Advanced Synthesis of Symmetric Imidazolium Ionic Liquids for Commercial Scale-Up
Advanced Synthesis of Symmetric Imidazolium Ionic Liquids for Commercial Scale-Up
The development of functionalized ionic liquids has become a cornerstone in modern materials science, particularly for applications requiring high stability and tunable physicochemical properties. Patent CN101250159A introduces a significant breakthrough in this domain by detailing the synthesis of 1,3-bis(2-methoxy-2-oxyethyl)imidazolium chloride, a novel symmetric ionic liquid. This specific compound distinguishes itself through the incorporation of symmetrical functional groups on the imidazolium cation, which enhances its potential utility in conductive materials and polyelectrolyte systems. Unlike traditional mono-functionalized variants, this symmetric architecture offers superior modulation of polymer chain interactions, thereby improving the overall performance of solid electrolytes. For R&D directors and procurement specialists in the electronic chemical sector, understanding the nuances of this synthesis route is critical for securing a reliable supply of high-performance precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of functional ionic liquids has predominantly focused on mono-substituted imidazolium derivatives, such as 1-methyl-3-(2-hydroxyethyl)imidazolium salts. These conventional methods often suffer from limited structural versatility, as the presence of a single functional group restricts the ability to modify polymer chains effectively. Furthermore, traditional alkylation processes frequently require harsh conditions or expensive phase-transfer catalysts that complicate downstream purification. The lack of symmetry in these molecules can lead to inconsistent packing in solid states, resulting in variable conductivity and thermal stability. For manufacturers aiming to produce high-purity electronic chemicals, these inconsistencies pose significant challenges in quality control and batch-to-batch reproducibility, ultimately driving up the cost of goods sold.
The Novel Approach
The methodology outlined in the patent data presents a robust alternative by utilizing a direct quaternization strategy between imidazole and methyl chloroacetate. This approach leverages the nucleophilicity of the imidazole nitrogen atoms to attack the electrophilic carbon of the chloroacetate, facilitated by an in-situ generated sodium methoxide catalyst. By carefully controlling the molar ratio of reactants to approximately 1:2.1, the process ensures double substitution, yielding the desired symmetric cation with high selectivity. This novel route eliminates the need for complex protecting group chemistry often required in asymmetric syntheses. Consequently, the process flow is significantly streamlined, reducing the number of unit operations and minimizing solvent waste, which aligns perfectly with the principles of green chemistry and sustainable manufacturing.
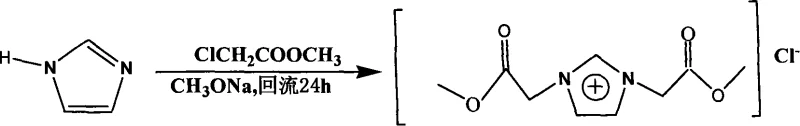
Mechanistic Insights into Sodium-Catalyzed Quaternization
The core of this synthesis lies in the precise generation of the alkoxide catalyst and the subsequent nucleophilic substitution mechanism. Initially, metallic sodium reacts with anhydrous methanol at cryogenic temperatures (-1 to 0°C) to form sodium methoxide. This strong base is essential for deprotonating trace impurities and activating the reaction medium without degrading the sensitive ester functionalities present in the methyl chloroacetate. As the imidazole and alkylating agent are introduced, the nitrogen atoms on the imidazole ring act as nucleophiles, displacing the chloride ion in an SN2 reaction. The low-temperature initiation is crucial to prevent poly-alkylation side reactions or ester hydrolysis, ensuring that the reaction proceeds cleanly towards the bis-substituted product. This mechanistic control is vital for maintaining the integrity of the ester groups, which serve as the reactive handles for future polymer modifications.
Impurity control is managed through a multi-stage purification protocol that addresses both organic byproducts and colored contaminants. Following the reaction, unreacted starting materials and solvents are removed under reduced pressure, leveraging the difference in volatility between the ionic liquid and the organic components. The crude product is then subjected to activated carbon treatment, which adsorbs high-molecular-weight colored impurities formed during the exothermic quaternization. Final purification via silica gel chromatography using a specific methanol-chloroform eluent system ensures the removal of any mono-substituted intermediates. This rigorous purification sequence guarantees a product with the stringent purity specifications required for electronic applications, where trace ionic contaminants can severely degrade device performance.
How to Synthesize 1,3-Bis(2-Methoxy-2-Oxyethyl)imidazolium Chloride Efficiently
Executing this synthesis at a commercial scale requires strict adherence to the thermal profiles and stoichiometric ratios defined in the patent examples. The process begins with the preparation of the catalyst solution, followed by the controlled addition of reactants to manage the exotherm. Detailed standard operating procedures regarding the specific crystallization temperatures and washing protocols are essential to achieve the reported yields of over 70%.
- Prepare sodium methoxide catalyst by reacting sodium metal with anhydrous methanol at -1 to 0°C for 5-7 hours.
- Add imidazole and methyl chloroacetate (molar ratio 1: 2.1-2.2) and react at low temperature followed by heating to 10-60°C for 20-24 hours.
- Purify the crude product via vacuum distillation, activated carbon decolorization, silica gel chromatography, and crystallization at 0 to -2°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this symmetric ionic liquid synthesis offers tangible benefits regarding cost structure and supply reliability. The primary advantage stems from the use of commodity chemicals; both imidazole and methyl chloroacetate are widely available bulk intermediates with stable global supply chains. This reduces the risk of raw material shortages that often plague specialty chemical manufacturing. Furthermore, the elimination of exotic catalysts or rare metal complexes simplifies the sourcing strategy and lowers the overall input costs. The process operates at moderate temperatures after the initial cooling phase, allowing for the use of standard stainless steel reactors rather than specialized lined vessels, which significantly reduces capital expenditure for scale-up.
- Cost Reduction in Manufacturing: The synthetic route achieves cost optimization by removing the need for expensive transition metal catalysts and complex purification resins. By relying on a straightforward filtration and crystallization workflow, the operational expenditure associated with consumables is drastically reduced. Additionally, the high atom economy of the quaternization reaction minimizes waste disposal costs, contributing to a leaner manufacturing budget. This efficiency allows for competitive pricing strategies when supplying high-purity electronic chemicals to downstream battery and polymer manufacturers.
- Enhanced Supply Chain Reliability: Utilizing readily available feedstocks ensures that production schedules are not disrupted by niche supplier bottlenecks. The robustness of the reaction conditions means that the process is less sensitive to minor fluctuations in utility supplies, such as cooling water temperature variations. This resilience translates to consistent lead times for customers, a critical factor for Just-In-Time manufacturing environments in the electronics sector. Establishing a supply line based on this chemistry provides a stable foundation for long-term procurement contracts.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from laboratory glassware to multi-ton industrial reactors without fundamental changes to the chemistry. The use of methanol and ethyl acetate, which are easily recoverable solvents, supports closed-loop recycling systems that minimize environmental impact. This alignment with green chemistry principles facilitates easier regulatory approval and reduces the burden of hazardous waste management. Consequently, manufacturers can expand capacity to meet growing demand for energy storage materials while maintaining strict environmental compliance standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this symmetric ionic liquid. These insights are derived directly from the experimental data and characterization results provided in the patent documentation. Understanding these details helps stakeholders evaluate the feasibility of integrating this material into their existing product lines.
Q: What is the typical yield for this ionic liquid synthesis?
A: According to patent CN101250159A, the optimized process yields between 65.2% and 76.2% pure product after crystallization and chromatography.
Q: Why is temperature control critical in the first stage of reaction?
A: Maintaining -1 to 0°C during the initial catalyst formation and addition prevents side reactions and ensures the selective formation of the symmetric bis-substituted cation.
Q: What are the electrochemical properties of the final product?
A: The synthesized ionic liquid exhibits a conductivity of 0.4-0.5 s/m and a wide electrochemical window of 2.0-2.2V, making it suitable for electrolyte applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,3-Bis(2-Methoxy-2-Oxyethyl)imidazolium Chloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced ionic liquids play in the next generation of energy storage and conductive polymer technologies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and speed. We maintain stringent purity specifications through our rigorous QC labs, utilizing state-of-the-art analytical instrumentation to verify every batch against the highest industry standards. Our commitment to quality ensures that the ionic liquids we supply perform consistently in your final applications, whether for solid-state batteries or specialized polyelectrolytes.
We invite you to collaborate with us to optimize your supply chain for these high-value intermediates. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your R&D and manufacturing goals. Let us be your partner in driving innovation through reliable chemical supply.
