Scalable Synthesis of 4-Bromo-2-Methylbenzoic Acid: A Cost-Effective Route for Veterinary Drug Manufacturing
The global demand for effective veterinary parasiticides continues to drive innovation in the synthesis of key intermediates like 4-bromo-2-methylbenzoic acid, a critical building block for Fluralaner. Patent CN115304472A introduces a robust, five-step synthetic methodology that addresses longstanding challenges in cost, safety, and scalability associated with traditional manufacturing routes. By utilizing 3-methylaniline as a readily available starting material, this novel approach eliminates the reliance on expensive precursors and toxic cyanide reagents, offering a streamlined pathway suitable for large-scale industrial production. The process integrates classic organic transformations including Vilsmeier-Haack formylation, oxime dehydration, and Sandmeyer bromination, optimized through precise temperature control and solvent selection to maximize yield and purity at every stage.
For R&D directors and process chemists, the significance of this patent lies in its ability to deliver high-purity intermediates while simplifying downstream processing. The strategic design of the reaction sequence ensures that impurities are minimized through selective para-formylation and efficient crystallization protocols, reducing the burden on purification units. This technical advancement not only enhances the economic viability of Fluralaner production but also aligns with modern green chemistry principles by avoiding heavy metal catalysts and hazardous cyanide salts. As the veterinary pharmaceutical sector seeks more sustainable and cost-efficient supply chains, this synthesis route represents a pivotal shift towards safer and more reliable manufacturing practices.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies for synthesizing 4-bromo-2-methylbenzoic acid have been plagued by significant economic and safety drawbacks that hinder their industrial applicability. For instance, Patent WO2017034994 describes a route starting from (4-bromo-2-methylphenyl)methanol, a precursor that is not only difficult to source commercially but also commands a prohibitively high price, making the overall process economically unfeasible for mass production. Another approach reported in Patent CN112441907 utilizes 4-bromo-1,2-xylene with noble metal catalysis; however, this method suffers from poor selectivity and low yields, with the main product yield reported at only 43%, alongside the generation of multiple difficult-to-separate by-products that complicate purification.
Furthermore, literature reports such as those by Chiarelli et al. rely on 4-bromo-2-methylbenzonitrile as a starting material, necessitating the use of highly toxic cyanide compounds. The handling of cyanides requires specialized equipment, rigorous safety protocols, and expensive waste treatment facilities, all of which inflate operational costs and introduce substantial liability risks. These conventional pathways collectively fail to meet the dual demands of cost-efficiency and environmental safety required by modern pharmaceutical supply chains, creating an urgent need for a superior alternative that leverages cheap, safe, and abundant raw materials.
The Novel Approach
The methodology disclosed in CN115304472A fundamentally restructures the synthesis logic by starting from 3-methylaniline, a commodity chemical that is inexpensive and widely available. This new route bypasses the need for precious metal catalysts and toxic cyanides entirely, replacing them with standard reagents like phosphorus oxychloride, hydroxylamine hydrochloride, and sodium formate. The process begins with a highly selective formylation to generate 2-methyl-4-aminobenzaldehyde, followed by conversion to an oxime and subsequent dehydration to the nitrile, effectively installing the carboxylic acid precursor without hazardous cyanation steps.
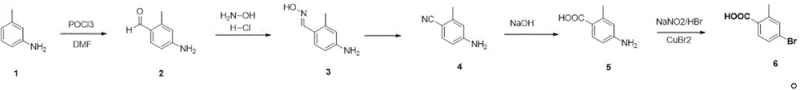
The final stages involve alkaline hydrolysis to form the benzoic acid derivative and a classic Sandmeyer bromination to install the bromine atom at the desired position. This sequence is characterized by high yields at each step, with the formylation step achieving over 87% yield and the final bromination reaching up to 89.4%. The use of common solvents like ethanol and DMF, which are miscible with water, allows for simple product isolation via water addition and filtration, eliminating the need for energy-intensive distillation or complex extraction procedures. This operational simplicity translates directly into reduced manufacturing costs and enhanced scalability for commercial production.
Mechanistic Insights into Vilsmeier-Haack Formylation and Sandmeyer Substitution
The cornerstone of this synthesis is the initial Vilsmeier-Haack formylation, where 3-methylaniline reacts with phosphorus oxychloride (POCl3) in a polar solvent like DMF or ethanol. Mechanistically, POCl3 reacts with the solvent to generate a reactive chloroiminium ion electrophile, which attacks the electron-rich aromatic ring of the aniline derivative. The amino group acts as a strong activating group, directing the electrophilic substitution selectively to the para-position due to steric hindrance at the ortho-positions caused by the methyl group. Strict temperature control between 0-5°C during the addition of POCl3 is critical to suppress side reactions such as over-chlorination or polymerization, ensuring that the reaction proceeds cleanly to form 2-methyl-4-aminobenzaldehyde with a purity exceeding 99.5%.
The final transformation involves a Sandmeyer reaction, a radical substitution mechanism that replaces the diazonium group with a bromine atom. In this step, 2-methyl-4-aminobenzoic acid is diazotized using sodium nitrite and hydrobromic acid at low temperatures (0-5°C) to form the unstable diazonium salt. The subsequent addition of copper(II) bromide facilitates a single-electron transfer process, generating an aryl radical that abstracts a bromine atom from the copper complex. This mechanism is highly specific and avoids the formation of phenolic by-products that often plague aqueous diazonium reactions. The careful maintenance of low temperatures throughout the diazotization and coupling phases prevents the decomposition of the diazonium intermediate, thereby maximizing the yield of the target bromo-acid and minimizing the formation of tar-like impurities.
How to Synthesize 4-Bromo-2-Methylbenzoic Acid Efficiently
The synthesis of this high-value veterinary intermediate requires precise adherence to reaction conditions to maintain the high purity and yield profiles described in the patent. The process is divided into five distinct operational stages, each requiring specific stoichiometric ratios and thermal management strategies to optimize the conversion of intermediates. While the general workflow is straightforward, the success of the scale-up depends heavily on the control of exothermic events during reagent addition and the efficiency of the crystallization steps used for product isolation. Detailed standard operating procedures for each transformation are essential for reproducibility and quality assurance in a GMP environment.
- Perform Vilsmeier-Haack formylation of 3-methylaniline using POCl3 in DMF or ethanol at 0-65°C to obtain 2-methyl-4-aminobenzaldehyde.
- React the aldehyde with hydroxylamine hydrochloride in ethanol or DMF under reflux to generate the corresponding oxime intermediate.
- Dehydrate the oxime using sodium formate and formic acid at 100°C to yield 2-methyl-4-aminobenzonitrile.
- Hydrolyze the nitrile group with sodium hydroxide in water under reflux conditions to produce 2-methyl-4-aminobenzoic acid.
- Execute Sandmeyer bromination using sodium nitrite, hydrobromic acid, and copper bromide at 0-5°C to finalize 4-bromo-2-methylbenzoic acid.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this synthetic route offers transformative advantages by decoupling production costs from volatile precious metal markets and scarce specialty reagents. The exclusive use of commodity chemicals such as 3-methylaniline, sodium hydroxide, and hydrobromic acid ensures a stable and resilient supply chain that is less susceptible to geopolitical disruptions or supplier monopolies. By eliminating the need for expensive palladium or platinum catalysts, the process significantly reduces the direct material cost per kilogram of the final product, allowing for more competitive pricing strategies in the global veterinary drug market.
- Cost Reduction in Manufacturing: The elimination of toxic cyanide reagents removes the substantial overhead costs associated with hazardous waste disposal and specialized containment infrastructure. Furthermore, the high selectivity of the formylation and bromination steps minimizes the loss of raw materials to by-products, effectively increasing the overall mass balance efficiency. The ability to isolate products through simple water-induced crystallization rather than complex chromatography or distillation further drives down utility and labor costs, resulting in a leaner and more profitable manufacturing operation.
- Enhanced Supply Chain Reliability: Sourcing 3-methylaniline is far more reliable than procuring specialized brominated alcohols or nitriles, as it is a bulk chemical produced by multiple manufacturers worldwide. This diversity of supply sources mitigates the risk of production stoppages due to raw material shortages. Additionally, the robustness of the reaction conditions, which tolerate standard industrial equipment without requiring exotic alloys or high-pressure vessels, ensures that production can be easily transferred between different manufacturing sites without significant re-validation efforts.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing solvents like ethanol and water that are environmentally benign and easy to recover or treat. The absence of heavy metal residues in the final product simplifies the regulatory approval process for veterinary applications, as extensive testing for metal impurities is not required. This environmental compatibility not only reduces the carbon footprint of the manufacturing process but also aligns with the increasingly strict sustainability mandates imposed by global regulatory bodies and end-users.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on safety, efficiency, and operational feasibility for potential partners and licensees.
Q: Why is this synthesis route preferred over cyanide-based methods?
A: Traditional routes often utilize highly toxic cyanides which pose severe safety and environmental risks. This patented method replaces cyanide chemistry with a safer dehydration-hydrolysis sequence using common reagents like sodium formate and sodium hydroxide, significantly reducing hazardous waste disposal costs and operational risks.
Q: What are the cost advantages of using 3-methylaniline as the starting material?
A: Unlike previous methods relying on expensive precursors like (4-bromo-2-methylphenyl)methanol, 3-methylaniline is a commodity chemical available at very low cost. Combined with the avoidance of precious metal catalysts, this raw material choice drastically lowers the overall production cost per kilogram.
Q: How does this process ensure high purity for pharmaceutical applications?
A: The process utilizes specific temperature controls (0-5°C for critical additions) and solvent systems (DMF/Ethanol) that facilitate easy crystallization and washing steps. Each intermediate achieves purity levels exceeding 99%, ensuring the final API intermediate meets stringent quality specifications without complex chromatographic purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Bromo-2-Methylbenzoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation veterinary therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of 4-bromo-2-methylbenzoic acid meets the exacting standards required for API synthesis, providing our clients with a foundation of quality and reliability.
We invite procurement leaders and R&D directors to collaborate with us to leverage this advanced synthetic technology for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize both the cost and security of your veterinary drug manufacturing operations.
