Advanced Synthetic Routes for 4-Fluoro-6-Trifluoromethyl-3-Picolinic Acid Intermediates
Introduction to Next-Generation Fluorinated Pyridine Intermediates
The landscape of fine chemical synthesis is constantly evolving, driven by the demand for highly functionalized heterocyclic scaffolds that serve as the backbone for modern agrochemicals and pharmaceuticals. Patent CN107778227B introduces a significant advancement in this domain by disclosing a novel synthetic pathway for 4-fluoro-6-trifluoromethyl-3-picolinate intermediates, specifically designated as Compound V. This technology addresses a critical gap in the prior art, where the direct introduction of fluorine at the 4-position of the 6-trifluoromethyl-3-picolinic acid framework was either non-existent or plagued by inefficiencies. The strategic placement of fluorine atoms within a molecular architecture is not merely a structural modification; it fundamentally alters the electron density distribution, acidity, and lipophilicity of the compound, thereby enhancing its biological activity and environmental profile. For R&D directors and procurement specialists alike, understanding the nuances of this patented route is essential for securing a reliable supply chain of high-purity agrochemical intermediates.
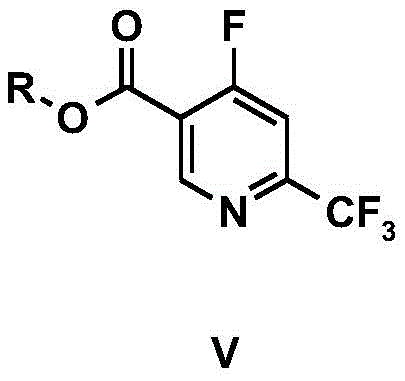
The core innovation lies in the versatility of the ester group (R), which can be tailored as n-propyl, isopropyl, n-butyl, tert-butyl, cyclopentyl, cyclohexyl, or benzyl. This modularity allows manufacturers to optimize the physical properties of the intermediate for downstream processing, such as crystallization or solubility in specific reaction media. By establishing a robust method to access these fluorinated esters, the patent provides a foundational technology that supports the commercial scale-up of complex pharmaceutical intermediates, ensuring that production capacities can meet the rigorous demands of global markets without compromising on quality or consistency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of fluorinated pyridine derivatives has been fraught with challenges, particularly when attempting to introduce fluorine into electron-deficient rings that already bear strong electron-withdrawing groups like trifluoromethyl. Conventional approaches often rely on harsh fluorinating agents that pose significant safety hazards and require specialized equipment, leading to escalated capital expenditures. Furthermore, traditional routes may involve multiple protection and deprotection steps, which inherently lower the overall atom economy and generate substantial chemical waste. In many existing processes, the selectivity for the 4-position is poor, resulting in difficult-to-separate isomeric impurities that compromise the purity specifications required for active pharmaceutical ingredients (APIs). These inefficiencies translate directly into higher production costs and extended lead times, creating bottlenecks for supply chain heads who are tasked with maintaining continuous manufacturing flows.
The Novel Approach
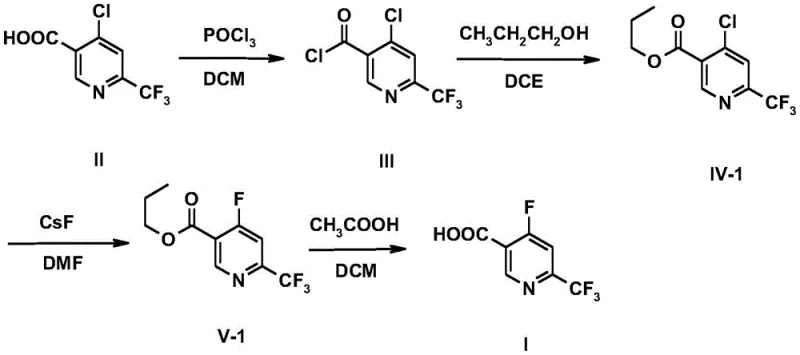
In stark contrast, the methodology outlined in CN107778227B employs a logical and step-wise progression that maximizes yield while minimizing operational complexity. The process begins with the activation of 4-chloro-6-trifluoromethyl-3-picolinic acid (Compound II), a readily accessible starting material, followed by esterification and a final nucleophilic aromatic substitution. This sequence effectively bypasses the need for direct fluorination of the acid, which can be problematic due to decarboxylation risks under aggressive conditions. Instead, by converting the acid to an ester first, the molecule becomes more amenable to the subsequent substitution reaction. The use of diverse alcohol partners allows for the fine-tuning of the intermediate's steric and electronic properties, facilitating easier purification. This novel approach not only simplifies the workflow but also enhances the reproducibility of the synthesis, making it ideally suited for mass production environments where batch-to-batch consistency is paramount.
Mechanistic Insights into Nucleophilic Aromatic Substitution and Esterification
The chemical elegance of this synthesis is rooted in the precise manipulation of reactivity at the pyridine ring. The initial step involves the conversion of the carboxylic acid moiety in Compound II to an acid chloride (Compound III) using chlorinating agents such as phosphorus oxychloride (POCl3), thionyl chloride (SOCl2), or oxalyl chloride. This activation is critical as it renders the carbonyl carbon highly electrophilic, priming it for the subsequent nucleophilic attack by an alcohol. The reaction conditions are carefully controlled, typically initiating at 0°C to manage exotherms before warming to room temperature, ensuring that the sensitive trifluoromethyl group remains intact while the acid chloride forms quantitatively. This attention to thermal management is a key factor in preventing side reactions that could degrade the product quality.
Following esterification to form Compound IV, the pivotal transformation occurs: the displacement of the 4-chloro atom with a fluorine atom to yield Compound V. This nucleophilic aromatic substitution (SnAr) is driven by the strong electron-withdrawing nature of both the pyridine nitrogen and the 6-trifluoromethyl group, which activate the 4-position towards nucleophilic attack. The choice of fluoride source is versatile, ranging from inorganic salts like cesium fluoride (CsF) and potassium fluoride (KF) to organic salts like tetramethylammonium fluoride. The reaction is conducted in polar aprotic solvents such as DMF, NMP, or DMSO at elevated temperatures (50°C to 120°C), which solvate the cation and enhance the nucleophilicity of the fluoride anion. This mechanistic understanding allows chemists to optimize the reaction kinetics, ensuring high conversion rates and minimizing the formation of defluorinated byproducts, thus securing a high-purity pharmaceutical intermediate suitable for stringent regulatory standards.
How to Synthesize 4-Fluoro-6-Trifluoromethyl-3-Picolinate Efficiently
Implementing this synthesis in a production setting requires a clear understanding of the operational parameters defined in the patent examples. The process is designed to be scalable, utilizing common industrial solvents and reagents that are easily sourced. The initial acylation and esterification steps can often be telescoped or performed in sequence with minimal workup, reducing solvent consumption and processing time. The final fluorination step, while requiring heat, proceeds cleanly with simple aqueous workups and standard extraction techniques. For technical teams looking to adopt this route, the detailed standardized synthesis steps provided in the patent serve as a robust blueprint for process validation.
- Activate 4-chloro-6-trifluoromethyl-3-picolinic acid (Compound II) using chlorinating agents like POCl3 or SOCl2 to form the acid chloride (Compound III).
- Perform esterification by reacting Compound III with various alcohols (e.g., n-propanol, isopropanol) or alkoxides to generate the chloro-ester intermediate (Compound IV).
- Execute nucleophilic aromatic substitution using fluoride salts (e.g., CsF, KF) in polar aprotic solvents to replace the 4-chloro group with fluorine, yielding Compound V.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers compelling advantages that resonate deeply with procurement managers and supply chain strategists. The primary benefit lies in the significant cost reduction in pharmaceutical manufacturing achieved through process simplification. By eliminating the need for exotic catalysts or multi-step protection strategies, the overall cost of goods sold (COGS) is drastically lowered. The use of commodity chemicals like alcohols and inorganic fluoride salts ensures that raw material costs remain stable and predictable, shielding the supply chain from the volatility often associated with specialized reagents. Furthermore, the high yields reported in the patent examples indicate a material-efficient process, meaning less raw material is wasted per kilogram of finished product, directly contributing to margin improvement.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts is a major financial driver. Traditional cross-coupling methods often require expensive palladium or nickel catalysts, along with costly ligands and extensive purification steps to remove trace metals to ppm levels. This metal-free approach removes those expenses entirely. Additionally, the ability to use various alcohols allows manufacturers to select the most cost-effective esterifying agent based on current market prices, providing flexibility in cost management. The simplified workup procedures, often involving basic aqueous washes and crystallization, reduce the load on utility systems and labor hours, further driving down operational expenditures.
- Enhanced Supply Chain Reliability: Supply continuity is critical for maintaining production schedules in the fine chemical industry. This route relies on Compound II, a stable and commercially available precursor, reducing the risk of bottlenecks associated with custom-synthesized starting materials. The robustness of the reaction conditions—tolerating a range of temperatures and solvents—means that production is less susceptible to minor fluctuations in process control, ensuring consistent output. By diversifying the acceptable fluoride sources (e.g., switching between CsF and KF based on availability), procurement teams can mitigate the risk of single-source supplier dependency, thereby enhancing the resilience of the supply chain against global disruptions.
- Scalability and Environmental Compliance: As regulatory pressures regarding waste disposal intensify, the environmental profile of a synthesis route becomes a key selection criterion. This method generates fewer hazardous byproducts compared to direct fluorination techniques that might utilize dangerous gases like elemental fluorine. The solvents used, such as DCM and alcohols, are well-understood and can be efficiently recovered and recycled in modern facilities. The high atom economy of the substitution step means less chemical waste is generated per unit of product. This alignment with green chemistry principles not only simplifies compliance with environmental regulations but also positions the manufacturer as a sustainable partner, a growing requirement for multinational corporations auditing their supplier base.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and claims within CN107778227B, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this intermediate into their existing product portfolios.
Q: What are the key advantages of this synthetic route for 4-fluoro-6-trifluoromethyl-3-picolinic acid?
A: The patented method offers a short reaction sequence with high operational simplicity. It utilizes readily available starting materials and avoids complex transition metal catalysts, significantly reducing purification burdens and heavy metal residue risks.
Q: Which fluoride sources are compatible with this nucleophilic substitution process?
A: The process demonstrates broad compatibility with various fluoride sources including cesium fluoride, potassium fluoride, sodium fluoride, pyridine hydrofluoride, and quaternary ammonium fluorides like tetramethylammonium fluoride, allowing for flexible cost optimization.
Q: How does this intermediate contribute to agrochemical and pharmaceutical development?
A: The introduction of fluorine atoms enhances lipophilicity and metabolic stability. This specific scaffold serves as a critical building block for next-generation herbicides and pharmaceuticals requiring precise electronic modulation of the pyridine ring.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Fluoro-6-Trifluoromethyl-3-Picolinate Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of high-quality intermediates in the development of next-generation crop protection agents and therapeutic drugs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with precision and speed. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our expertise in handling fluorinated compounds ensures that safety and quality are never compromised, providing you with a secure foundation for your downstream synthesis.
We invite you to collaborate with us to leverage this advanced synthetic technology for your specific applications. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your project needs, demonstrating how our optimized processes can enhance your bottom line. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate why we are the preferred partner for reliable agrochemical intermediate supplier solutions in the global market.
