Advanced Catalytic Protection of Shikimic Acid Derivatives for Scalable Pharmaceutical Manufacturing
Introduction to Next-Generation Shikimic Acid Derivative Synthesis
The pharmaceutical industry continuously seeks robust and sustainable pathways for synthesizing complex chiral intermediates, particularly those derived from natural products like shikimic acid. Patent CN113336738A introduces a transformative methodology for the preparation of shikimic acid ether compounds, addressing critical bottlenecks in traditional synthetic routes. This innovation leverages the unique properties of solid-state cation exchange resins to catalyze the protection of hydroxyl groups on the shikimate scaffold, replacing conventional liquid acid catalysts that have long plagued manufacturers with issues of corrosion and difficult separation. By shifting from homogeneous to heterogeneous catalysis, this technology not only enhances the purity profile of the resulting intermediates but also aligns with modern green chemistry principles essential for a reliable pharmaceutical intermediate supplier. The ability to recycle the catalyst directly within the reaction vessel represents a paradigm shift in process economics, offering a compelling value proposition for procurement teams focused on long-term cost stability.
Furthermore, the versatility of this approach allows for the modulation of steric bulk around the protected diol system by varying the ketone source, ranging from simple acetone to more bulky ketones like 3-pentanone. This structural flexibility is crucial for downstream transformations where specific protecting group lability or steric shielding is required to control stereoselectivity in subsequent steps. For R&D directors evaluating new routes for antiviral drug precursors, such as those leading to neuraminidase inhibitors, this method provides a high-fidelity entry point into the shikimate pathway with minimized impurity generation. The integration of this technology into existing manufacturing frameworks promises to streamline operations, reduce solvent usage associated with extensive workups, and ultimately deliver a higher quality starting material for complex multistep syntheses.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of shikimic acid ether compounds has relied heavily on the use of strong organic acids, such as p-toluenesulfonic acid (PTSA), or inorganic acids as homogeneous catalysts. While effective in driving the ketalization reaction, these liquid acids introduce severe operational challenges that impact both the economic and environmental footprint of the process. The primary drawback lies in the difficulty of catalyst recovery; once the reaction is complete, the acid remains dissolved in the reaction mixture, necessitating complex neutralization steps involving bases, followed by extensive aqueous workups and extractions to remove the resulting salts. This not only increases the volume of wastewater generated but also leads to significant product loss during phase separations, thereby reducing the overall yield and increasing the cost of goods sold.
Additionally, the corrosive nature of strong liquid acids poses a significant threat to standard stainless steel reactor vessels, often requiring the use of expensive glass-lined equipment or specialized alloys to prevent degradation and metal leaching into the product stream. Metal contamination is a critical quality attribute in pharmaceutical manufacturing, and removing trace metals to meet stringent regulatory specifications adds further purification steps and cost. Moreover, the inability to reuse the catalyst means that every batch requires a fresh charge of acid, creating a recurring raw material cost that accumulates significantly over large-scale production campaigns. These factors combined create a fragile supply chain vulnerable to fluctuations in raw material prices and waste disposal regulations, making conventional methods less attractive for modern, sustainability-focused manufacturing.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes solid strong acid cation exchange resins as heterogeneous catalysts, fundamentally altering the reaction dynamics and workup procedure. As illustrated in the reaction schemes below, the shikimate ester reacts with ketones such as acetone or 3-pentanone in the presence of the resin to form the corresponding protected ether derivatives with exceptional efficiency.
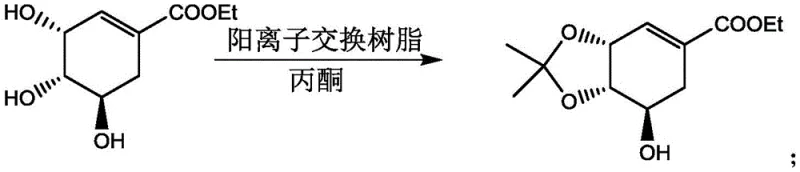
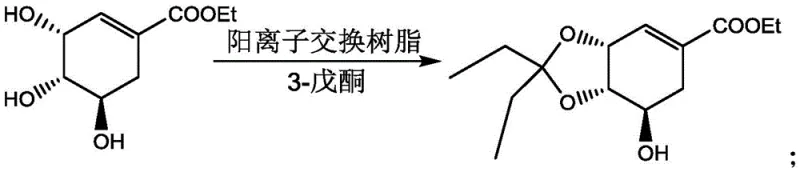
This heterogeneous system allows the catalyst to function as a fixed bed or a suspendable solid that can be effortlessly separated from the reaction mixture via simple filtration upon completion. The elimination of neutralization steps drastically simplifies the downstream processing, reducing solvent consumption and minimizing the generation of saline waste streams. Furthermore, the solid resin exhibits remarkable stability and can be regenerated and reused for multiple cycles without significant loss of catalytic activity, effectively amortizing the catalyst cost over tons of product. This approach not only mitigates equipment corrosion risks, extending the lifespan of standard reactors, but also ensures a cleaner product profile with lower levels of inorganic impurities, thereby facilitating easier compliance with rigorous pharmaceutical quality standards.
Mechanistic Insights into Cation Exchange Resin Catalyzed Ketalization
The core of this technological advancement lies in the mechanism of acid-catalyzed acetal formation facilitated by the sulfonic acid functional groups anchored on the polymeric matrix of the cation exchange resin. These immobilized proton sources activate the carbonyl oxygen of the ketone reactant, increasing its electrophilicity and promoting nucleophilic attack by the vicinal hydroxyl groups on the shikimate ring. The polymeric support provides a unique microenvironment that can enhance local concentration effects and potentially stabilize transition states through hydrogen bonding interactions within the resin pores. Unlike free protons in solution which are highly mobile and non-selective, the protons on the resin surface offer a controlled acidic environment that drives the equilibrium towards the acetal product while minimizing side reactions such as elimination or polymerization that are common with stronger liquid acids.
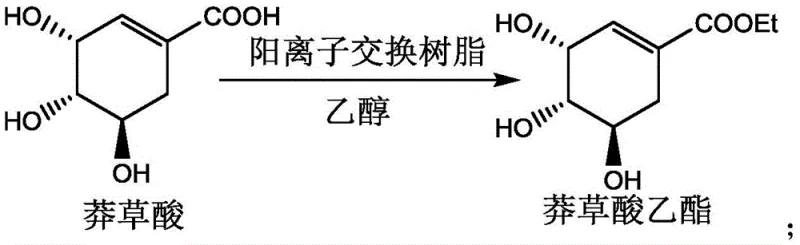
Moreover, the patent describes a comprehensive synthetic ecosystem where the shikimate starting material itself can be prepared via a similar resin-catalyzed esterification of shikimic acid with ethanol, as shown in the figure below, ensuring consistency in catalyst technology throughout the upstream process.
Following the etherification, the protected intermediate serves as a robust platform for further functionalization, such as the selective sulfonation of the remaining free hydroxyl group. The subsequent transformation into shikimic acid sulfonates, depicted in the following scheme, utilizes anion exchange resins as acid scavengers, maintaining the theme of heterogeneous reagents to simplify purification and enhance process safety.

This dual-resin strategy (cation for acid catalysis, anion for acid binding) creates a closed-loop system where solid reagents are easily removed by filtration, leaving the product in solution with minimal contamination. For R&D teams, understanding this mechanistic nuance is vital for optimizing reaction parameters such as temperature and residence time to maximize the selectivity for the desired diastereomer and minimize the formation of regioisomers. The steric bulk of the ketone, whether it is the smaller acetone or the larger 3-pentanone, influences the conformational preference of the resulting dioxolane ring, which can be exploited to direct the stereochemical outcome of subsequent nucleophilic substitutions on the cyclohexene ring.
How to Synthesize Shikimic Acid Ether Efficiently
Implementing this synthesis route requires careful attention to reactor configuration and parameter control to fully leverage the benefits of the heterogeneous catalyst. The process is designed to be adaptable to both batch and continuous flow modes, with the patent specifically highlighting the use of an esterification dehydration tower where the resin acts as a packing material. In this setup, reactants are introduced from opposite ends of the column, maximizing contact time and mass transfer efficiency while allowing for the continuous removal of water, which drives the equilibrium towards the ether product. Operators must maintain precise temperature control within the 40°C to 70°C range to ensure optimal reaction kinetics without degrading the thermally sensitive shikimate scaffold. Detailed standardized operating procedures regarding resin pre-treatment, loading densities, and regeneration protocols are essential for consistent batch-to-batch reproducibility.
- Prepare the reaction system by loading a strong acid cation exchange resin into an esterification dehydration tower to serve as the fixed-bed catalyst filler.
- Introduce the shikimate compound (e.g., shikimate ethyl ester) from the top of the tower and the ketone compound (e.g., acetone or 3-pentanone) from the bottom.
- Maintain the reaction temperature between 40°C and 70°C for 1 to 5 hours to achieve high-yield conversion to the protected shikimic acid ether.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this resin-catalyzed technology translates into tangible strategic advantages that extend beyond simple yield improvements. The shift to a recyclable catalyst system fundamentally alters the cost structure of the manufacturing process by eliminating the recurring expense of stoichiometric or near-stoichiometric amounts of homogeneous acid catalysts. Instead of purchasing fresh acid for every batch, the facility invests in a fixed quantity of resin that serves as a durable asset, depreciating over a much longer timeline. This reduction in consumable costs directly improves the gross margin of the final API intermediate, providing a competitive edge in price-sensitive markets. Additionally, the simplification of the workup procedure reduces the demand for auxiliary chemicals such as neutralizing bases and extraction solvents, further compounding the cost savings.
- Cost Reduction in Manufacturing: The implementation of reusable cation exchange resins eliminates the need for expensive and hazardous liquid acid catalysts, leading to substantial cost savings in raw material procurement. By avoiding complex neutralization and salt-removal steps, the process significantly reduces the consumption of water and organic solvents, lowering utility bills and waste disposal fees. The extended lifespan of the reactor equipment due to reduced corrosion also decreases capital expenditure on maintenance and replacement, contributing to a lower total cost of ownership for the production line.
- Enhanced Supply Chain Reliability: Relying on solid catalysts that can be stocked and reused mitigates the risk of supply disruptions associated with specialized liquid reagents. The robustness of the process allows for flexible production scheduling, as the catalyst does not require immediate replenishment after every run, ensuring continuous operation even during periods of raw material scarcity. Furthermore, the simplified purification train reduces the number of unit operations, decreasing the likelihood of mechanical failures or bottlenecks that could delay order fulfillment and impact delivery timelines to downstream pharmaceutical partners.
- Scalability and Environmental Compliance: The heterogeneous nature of the reaction facilitates seamless scale-up from pilot plant to commercial tonnage without the engineering challenges associated with heat management in highly exothermic liquid acid reactions. The drastic reduction in saline wastewater generation aligns with increasingly stringent environmental regulations, reducing the regulatory burden and permitting risks for manufacturing sites. This green chemistry profile enhances the corporate sustainability image, making the supply chain more resilient to future carbon taxes or environmental compliance costs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel shikimic acid ether synthesis technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this route into their existing portfolio of chemical processes.
Q: What are the advantages of using cation exchange resin over traditional organic acids?
A: Unlike traditional homogeneous catalysts like p-toluenesulfonic acid, cation exchange resins are heterogeneous solids that can be easily filtered and reused multiple times, significantly reducing catalyst consumption and waste treatment costs while minimizing equipment corrosion.
Q: Can this method be scaled for industrial production of antiviral intermediates?
A: Yes, the process utilizes continuous flow or fixed-bed reactor configurations (esterification dehydration towers), which are inherently scalable and suitable for large-scale commercial manufacturing of complex pharmaceutical intermediates.
Q: What specific ketone compounds are compatible with this protection strategy?
A: The patent demonstrates compatibility with various ketones including acetone for binary ether formation and 3-pentanone for ternary ether formation, allowing flexibility in designing the steric environment of the protected diol system.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Shikimic Acid Ether Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global pharmaceutical supply chain. Our team of expert chemists has extensively evaluated the resin-catalyzed pathways described in recent patents and possesses the technical capability to adapt these methodologies for your specific project needs. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale discovery to industrial manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of shikimic acid derivative we supply meets the highest standards of quality and consistency required for GMP environments.
We invite you to collaborate with us to explore how this advanced catalytic technology can optimize your production costs and enhance your supply chain security. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your current manufacturing setup, demonstrating exactly where efficiencies can be gained. Please contact our technical procurement team today to request specific COA data for our shikimic acid intermediates and to discuss detailed route feasibility assessments for your upcoming projects. Let us be your partner in delivering innovative chemical solutions that drive your business forward.
