Advanced Asymmetric Synthesis of Tetrahydrobenzothiaheptine Oxides for Commercial API Manufacturing
The pharmaceutical industry's relentless pursuit of potent lipid-lowering agents has placed significant focus on Ileal Bile Acid Transport (IBAT) inhibitors, a class of compounds where stereochemistry plays a pivotal role in biological efficacy. Patent CN1106395C discloses a groundbreaking methodology for the preparation of enantiomerically enriched tetrahydrobenzothiaheptine oxides, which serve as critical intermediates for these therapeutic agents. Unlike conventional approaches that struggle with controlling multiple chiral centers within a flexible seven-membered ring system, this invention leverages the chirality of a sulfur atom to direct the stereochemical outcome of the ring closure. This represents a paradigm shift from resolution-based strategies to true asymmetric synthesis, offering a robust pathway for producing high-purity intermediates essential for next-generation cardiovascular drugs.
For R&D directors evaluating process viability, the ability to dictate the configuration at the 4 and 5 positions of the tetrahydrobenzothiaheptine scaffold through a sulfur-centered chiral auxiliary is a significant technical advancement. The patent outlines a sequence where an aryl-3-propionaldehyde sulfoxide, possessing a chiral sulfur center, undergoes cyclization to form the target oxide with high diastereoselectivity. This approach circumvents the limitations of earlier methods described by Higashikawa or Giordano, which were either restricted to microgram scales or failed to control stereochemistry at all relevant positions. By establishing the chiral environment early in the synthesis via enantioselective oxidation of a sulfide precursor, the subsequent carbon-carbon bond formation becomes highly predictable and efficient.
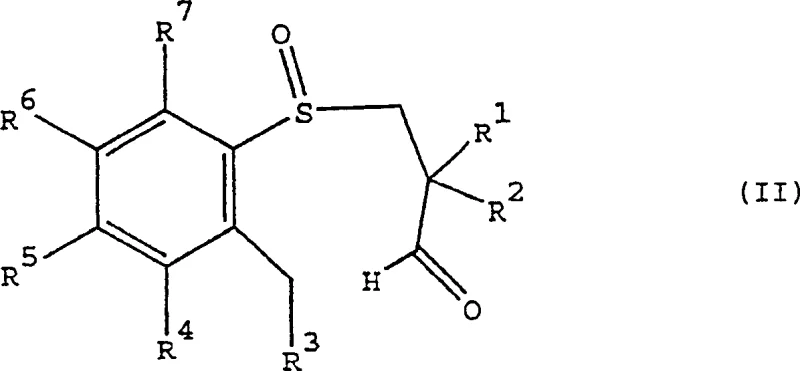
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzothiazepine and related seven-membered heterocycles has been plagued by inefficiencies in stereocontrol. Prior art methods, such as those cited in the background of CN1106395C, often relied on the optical resolution of racemic mixtures. For instance, separating enantiomers using chiral crown ether chromatography is technically feasible but economically prohibitive for commercial manufacturing due to the high cost of stationary phases and the inherent 50% yield loss of the unwanted isomer. Furthermore, microbial reduction methods, while capable of inducing chirality, introduce significant supply chain volatility; maintaining viable bacterial cultures requires stringent biological controls and is difficult to scale beyond laboratory quantities. These traditional pathways also frequently fail to address the stereochemistry at the 4 and 5 positions of the ring simultaneously, leading to complex mixtures of diastereomers that require tedious and yield-eroding purification steps.
The Novel Approach
The methodology presented in CN1106395C offers a decisive break from these constraints by utilizing a chiral sulfoxide strategy. The core innovation lies in the cyclization of an enantiomerically enriched aryl-3-propionaldehyde sulfoxide. In this system, the sulfur atom acts as a chiral center that influences the conformation of the transition state during ring closure. This allows for the simultaneous establishment of chirality at the sulfur and the adjacent carbon atoms (positions 4 and 5) with high fidelity. The process avoids the need for expensive chiral columns or fragile biological catalysts, relying instead on well-defined chemical oxidants and base-catalyzed condensation reactions. This chemical robustness translates directly into a more reliable supply chain, as the reagents are stable, shelf-stable, and readily sourced from global chemical suppliers, ensuring consistent batch-to-batch quality.
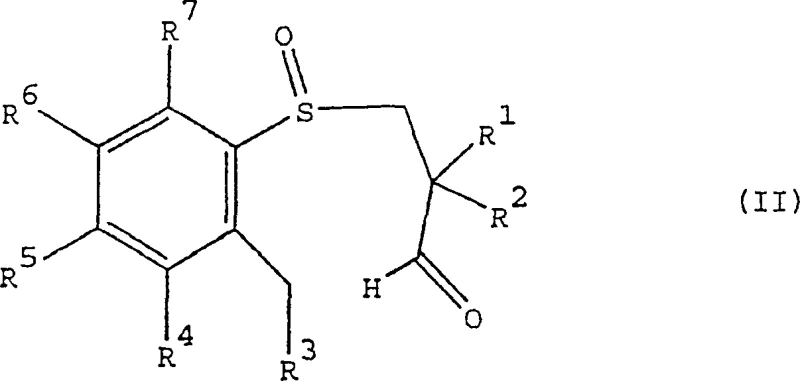
Mechanistic Insights into Chiral Sulfoxide-Directed Cyclization
The mechanistic elegance of this process resides in the transfer of chirality from the sulfur atom to the carbon framework. The synthesis begins with the enantioselective oxidation of an achiral aryl-3-hydroxypropyl sulfide. Using chiral oxidants such as camphorsulfonyl oxaziridines or chiral (salen) metal complexes, the sulfur atom is converted into a chiral sulfoxide with high enantiomeric excess. This chiral information is then preserved during the subsequent oxidation of the primary alcohol to an aldehyde. When this chiral aldehyde-sulfoxide intermediate is treated with a base, such as potassium tert-butoxide, it undergoes an intramolecular aldol-type condensation. The bulky lone pair and oxygen on the chiral sulfur create a steric environment that favors the formation of one specific diastereomer of the seven-membered ring. This ensures that the resulting tetrahydrobenzothiaheptine-1-oxide possesses the desired (4R,5R) or (4S,5S) configuration, which is critical for the biological activity of the final IBAT inhibitor drug substance.
From an impurity profile perspective, this mechanism is highly advantageous. Because the stereochemistry is driven by the thermodynamic and kinetic preferences of the cyclization transition state, the formation of the wrong diastereomer is minimized at the source. This contrasts sharply with resolution methods where the unwanted isomer is produced in equal amounts and must be physically removed. In the patented process, side reactions are primarily limited to over-oxidation or incomplete cyclization, both of which are chemically distinct from the product and can be removed via standard crystallization or chromatography. The result is a cleaner crude reaction mixture, reducing the burden on downstream purification units and increasing the overall mass balance efficiency of the manufacturing campaign.
How to Synthesize Tetrahydrobenzothiaheptine Oxides Efficiently
The synthesis protocol detailed in the patent provides a clear roadmap for laboratory and pilot-scale production. It involves three critical stages: the generation of the chiral sulfoxide, the oxidation to the aldehyde, and the final cyclization. Each step utilizes standard organic transformations that are well-understood by process chemists, minimizing the risk of unexpected scale-up issues. The use of common solvents like dichloromethane, THF, and diglyme further simplifies the engineering requirements for the reaction vessels. For a detailed breakdown of the specific molar ratios, temperature profiles, and workup procedures required to achieve the reported yields and enantiomeric excess, please refer to the standardized guide below.
- Perform enantioselective oxidation of an aryl-3-hydroxypropyl sulfide using a chiral oxidant like camphorsulfonyl oxaziridine to generate a chiral sulfoxide.
- Oxidize the hydroxyl group of the chiral sulfoxide intermediate to an aldehyde using sulfur trioxide-pyridine complex.
- Execute intramolecular cyclization of the resulting aryl-3-propionaldehyde sulfoxide using a strong base like potassium tert-butoxide to close the seven-membered ring.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic benefits beyond mere technical feasibility. The shift from biological or chromatographic resolution to chemical asymmetric synthesis fundamentally alters the cost structure and risk profile of the intermediate's production. By eliminating the dependency on specialized chiral stationary phases or live bacterial cultures, the process reduces exposure to single-source supplier risks and volatile pricing associated with biotech-derived reagents. The robustness of the chemical steps ensures that production schedules are less likely to be disrupted by biological contamination or column degradation, leading to more predictable lead times for API manufacturing.
- Cost Reduction in Manufacturing: The most significant economic driver here is the avoidance of yield-limiting resolution steps. Traditional resolution caps the maximum theoretical yield at 50%, effectively doubling the raw material cost per unit of active isomer. This asymmetric synthesis pathway aims for yields significantly higher than this limit by constructing the desired isomer directly. Furthermore, the removal of preparative chiral HPLC steps eliminates a major cost center, as these columns are expensive to purchase, have limited lifespans, and require large volumes of high-grade solvents. The net result is a substantial reduction in the cost of goods sold (COGS) for the intermediate.
- Enhanced Supply Chain Reliability: The reagents specified in the patent, such as potassium tert-butoxide, sulfur trioxide-pyridine complex, and various oxaziridines, are commodity chemicals available from multiple global vendors. This diversification of the supply base mitigates the risk of shortages that often plague proprietary enzymes or custom-synthesized chiral catalysts. Additionally, the chemical stability of the intermediates allows for potential storage between steps, providing flexibility in production planning and inventory management that is not possible with unstable biological cultures.
- Scalability and Environmental Compliance: The process is designed for scalability, utilizing reaction conditions that are compatible with standard stainless steel reactors. The absence of biological waste streams simplifies effluent treatment, as the waste consists primarily of organic solvents and salts that can be managed through established incineration or recovery protocols. The high selectivity of the reaction also means less solvent is consumed per kilogram of product, as fewer purification cycles are needed to remove diastereomeric impurities, aligning with green chemistry principles and reducing the environmental footprint of the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. They are derived from the specific embodiments and claims within CN1106395C, focusing on the practical aspects of stereocontrol and process optimization. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: How does this process control stereochemistry at the 4 and 5 positions?
A: The process utilizes a chiral sulfur atom in the sulfoxide intermediate as a stereochemical director. During the base-catalyzed cyclization, the existing chirality at the sulfur atom induces high diastereoselectivity, establishing the desired configuration at the carbon centers of the seven-membered ring without needing external chiral auxiliaries on the carbon chain.
Q: What are the advantages over traditional resolution methods?
A: Traditional methods often rely on resolving racemic mixtures via chiral chromatography or enzymatic reduction, which inherently limits yield to a maximum of 50% and requires expensive separation media. This asymmetric synthesis builds chirality directly, potentially doubling theoretical yield and eliminating the need for costly preparative chiral HPLC steps.
Q: Is this method suitable for large-scale production?
A: Yes, the reagents used, such as potassium tert-butoxide and standard oxidants, are commercially available and the reaction conditions do not require extreme pressures or temperatures. The elimination of complex biological fermentation steps or specialized chromatographic columns makes the process highly amenable to kilogram and ton-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrahydrobenzothiaheptine Oxides Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of life-saving cardiovascular medications. Our technical team has extensively analyzed the methodologies described in CN1106395C and possesses the expertise to execute this complex asymmetric synthesis with precision. We offer extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from clinical trials to market launch. Our facility is equipped with rigorous QC labs and stringent purity specifications to guarantee that every batch of tetrahydrobenzothiaheptine oxides meets the exacting standards required for pharmaceutical applications.
We invite you to collaborate with us to optimize this synthesis for your specific needs. Our engineers can provide a Customized Cost-Saving Analysis to demonstrate how implementing this route can lower your overall production expenses. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us partner with you to secure a reliable, cost-effective, and high-quality supply of these essential pharmaceutical intermediates.
