Advanced 7β-Hydroxysteroid Dehydrogenase Mutants Enable Scalable Ursodeoxycholic Acid Production
The pharmaceutical industry's demand for high-purity ursodeoxycholic acid (UDCA) continues to drive innovation in biocatalytic synthesis, particularly as traditional extraction methods face ethical and supply constraints. Patent CN107099516B introduces a groundbreaking advancement in this sector by disclosing a series of 7β-hydroxysteroid dehydrogenase (7β-HSDH) mutants with significantly improved activity and thermal stability. This technology addresses critical bottlenecks in the enzymatic production of UDCA, specifically targeting the inefficient conversion rates and poor stability of previously available enzymes. By leveraging directed evolution techniques such as error-prone PCR and DNA shuffling, the inventors have successfully engineered variants that operate effectively at high substrate concentrations, offering a robust solution for industrial-scale manufacturing. The strategic shift towards using these enhanced biocatalysts represents a pivotal move away from harsh chemical synthesis routes, aligning with global trends towards greener, more sustainable pharmaceutical intermediate production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of ursodeoxycholic acid has been plagued by significant technical and economic hurdles that limit its commercial viability and scalability. Traditional chemical methods often rely on heavy metal oxidants and high-pressure hydrogenation conditions, which pose severe safety risks and generate substantial toxic waste streams requiring complex disposal protocols. Furthermore, earlier enzymatic approaches utilizing wild-type 7β-HSDH expressed in Escherichia coli suffered from low catalytic efficiency and poor thermal stability, necessitating long reaction times and frequent enzyme replenishment. The intracellular nature of E. coli expression also mandates energy-intensive cell disruption steps to access the enzyme, adding unnecessary complexity and cost to the downstream processing workflow. These cumulative inefficiencies result in lower space-time yields and inconsistent product quality, making it difficult for manufacturers to meet the stringent purity requirements of modern pharmacopeia standards without incurring prohibitive operational expenses.
The Novel Approach
The novel approach detailed in the patent fundamentally reengineers the biocatalytic landscape by introducing highly stable 7β-HSDH mutants expressed via a Pichia pastoris host system. This strategic choice of host organism facilitates the extracellular secretion of the enzyme, thereby eliminating the need for cell lysis and simplifying the recovery process to mere centrifugation and filtration. The engineered mutants, particularly the M6 variant, exhibit exceptional resistance to thermal denaturation, allowing reactions to proceed at elevated temperatures that enhance substrate solubility and reaction kinetics without compromising enzyme integrity. Moreover, the technology enables the use of immobilized enzyme systems coupled with cofactor regeneration enzymes, creating a continuous flow process that can handle substrate concentrations as high as 120 g/L. This leap in performance capability transforms the synthesis from a batch-limited operation into a highly efficient, continuous manufacturing process suitable for large-scale commercial deployment.
Mechanistic Insights into Directed Evolution and Enzyme Immobilization
The core of this technological breakthrough lies in the sophisticated application of protein engineering to optimize the active site and structural stability of the 7β-hydroxysteroid dehydrogenase. Through a rigorous screening process involving high-throughput primary screening and automatic sample injection HPLC secondary screening, specific amino acid substitutions were identified that confer superior properties. For instance, the M6 mutant involves replacing glycine at position 57 with threonine and alanine at position 172 with leucine, modifications that appear to rigidify the protein structure against thermal stress while maintaining or enhancing substrate binding affinity. The patent data illustrates a clear correlation between these specific mutations and the observed improvements in specific activity and residual activity after heat treatment, validating the efficacy of the combined mutation strategy employed.
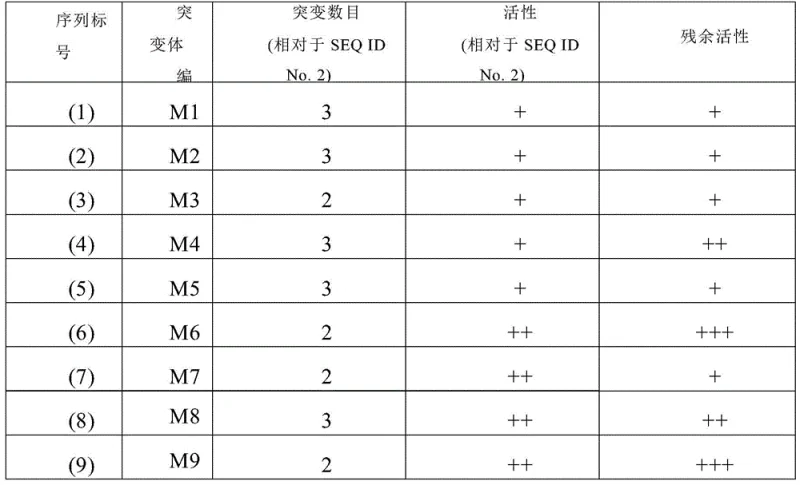
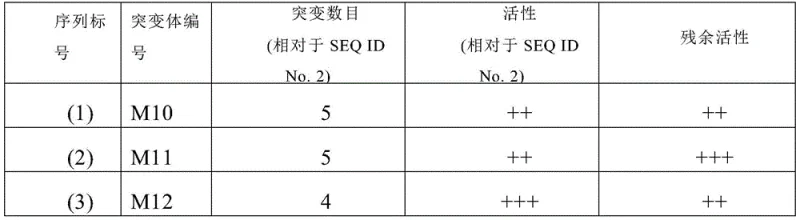
Beyond the genetic modifications, the mechanistic advantage is further amplified by the implementation of co-immobilization techniques on epoxy resin carriers. By physically anchoring the 7β-HSDH mutant alongside glucose dehydrogenase on the same solid support, the system creates a localized microenvironment that facilitates rapid cofactor recycling. This spatial proximity ensures that the oxidized cofactor NADP+ is immediately reduced back to NADPH by the glucose dehydrogenase using glucose as a sacrificial donor, maintaining the driving force for the asymmetric reduction of 7-ketolithocholic acid. This dual-enzyme immobilization not only stabilizes the biocatalyst against mechanical shear and solvent exposure but also allows for easy separation from the reaction mixture, enabling the enzyme to be reused multiple times without significant loss of activity, which is a critical factor for reducing the overall cost of goods in pharmaceutical manufacturing.
How to Synthesize Ursodeoxycholic Acid Efficiently
The synthesis protocol outlined in the patent provides a streamlined pathway for producing high-purity ursodeoxycholic acid using the engineered 7β-HSDH mutants. The process begins with the cultivation of recombinant Pichia pastoris strains, followed by the induction of enzyme secretion and subsequent immobilization on epoxy resin. This preparation yields a robust biocatalyst capable of operating under mild aqueous conditions, significantly reducing the environmental footprint compared to organic solvent-based chemical synthesis. The detailed standardized synthesis steps below outline the precise conditions for reaction setup, cofactor management, and product isolation to ensure optimal yield and purity.
- Construct recombinant expression vectors containing the mutated 7β-HSDH gene (e.g., M6 variant) and transform into Pichia pastoris for extracellular secretion.
- Immobilize the secreted enzyme onto epoxy resin carriers, optionally co-immobilizing with glucose dehydrogenase for cofactor regeneration.
- Perform the biocatalytic reaction at 30-35°C with substrate concentrations up to 120 g/L, achieving >99% conversion within 6 hours.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented enzyme technology offers compelling advantages that directly impact the bottom line and operational resilience. The ability to utilize cheaper substrates like chenodeoxycholic acid (CDCA) through an efficient epimerization process reduces reliance on more expensive intermediates, effectively lowering the raw material cost basis. The enhanced thermal stability of the M6 mutant means that the enzyme inventory has a longer shelf life and can withstand variations in shipping and storage conditions better than conventional biocatalysts, reducing the risk of supply chain disruptions due to spoiled materials. Furthermore, the extracellular expression system simplifies the supply of the enzyme itself, as it can be supplied as a crude supernatant or immobilized beads, removing the need for customers to invest in complex fermentation and lysis infrastructure.
- Cost Reduction in Manufacturing: The transition to this high-efficiency biocatalytic route eliminates the need for expensive heavy metal catalysts and high-pressure equipment associated with traditional chemical synthesis, leading to substantial capital expenditure savings. The high conversion rate of over 99.5% achieved in just 6 hours significantly reduces utility costs related to heating, stirring, and reactor occupancy time, allowing for higher throughput with existing assets. Additionally, the reusability of the immobilized enzyme system means that the effective cost per kilogram of product attributed to the biocatalyst is drastically reduced over multiple batches, providing a clear path to margin expansion.
- Enhanced Supply Chain Reliability: By enabling the use of abundant and low-cost poultry bile-derived chenodeoxycholic acid as a starting material, this technology diversifies the raw material base away from limited bear bile sources or complex synthetic precursors. The robustness of the Pichia pastoris expression system ensures consistent and scalable production of the enzyme catalyst, mitigating the risk of batch-to-batch variability that often plagues biological manufacturing. This reliability translates into more predictable lead times for the final API intermediate, allowing downstream pharmaceutical manufacturers to plan their production schedules with greater confidence and reduced safety stock requirements.
- Scalability and Environmental Compliance: The process operates in aqueous buffers at near-neutral pH, generating minimal hazardous waste and avoiding the use of volatile organic compounds, which simplifies compliance with increasingly stringent environmental regulations. The continuous flow capability demonstrated by the column-packed immobilized enzymes allows for seamless scale-up from laboratory to commercial production volumes without the need for extensive process re-validation. This scalability ensures that suppliers can rapidly respond to surges in market demand for ursodeoxycholic acid, securing a competitive position in the global market for liver disease therapeutics.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this 7β-hydroxysteroid dehydrogenase mutant technology in industrial settings. These answers are derived directly from the experimental data and embodiments provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers. Understanding these specifics is crucial for evaluating the feasibility of integrating this biocatalytic route into existing manufacturing workflows.
Q: How does the M6 mutant improve upon wild-type 7β-HSDH?
A: The M6 mutant (G57T/A172L) demonstrates significantly enhanced thermal stability and catalytic activity. Data shows it retains 78% residual activity after reaction compared to only 21% for the parent strain, while achieving >99.5% conversion in just 6 hours versus 10 hours for the control.
Q: What are the advantages of using Pichia pastoris over E. coli for this enzyme?
A: Using Pichia pastoris allows for extracellular secretion of the enzyme, which drastically simplifies the downstream purification process by eliminating the need for cell disruption and intracellular extraction required with E. coli systems.
Q: Can this process be scaled for continuous production?
A: Yes, the patent describes a continuous flow system where co-immobilized enzymes are packed into columns. This setup allows for the direct conversion of cheap substrates like chenodeoxycholic acid into ursodeoxycholic acid with high space-time yield.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ursodeoxycholic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the 7β-HSDH mutant technology described in Patent CN107099516B for the production of high-value bile acid derivatives. As a leading CDMO partner, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this enzymatic process are fully realized in a GMP-compliant environment. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of ursodeoxycholic acid intermediate we produce adheres to the highest international quality standards. We are committed to leveraging our expertise in enzyme immobilization and biocatalysis to deliver cost-effective and sustainable solutions for our global clientele.
We invite forward-thinking pharmaceutical companies to collaborate with us to harness the full potential of this advanced synthesis route. By partnering with our technical procurement team, you can obtain a Customized Cost-Saving Analysis tailored to your specific volume requirements and supply chain constraints. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized biocatalytic processes can enhance your product portfolio while driving down manufacturing costs and improving supply security.
