Advanced Catalytic Acylation for Commercial Scale Hydroquinone Diester Manufacturing
Advanced Catalytic Acylation for Commercial Scale Hydroquinone Diester Manufacturing
The chemical landscape for producing high-value pharmaceutical intermediates has been significantly transformed by the innovations detailed in patent CN1219531A, filed in June 1999. This pivotal intellectual property introduces a robust methodology for synthesizing highly pure hydroquinone diester derivatives, specifically targeting the efficient conversion of ketoisophorone into valuable precursors for Vitamin E and polymer stabilizers. The core breakthrough lies in the simplification of the purification stage, moving away from labor-intensive extraction and low-efficiency recrystallization techniques towards a streamlined crystallization process using aqueous organic solvents. For R&D directors and process engineers, this represents a critical evolution in synthetic strategy, offering a pathway to achieve purity levels exceeding 99.9% while drastically reducing the operational complexity associated with traditional methods. The ability to control impurity profiles, particularly the suppression of catechol diester derivatives, positions this technology as a cornerstone for modern fine chemical manufacturing where specification adherence is paramount.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the advent of this technology, the industry relied heavily on methods disclosed in earlier literature, such as Japanese Patent Application No. 7632/1972, which presented substantial bottlenecks for commercial scale-up. These conventional processes typically involved neutralizing the reaction mixture followed by extraction and concentration under reduced pressure, creating multiple opportunities for yield loss and contamination. A critical flaw in these legacy methods was the reliance on hexane for recrystallization; due to the low solubility of the target compound in hexane, massive volumes of solvent were required, driving up both raw material costs and environmental waste management burdens. Furthermore, the separation efficiency was poor, often resulting in products with broad melting point ranges (97-107°C) and limited purity, as the by-products were not effectively removed. This inefficiency not only compromised the quality of the final pharmaceutical intermediate but also created significant safety hazards associated with handling large quantities of volatile hydrocarbon solvents in an industrial setting.
The Novel Approach
The novel approach described in the patent fundamentally reengineers the downstream processing by leveraging the unique solubility characteristics of the reaction product in polar solvent systems. Instead of resorting to complex extraction workflows, the process allows the reaction product of ketoisophorone and an acylating agent to undergo direct crystallization using a mixed solvent system comprising an organic carboxylic acid and water. 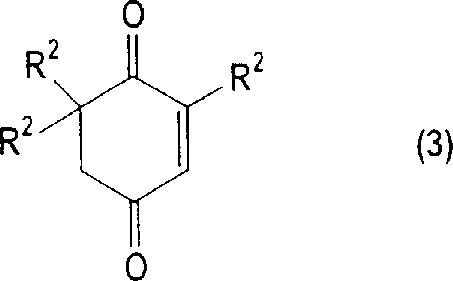 This strategic shift eliminates the need for solvent evaporation and recovery steps that plague traditional methods, thereby enhancing the overall mass balance of the production line. By utilizing solvents that correspond to the acylating agent, such as acetic acid when using acetic anhydride, the process creates a synergistic environment where the reaction by-products are seamlessly integrated into the crystallization medium. This results in a highly efficient operation where the target hydroquinone diester precipitates with exceptional clarity and definition, bypassing the limitations of hexane-based systems and delivering a product that meets stringent international quality standards with minimal post-processing.
This strategic shift eliminates the need for solvent evaporation and recovery steps that plague traditional methods, thereby enhancing the overall mass balance of the production line. By utilizing solvents that correspond to the acylating agent, such as acetic acid when using acetic anhydride, the process creates a synergistic environment where the reaction by-products are seamlessly integrated into the crystallization medium. This results in a highly efficient operation where the target hydroquinone diester precipitates with exceptional clarity and definition, bypassing the limitations of hexane-based systems and delivering a product that meets stringent international quality standards with minimal post-processing.
Mechanistic Insights into Acid-Catalyzed Cyclization and Acylation
The chemical transformation at the heart of this process involves the acid-catalyzed reaction of a cyclohex-2-ene-1,4-dione derivative, specifically 2,6,6-trimethylcyclohex-2-ene-1,4-dione (Ketoisophorone), with various acylating agents. The mechanism proceeds through the activation of the carbonyl groups by protonic or Lewis acid catalysts, facilitating the nucleophilic attack by the acylating species to form the diester linkage. The versatility of the catalytic system is a key feature, accommodating a broad spectrum of acids ranging from mineral acids like sulfuric acid and hydrochloric acid to sophisticated superacids and solid acid catalysts like ion-exchange resins. This flexibility allows manufacturers to tune the reaction kinetics and selectivity based on specific substrate requirements, ensuring high conversion rates often exceeding 95% within moderate temperature ranges of 50-110°C. The precise control over reaction conditions prevents the degradation of the sensitive quinone structure, maintaining the integrity of the molecular framework essential for downstream applications in antioxidant synthesis.
A critical aspect of this mechanistic pathway is the inherent control over impurity formation, specifically the minimization of catechol diester derivatives which are structurally similar by-products. 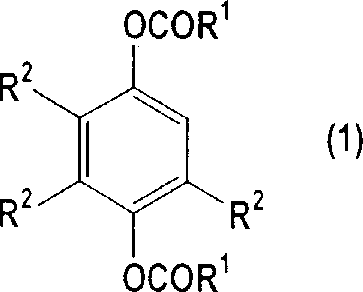 Through the optimized crystallization protocol, the process exploits the differential solubility between the desired hydroquinone diester (Formula 1) and the catechol impurity (Formula 2). The patent data indicates that by carefully adjusting the ratio of organic carboxylic acid to water in the crystallization solvent, the content of the catechol by-product can be suppressed to levels as low as 0.001-2% by weight. This level of impurity control is achieved without the need for chromatographic separation, relying instead on thermodynamic principles during the phase transition from solution to solid state. For quality assurance teams, this means a consistent and predictable impurity profile that simplifies analytical validation and ensures the final API intermediate meets the rigorous specifications required for human consumption or high-performance polymer applications.
Through the optimized crystallization protocol, the process exploits the differential solubility between the desired hydroquinone diester (Formula 1) and the catechol impurity (Formula 2). The patent data indicates that by carefully adjusting the ratio of organic carboxylic acid to water in the crystallization solvent, the content of the catechol by-product can be suppressed to levels as low as 0.001-2% by weight. This level of impurity control is achieved without the need for chromatographic separation, relying instead on thermodynamic principles during the phase transition from solution to solid state. For quality assurance teams, this means a consistent and predictable impurity profile that simplifies analytical validation and ensures the final API intermediate meets the rigorous specifications required for human consumption or high-performance polymer applications.
How to Synthesize Trimethyl Hydroquinone Diacetate Efficiently
The synthesis of Trimethyl Hydroquinone Diacetate via this patented route offers a practical blueprint for laboratories and pilot plants aiming to replicate these high-efficiency results. The procedure begins with the charging of Ketoisophorone and an excess of acetic anhydride into a reactor equipped with temperature control and agitation, followed by the addition of a catalytic amount of sulfuric acid. The mixture is heated to facilitate the acylation reaction until gas chromatography confirms the complete consumption of the starting material, typically indicated by a conversion rate of nearly 100%. Following the reaction, the mixture is neutralized with a dilute aqueous base, such as sodium hydroxide, to quench the acid catalyst and prepare the system for the critical crystallization step. The detailed standardized synthesis steps, including specific molar ratios, temperature ramping rates, and filtration protocols, are outlined below to ensure reproducibility and safety during scale-up operations.
- React ketoisophorone with an acylating agent like acetic anhydride in the presence of an acid catalyst at 50-110°C.
- Neutralize the reaction mixture with an alkali solution if liquid acid catalysts are used, or filter if solid catalysts are employed.
- Add a mixed solvent of organic carboxylic acid and water to the mixture and cool gradually to crystallize the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthesis methodology translates into tangible strategic advantages that extend beyond mere technical feasibility. The elimination of hexane from the purification process removes a significant cost center associated with solvent purchase, recovery, and hazardous waste disposal, leading to substantial cost savings in the overall manufacturing budget. Furthermore, the simplified workflow reduces the number of unit operations required, which directly correlates to lower energy consumption and reduced labor hours per batch. This operational efficiency enhances the reliability of supply by minimizing the risk of production delays caused by equipment bottlenecks or solvent shortages, ensuring a steady flow of high-purity intermediates to downstream customers. The robustness of the process also means that raw material variability can be better tolerated, providing a buffer against market fluctuations in the price of specialty chemicals.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the drastic simplification of the purification stage, which removes the need for energy-intensive distillation and large-volume solvent extraction. By utilizing acetic acid and water, which are commodity chemicals with low procurement costs, the variable cost per kilogram of the final product is significantly lowered compared to processes relying on expensive or regulated solvents. Additionally, the high yield and purity reduce the need for reprocessing or blending off-spec material, maximizing the return on raw material investment. The absence of heavy metal catalysts in many embodiments further reduces the cost associated with metal scavenging and residual analysis, streamlining the quality control budget.
- Enhanced Supply Chain Reliability: From a logistics perspective, the use of non-volatile, non-flammable aqueous solvent systems improves the safety profile of the manufacturing facility, reducing insurance premiums and regulatory compliance burdens. The ability to produce high-purity material in fewer steps shortens the manufacturing cycle time, allowing for faster response to customer demand spikes and tighter delivery windows. This agility is crucial in the pharmaceutical sector, where supply continuity is often linked to regulatory filings and patient access. Moreover, the scalability of the reaction ensures that production volumes can be increased from pilot scale to multi-ton commercial batches without fundamental changes to the process chemistry, securing long-term supply stability.
- Scalability and Environmental Compliance: The environmental footprint of this manufacturing route is markedly smaller than traditional methods, aligning with global sustainability goals and increasingly strict environmental regulations. The replacement of hydrocarbon solvents with biodegradable organic acids and water minimizes VOC emissions and wastewater toxicity, facilitating easier permitting and community relations. The high atom economy of the acylation reaction, combined with the efficient recovery of solvents through simple crystallization, supports a circular economy approach within the plant. This green chemistry profile not only mitigates regulatory risk but also enhances the brand value of the end-product for eco-conscious customers in the nutraceutical and personal care industries.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this hydroquinone diester synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this process into existing production lines or for procurement specialists assessing the quality credentials of potential suppliers. The answers reflect the consensus on best practices for catalyst selection, solvent management, and impurity control as established by the inventors.
Q: How does this patent improve purity compared to conventional hexane recrystallization?
A: The patented method utilizes a mixed solvent of organic carboxylic acid and water for crystallization, which effectively separates the target hydroquinone diester from catechol diester impurities, achieving purity levels of 99.9% or higher, whereas conventional hexane methods often result in broad melting points and lower purity due to poor solubility differentiation.
Q: What types of catalysts are suitable for this acylation reaction?
A: The process supports a wide range of acid catalysts including protonic acids like sulfuric acid and tosic acid, as well as Lewis acids such as BF3·OEt2 and AlCl3, allowing manufacturers to select based on cost, availability, and downstream processing requirements.
Q: Can this process be scaled for industrial vitamin E production?
A: Yes, the method is designed for industrial scalability by eliminating complex extraction steps and using common solvents like acetic acid and water, making it highly suitable for the large-scale manufacturing of trimethyl hydroquinone derivatives used in Vitamin E synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trimethyl Hydroquinone Diacetate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of deploying advanced synthetic routes like the one described in CN1219531A to meet the evolving demands of the global pharmaceutical and fine chemical markets. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a GMP-compliant manufacturing environment. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify that every batch of Trimethyl Hydroquinone Diacetate meets the highest standards of quality and consistency. Our commitment to process excellence means that we can deliver this complex intermediate with the reliability and transparency that top-tier multinational corporations require for their supply chains.
We invite you to engage with our technical procurement team to discuss how our capabilities align with your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages of switching to our optimized manufacturing process. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to validate our performance metrics against your internal benchmarks. Let us collaborate to drive efficiency and innovation in your supply chain, ensuring that your access to high-purity hydroquinone derivatives remains uninterrupted and cost-effective.
