Advanced Visible Light-Sensitized Europium Complexes for High-Performance Optical Applications
Advanced Visible Light-Sensitized Europium Complexes for High-Performance Optical Applications
The development of advanced luminescent materials has reached a critical juncture where efficiency and biocompatibility are paramount. Patent CN101066975B introduces a groundbreaking class of europium complexes that overcome the historical limitations of ultraviolet excitation. These novel compounds, characterized by a unique triazine-pyrazole ligand system coordinated with a europium center, demonstrate exceptional visible light-sensitized luminescent properties. For R&D directors and procurement specialists in the electronic materials sector, this technology represents a significant leap forward in creating high-purity optical materials. The ability to excite these complexes using long-wave visible light minimizes background interference and biological damage, making them ideal candidates for next-generation biosensors, imaging agents, and optoelectronic devices. This report analyzes the technical merits and commercial viability of this synthesis pathway.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional rare earth luminescent materials have long suffered from intrinsic weaknesses related to their excitation mechanisms. Most conventional europium complexes rely heavily on ultraviolet (UV) light for excitation due to the weak f-f transition absorption of the lanthanide ion itself. This dependency on UV radiation poses severe challenges in practical applications, particularly in biological contexts where UV light can cause significant photodamage to living tissues and induce strong autofluorescence from biological matrices. Furthermore, the molar extinction coefficients of these traditional complexes are typically very low, necessitating high concentrations to achieve detectable signal levels. This inefficiency translates to higher material costs and potential toxicity issues in sensitive applications. The narrow excitation windows of older generations of materials also limit their versatility in multiplexed detection systems where distinct excitation sources are required.
The Novel Approach
The methodology outlined in patent CN101066975B fundamentally shifts the paradigm by engineering a highly efficient "antenna effect" within the ligand structure. By incorporating a specific 2-(N,N-dialkylaniline-4-yl)-4,6-di(pyrazol-1-yl)-1,3,5-triazine framework, the new complexes absorb light effectively in the visible region. This structural innovation allows the ligand to harvest photon energy at wavelengths up to 441nm and transfer it efficiently to the europium ion's emitting state. The result is a dramatic enhancement in luminescence efficiency, with quantum yields reaching unprecedented levels for visible-light excitation. This approach not only eliminates the need for harmful UV sources but also significantly improves the signal-to-noise ratio in detection assays. For manufacturers, this means producing materials that offer superior performance metrics without the regulatory and safety burdens associated with high-energy UV excitation sources.
Mechanistic Insights into Triazine-Pyrazole Ligand Sensitization
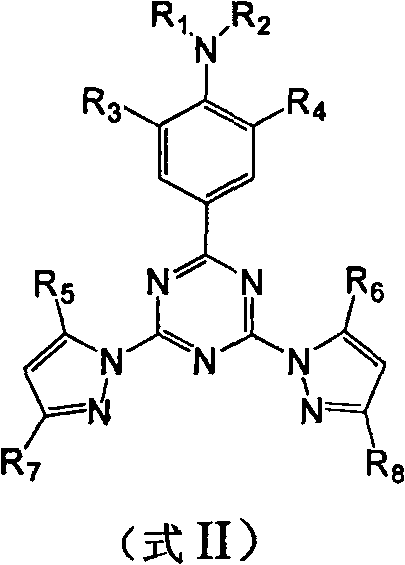
The core of this technological advancement lies in the precise molecular architecture of the sensitizer ligand. The synthesis begins with the formation of a chlorotriazine intermediate, which acts as an electrophilic hub for subsequent nucleophilic attacks by pyrazole anions. This creates a rigid, conjugated system that facilitates effective energy transfer. The electron-donating dialkylaniline group at the 2-position of the triazine ring plays a crucial role in red-shifting the absorption spectrum into the visible range. Once the ligand (Formula II) is formed, it coordinates with the europium center, typically provided as a beta-diketonate complex like Eu(tta)3. The energy transfer mechanism involves the absorption of a photon by the ligand's singlet state, followed by intersystem crossing to the triplet state, and finally resonant energy transfer to the resonant energy level of the Eu3+ ion. This cascade is highly optimized in the disclosed structures, minimizing non-radiative decay pathways.
Impurity control is inherently managed through the robustness of the triazine-pyrazole bond formation. The reaction conditions, which involve moderate temperatures and standard organic solvents, favor the formation of the desired thermodynamic product while minimizing side reactions such as hydrolysis of the chlorotriazine intermediate. The use of specific substituents, such as methyl or ethyl groups on the aniline nitrogen and pyrazole rings, provides steric protection that further stabilizes the complex against degradation. This structural stability ensures that the final product maintains its high quantum yield over extended periods, a critical factor for commercial shelf-life and reliability in field applications. The purity of the final complex is thus a direct function of the controlled stoichiometry and the inherent selectivity of the coordination chemistry involved.
How to Synthesize Visible Light-Sensitized Europium Complex Efficiently
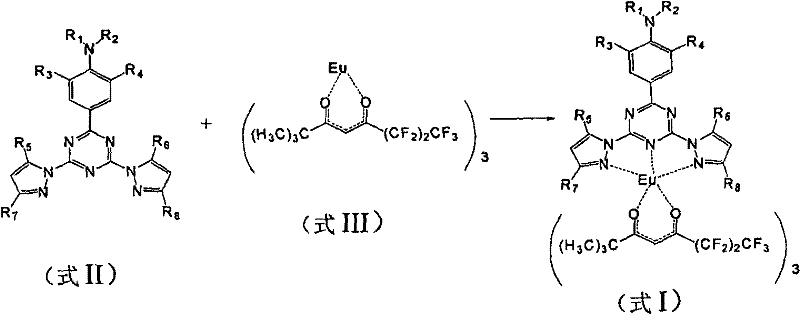
The synthesis protocol described in the patent offers a scalable and reproducible route to these high-value materials. It involves a two-stage process: first, the construction of the organic ligand via nucleophilic substitution, and second, the coordination of this ligand with the europium source. The process is designed to be compatible with standard chemical manufacturing equipment, utilizing common solvents like tetrahydrofuran, toluene, and chloroform. The reaction parameters are well-defined, with temperature ranges spanning from cryogenic conditions for lithiation steps to mild heating for complexation. This flexibility allows for optimization based on specific production scales. Detailed standardized synthesis steps see the guide below.
- Prepare the photosensitive ligand (Formula II) by reacting an aniline derivative with cyanuric chloride followed by substitution with a pyrazole anion.
- Dissolve the europium precursor (Formula III) and the ligand (Formula II) in an organic solvent such as tetrahydrofuran or toluene.
- Mix the solutions at a 1: 1 molar ratio and stir at temperatures between -10°C and 100°C to form the final europium complex (Formula I).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers distinct strategic advantages over legacy technologies. The primary benefit stems from the use of commodity chemical feedstocks. Starting materials such as cyanuric chloride, substituted anilines, and pyrazoles are produced on a massive industrial scale globally, ensuring a stable and competitive pricing structure. Unlike exotic rare earth ligands that require multi-step custom synthesis from scarce precursors, this pathway leverages established supply chains. This reduces the risk of supply disruption and allows for more accurate long-term cost forecasting. Additionally, the high efficiency of the energy transfer means that less active material is required to achieve the same optical performance, effectively reducing the cost-in-use for downstream customers.
- Cost Reduction in Manufacturing: The synthetic route eliminates the need for expensive transition metal catalysts often required in cross-coupling reactions for similar structures. Instead, it relies on straightforward nucleophilic substitutions and coordination chemistry, which are inherently less costly to operate. The reaction proceeds with high atom economy, particularly in the complexation step where the molar ratio is strictly 1:1, minimizing waste of the valuable europium component. Furthermore, the work-up procedures involve standard techniques like solvent evaporation and recrystallization, avoiding the need for costly chromatographic purification on a large scale. These factors combine to drastically simplify the production process and lower the overall manufacturing overhead.
- Enhanced Supply Chain Reliability: The robustness of the chemical bonds formed in the triazine-pyrazole framework ensures that the final product has excellent storage stability. This reduces the logistical complexities associated with cold-chain shipping or inert atmosphere storage that some sensitive luminescent materials require. The synthesis can be performed in a variety of common organic solvents, giving production facilities the flexibility to source solvents locally based on availability and price fluctuations. This adaptability strengthens the resilience of the supply chain against regional disruptions. Moreover, the scalability of the reaction from milligram to kilogram scales has been demonstrated in the patent examples, confirming its viability for commercial volume production without significant re-engineering.
- Scalability and Environmental Compliance: The process operates at moderate temperatures, generally between -10°C and 100°C, which reduces energy consumption compared to high-temperature pyrolysis or extreme cryogenic processes. The solvents used, such as toluene and THF, are widely recycled in industrial settings, facilitating compliance with environmental regulations regarding volatile organic compound (VOC) emissions. The absence of heavy metal catalysts other than the intended lanthanide simplifies waste treatment protocols, as there is no need for specialized removal of toxic transition metal residues. This cleaner profile aligns with increasingly stringent global environmental standards, reducing the regulatory burden on manufacturing sites and enhancing the sustainability credentials of the final product.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these europium complexes. They are derived from the specific experimental data and beneficial effects reported in the patent documentation. Understanding these details is crucial for integrating this material into your product development pipeline.
Q: What is the excitation wavelength range for these europium complexes?
A: The complexes exhibit an excitation window with a peak around 403nm and a tail extending beyond 440nm, allowing for efficient visible light sensitization.
Q: How does the quantum yield compare to traditional UV-excited materials?
A: The disclosed complexes achieve a fluorescence quantum yield as high as 0.94, which is significantly higher than many traditional UV-excited rare earth materials.
Q: Are the raw materials for this synthesis commercially available?
A: Yes, the synthesis utilizes readily available starting materials such as substituted anilines, cyanuric chloride, and pyrazoles, ensuring supply chain stability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Europium Complex Supplier
The technical potential of these visible light-sensitized europium complexes is immense, offering a pathway to superior performance in optical devices and bio-assays. NINGBO INNO PHARMCHEM stands ready to support your transition to this advanced technology. As a seasoned CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch meets the high quantum yield standards defined in the patent. We understand the critical nature of supply continuity for high-purity electronic chemicals and have established robust protocols to maintain consistent quality.
We invite you to engage with our technical procurement team to discuss how this technology can be integrated into your specific applications. Whether you require a Customized Cost-Saving Analysis for your current material spend or need specific COA data to validate performance in your systems, we are prepared to provide comprehensive support. Please reach out to request route feasibility assessments and discover how our manufacturing capabilities can drive value for your organization.
