class of benzothiadiazoloimidazole fluorescent dyes. Unlike traditional benzothiadiazole derivatives which often struggle with aggregation-caused quenching and poor aqueous solubility, this novel molecular architecture integrates an imidazole ring directly into the benzothiadiazole core. This structural modification creates a potent intramolecular donor-acceptor push-pull system that significantly boosts fluorescence intensity while mitigating the stacking issues common in planar aromatic systems. For R&D directors and procurement specialists seeking a reliable OLED material supplier, this technology represents a significant leap forward in designing high-performance emitters for next-generation displays and biological imaging probes.
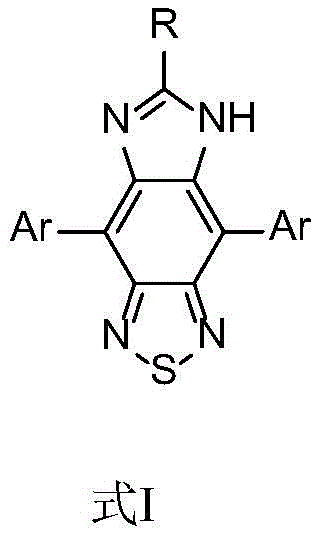
The general structure, designated as Formula I in the patent documentation, highlights the versatility of this chemical platform. By varying the substituted aryl groups (Ar) at the 4 and 7 positions and modifying the alkyl group (R) on the imidazole nitrogen, manufacturers can precisely engineer the electronic properties of the dye. This tunability allows for the adjustment of emission colors spanning from green to red, catering to the specific spectral requirements of full-color display technologies. Furthermore, the retention of an active hydrogen on the imidazole ring provides a functional handle for further derivatization, opening avenues for conjugation with biomolecules in sensing applications. The synthesis of such complex heterocyclic systems requires a robust and reproducible pathway, which the patent elucidates through a logical four-step sequence designed for high purity and yield.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of small-molecule organic fluorescent materials has been hindered by inherent structural defects that limit their practical utility in commercial devices. Conventional dyes such as cyanines, while bright, often exhibit poor photostability, leading to rapid degradation under operational conditions in OLED panels or during prolonged biological imaging sessions. Similarly, BODIPY dyes, although popular, frequently suffer from small Stokes shifts, which complicates the separation of excitation and emission signals and reduces detection sensitivity in probe applications. Most critically for aqueous applications, traditional benzothiadiazole-based acceptors possess large planar structures that facilitate strong pi-pi stacking interactions. This tendency to aggregate in polar solvents results in severe fluorescence quenching and precipitation, rendering them ineffective for water-based processing or bio-imaging without extensive and costly solubilizing modifications.
The Novel Approach
The synthetic strategy outlined in the patent overcomes these legacy challenges by fundamentally altering the electronic landscape of the fluorophore. By fusing an imidazole ring onto the benzothiadiazole scaffold, the new method introduces a strong electron-donating amino functionality that balances the electron-withdrawing nature of the thiadiazole moiety. This donor-acceptor architecture not only enhances the intramolecular charge transfer (ICT) efficiency but also disrupts the perfect planarity that leads to aggregation. The result is a dye with a remarkably large Stokes shift, reaching up to 140nm in certain derivatives, which drastically minimizes self-absorption and improves signal-to-noise ratios. Moreover, the ability to introduce bulky alkyl groups (such as tert-butyl) and diverse aryl substituents during the synthesis ensures excellent solubility in both organic and aqueous media, solving the processing bottlenecks that have plagued previous generations of benzothiadiazole dyes.
Mechanistic Insights into the Multi-Step Synthetic Route
The construction of the benzothiadiazoloimidazole core relies on a sophisticated yet industrially viable cascade of transformations, beginning with a palladium-catalyzed cross-coupling reaction. The process initiates with the Suzuki coupling of 4,7-dibromo-5,6-dinitrobenzothiadiazole with various arylboronic acids. This step is crucial for installing the electron-donating aryl wings that define the final emission color. Following this, a chemoselective reduction is performed using iron powder in acetic acid. This choice of reductant is particularly strategic for cost reduction in electronic chemical manufacturing, as iron is significantly cheaper and less toxic than alternative catalytic hydrogenation methods or hydride reagents, while effectively converting the nitro groups to the requisite amines without affecting the sensitive benzothiadiazole ring.
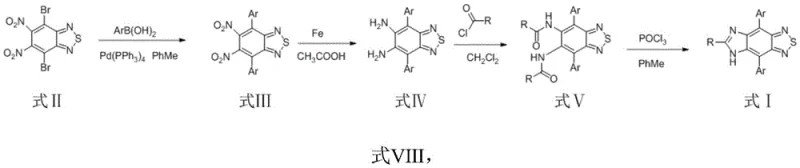
Subsequent acylation with acid chlorides installs the precursors for the imidazole ring, setting the stage for the final cyclization. The closing of the imidazole ring is achieved using phosphorus oxychloride (POCl3), a standard dehydrating agent that facilitates the condensation of the bis-amide intermediate. This cyclization step is the key to locking in the rigid, planar conjugated system responsible for the dye's high quantum yield. The entire pathway is designed to minimize side reactions; for instance, the use of mild temperatures (0-40°C) during the acylation and cyclization steps prevents the decomposition of the thermally sensitive intermediates. This careful control of reaction parameters ensures a clean impurity profile, which is essential for achieving the high purity standards required in semiconductor and display manufacturing.
How to Synthesize Benzothiadiazoloimidazole Efficiently
Executing this synthesis requires precise control over stoichiometry and temperature to maximize yield and minimize the formation of byproducts. The protocol begins with the activation of the dibromo precursor followed by the sequential addition of reagents under inert atmosphere to prevent oxidation of the sensitive amine intermediates. While the general chemistry is straightforward, the workup procedures, particularly the extraction and column chromatography steps described in the examples, are vital for isolating the high-purity material needed for optical testing. For detailed operational parameters, including specific molar ratios, solvent volumes, and purification techniques, please refer to the standardized guide below.
- Perform Suzuki coupling of 4,7-dibromo-5,6-dinitrobenzothiadiazole with arylboronic acid using Pd(PPh3)4 catalyst in toluene at 80-120°C.
- Reduce the resulting dinitro compound to the diamine derivative using iron powder in acetic acid at 60-120°C.
- React the diamine intermediate with acid chloride in the presence of triethylamine at 0-40°C to form the bis-amide precursor.
- Execute ring closure using phosphorus oxychloride (POCl3) in dichloromethane or toluene at 0-40°C to yield the final benzothiadiazoloimidazole core.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this synthetic route offers substantial logistical and economic benefits compared to alternative fluorophore syntheses. The reliance on commodity chemicals such as arylboronic acids, iron powder, and acid chlorides means that raw material sourcing is not constrained by single-source suppliers or geopolitical volatility. This abundance of starting materials ensures a stable supply chain, reducing the risk of production delays that can occur with exotic or proprietary reagents. Furthermore, the reaction conditions are compatible with standard stainless steel reactors commonly found in fine chemical manufacturing facilities, eliminating the need for specialized high-pressure or cryogenic equipment.
- Cost Reduction in Manufacturing: The synthetic pathway is engineered to utilize cost-effective reagents without compromising on performance. Specifically, the substitution of expensive noble metal catalysts or complex hydride reducing agents with iron powder for the reduction step represents a significant saving in raw material costs. Additionally, the high yields reported in the patent examples (ranging from 75% to 96% for individual steps) imply a lower consumption of solvents and energy per kilogram of final product. This efficiency translates directly into a lower cost of goods sold (COGS), allowing for more competitive pricing in the high-volume OLED material market.
- Enhanced Supply Chain Reliability: The robustness of the chemistry contributes to a more predictable production schedule. The tolerance of the Suzuki coupling and subsequent steps to minor variations in conditions suggests that the process is scalable with minimal risk of batch failure. For procurement managers, this reliability means shorter lead times and the ability to secure long-term contracts with confidence. The use of common solvents like toluene, dichloromethane, and acetic acid further simplifies logistics, as these are widely available and easy to recycle, supporting a circular economy approach within the manufacturing plant.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the absence of extremely hazardous reagents or extreme conditions. The waste streams generated, primarily consisting of iron salts and aqueous acidic washes, are well-understood and can be treated using standard effluent treatment protocols. This ease of waste management reduces the environmental compliance burden and associated disposal costs. Moreover, the high atom economy of the cyclization step ensures that a maximum proportion of the input mass ends up in the final product, aligning with green chemistry principles and corporate sustainability goals.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this new dye class is essential for stakeholders evaluating its integration into existing product lines. The following questions address common inquiries regarding the stability, customization, and application potential of these benzothiadiazoloimidazole derivatives. The answers are derived directly from the experimental data and structural analysis provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: How does the benzothiadiazoloimidazole structure improve upon traditional benzothiadiazole dyes?
A: Traditional benzothiadiazole dyes often suffer from poor water solubility and fluorescence quenching due to planar stacking. The incorporation of an imidazole ring introduces an electron-donating amino group that enhances fluorescence intensity and disrupts excessive stacking, significantly improving solubility and quantum yield in aqueous environments.
Q: What is the range of emission wavelengths achievable with this synthesis method?
A: By modifying the substituted aryl groups (Ar) and the alkyl groups (R) on the imidazole ring, the emission wavelength can be precisely tuned. The patent data demonstrates a tunable range from green light (507nm) to red light (628nm), making it highly versatile for different optoelectronic applications.
Q: Is this synthetic route suitable for large-scale commercial production?
A: Yes, the route utilizes robust and scalable reactions such as Suzuki coupling and iron-mediated reduction. The reagents, including arylboronic acids and iron powder, are commercially available commodities, and the reaction conditions (temperatures between 0°C and 120°C) are manageable in standard industrial reactors, facilitating commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzothiadiazoloimidazole Dye Supplier
As the demand for high-performance optoelectronic materials continues to surge, partnering with an experienced CDMO is critical for translating laboratory innovations into commercial reality. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including trace metal analysis and detailed impurity profiling, which are paramount for electronic grade chemicals. We understand that the performance of your final device depends entirely on the quality of the intermediate materials we provide.
We invite you to collaborate with our technical team to explore the full potential of this benzothiadiazoloimidazole platform for your specific application. Whether you require custom derivatives with shifted emission wavelengths or optimization of the synthesis for even greater cost efficiency, our experts are ready to assist. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis. We are prepared to provide specific COA data and comprehensive route feasibility assessments to support your R&D and procurement strategies, ensuring a seamless transition from concept to mass production.
