Advanced Continuous Synthesis of Chloroquine Side Chain Intermediates Using Supported Nano-Nickel Catalysts
The global demand for antimalarial and antiviral therapeutics has placed unprecedented pressure on the supply chains of critical active pharmaceutical ingredients (APIs), particularly Chloroquine Phosphate. At the heart of this supply chain lies the efficient production of its key side chain, N,N-diethyl-1,4-pentanediamine. Traditional manufacturing methods have long struggled with safety hazards, environmental burdens, and batch-to-batch inconsistency. However, a significant technological breakthrough detailed in patent CN111482178A introduces a novel supported nano-nickel-based catalyst system designed specifically for the continuous reductive amination of 5-diethylamino-2-pentanone. This innovation represents a paradigm shift from hazardous batch processing to streamlined, continuous flow chemistry, offering a robust solution for reliable pharmaceutical intermediate supplier networks seeking to modernize their production capabilities. By leveraging non-precious metal catalysis in a fixed-bed configuration, this technology addresses the twin challenges of cost efficiency and operational safety that have historically plagued the synthesis of this vital chloroquine precursor.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of N,N-diethyl-1,4-pentanediamine has relied heavily on batch-wise reductive amination using Raney nickel catalysts under high-pressure liquid ammonia conditions. This legacy approach is fraught with significant operational deficiencies that hinder modern cost reduction in API manufacturing. The use of Raney nickel, a pyrophoric material derived from nickel-aluminum alloys, necessitates rigorous safety protocols to prevent spontaneous combustion, while the dissolution of aluminum during catalyst preparation generates substantial quantities of alkaline waste containing heavy metals. Furthermore, the batch autoclave process requires the use of anhydrous liquid ammonia, a toxic and volatile reagent that poses severe storage and handling risks. Perhaps most critically for supply chain efficiency, the slurry nature of the reaction mandates complex downstream processing, including filtration and centrifugation, to separate the fine catalyst powder from the product stream, leading to inevitable catalyst loss, lower overall yields typically around 88%, and extended production cycles that limit scalability.
The Novel Approach
In stark contrast, the technology disclosed in patent CN111482178A utilizes a heterogeneous supported nano-nickel catalyst within a continuous fixed-bed reactor system, fundamentally altering the process economics and safety profile. This advanced methodology replaces the hazardous liquid ammonia with safer aqueous ammonia or urea sources and eliminates the need for post-reaction filtration since the catalyst remains stationary within the reactor column. The transition to a continuous flow regime allows for precise control over reaction parameters such as temperature (80-140°C) and pressure (0.1-1.0 MPa), resulting in significantly higher conversion rates exceeding 99% and selectivity towards the target diamine. By removing the aluminum component entirely and utilizing stable oxide supports like alumina or zirconia, the process drastically reduces environmental pollution and simplifies waste treatment. This shift not only enhances the commercial scale-up of complex pharmaceutical intermediates but also ensures a consistent, high-purity output suitable for stringent regulatory standards without the variability inherent in batch processing.
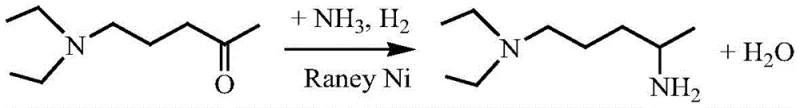
Mechanistic Insights into Supported Nano-Nickel Catalytic Reductive Amination
The core of this technological advancement lies in the sophisticated design of the supported nano-nickel catalyst, which integrates a primary active nickel component with specific promoters such as Cobalt, Lanthanum, or Cerium on stable oxide carriers. The preparation involves a co-precipitation or deposition-precipitation method that ensures the metallic nickel crystallites maintain a nano-scale dimension, typically below 8nm, which is critical for maximizing the active surface area available for hydrogenation. The inclusion of promoter metals plays a synergistic role in modifying the electronic state of the nickel sites, thereby enhancing the adsorption of the ketone substrate and facilitating the cleavage of hydrogen molecules. This structural optimization prevents the sintering of nickel particles under reaction conditions, a common failure mode in traditional catalysts, thus ensuring prolonged stability. The mechanism proceeds through the formation of an imine intermediate upon the reaction of the ketone with the nitrogen source, followed by rapid hydrogenation on the nickel surface to yield the secondary amine, with the solid support providing the necessary thermal stability to withstand continuous operation at elevated temperatures.
From an impurity control perspective, the fixed-bed continuous flow architecture offers superior selectivity management compared to batch systems. In traditional slurry reactors, local hot spots and concentration gradients often promote side reactions, such as the over-reduction of the ketone to the corresponding alcohol (5-diethylamino-2-pentanol) or the formation of tertiary amines. The plug-flow characteristics of the fixed-bed reactor ensure a uniform residence time distribution, minimizing the exposure of the product to reactive conditions that could lead to degradation. Furthermore, the specific acidity and basicity of the catalyst support can be tuned to favor the dehydration step of the hemiaminal intermediate, driving the equilibrium towards the desired amine product. This precise control results in product purities consistently above 99%, significantly reducing the burden on downstream purification units like distillation columns and ensuring that the final high-purity chloroquine side chain meets the rigorous specifications required for pharmaceutical grade applications without extensive reprocessing.
How to Synthesize N,N-Diethyl-1,4-Pentanediamine Efficiently
The implementation of this continuous synthesis route requires a systematic approach to catalyst activation and reactor engineering to fully realize the benefits of the nano-nickel system. Operators must first ensure the catalyst is properly reduced in a hydrogen atmosphere to generate the active metallic phase before introducing the organic feed stream. The process involves pumping a homogeneous mixture of the ketone substrate, an alcohol solvent to enhance miscibility, and the nitrogen source through the heated catalyst bed. Maintaining the correct hydrogen-to-substrate molar ratio is crucial to prevent catalyst coking and ensure complete conversion. While the general principles are straightforward, the specific operational parameters regarding temperature gradients and flow rates are critical for long-term stability. For a detailed breakdown of the standardized synthesis protocol, please refer to the technical guide below.
- Prepare the supported nano-nickel catalyst via co-precipitation, ensuring nickel grain sizes remain below 8nm for optimal activity.
- Load the activated catalyst into a fixed-bed reactor and establish a reducing atmosphere with hydrogen flow.
- Continuously pump a mixed solution of 5-diethylamino-2-pentanone, ammonia source, and alcohol solvent through the reactor at 80-140°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this supported nano-nickel technology translates into tangible strategic advantages that extend far beyond simple yield improvements. The elimination of pyrophoric Raney nickel and hazardous liquid ammonia significantly lowers the insurance and safety compliance costs associated with the facility, while the removal of filtration and centrifugation steps reduces capital expenditure on downstream processing equipment. The continuous nature of the process allows for a smaller physical footprint to achieve the same production volume as a large batch plant, optimizing facility utilization rates. Moreover, the stability of the catalyst over hundreds of hours of operation ensures predictable production schedules, reducing lead time for high-purity pharmaceutical intermediates and mitigating the risk of supply disruptions caused by frequent catalyst changeovers or batch failures. This reliability is essential for maintaining the continuity of supply for critical antimalarial drugs in the global market.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of expensive and hazardous reagents with cheaper, safer alternatives and the drastic simplification of the unit operations. By utilizing a fixed-bed reactor, the energy-intensive steps of heating and cooling large batch vessels repeatedly are replaced by a steady-state thermal profile, leading to substantial energy savings. Additionally, the high selectivity of the nano-nickel catalyst minimizes the formation of by-products, which reduces the load on purification systems and increases the overall mass efficiency of the plant. The absence of aluminum waste treatment further lowers environmental compliance costs, creating a leaner and more cost-effective manufacturing model that enhances profit margins without compromising product quality.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the robust nature of the supported catalyst, which demonstrates excellent resistance to deactivation and mechanical attrition. Unlike batch processes that are susceptible to variability between runs, the continuous flow system provides a consistent output quality, simplifying inventory management and quality assurance protocols. The ability to run the reactor for extended periods without shutdown means that production targets can be met with greater certainty, allowing suppliers to honor long-term contracts with confidence. This stability is particularly valuable in the pharmaceutical sector, where regulatory audits require demonstrable control over the manufacturing process and consistent product specifications over time.
- Scalability and Environmental Compliance: Scaling this technology from pilot to commercial production is inherently simpler due to the modular nature of fixed-bed reactors, avoiding the complex mixing and heat transfer issues often encountered when scaling up batch autoclaves. The process aligns perfectly with green chemistry principles by eliminating heavy metal waste streams and reducing solvent usage through higher concentration feeds. The use of aqueous ammonia instead of anhydrous ammonia removes the need for specialized high-pressure storage infrastructure, making the technology accessible to a wider range of manufacturing sites. This environmental friendliness not only future-proofs the production facility against tightening regulations but also enhances the corporate sustainability profile of the manufacturer.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this continuous reductive amination technology. These insights are derived directly from the experimental data and process descriptions found in the underlying patent literature, providing a factual basis for evaluating the feasibility of adopting this method. Understanding these details is crucial for technical teams assessing the integration of this catalyst system into existing production lines or new facility designs.
Q: How does the supported nano-nickel catalyst improve safety compared to Raney nickel?
A: Unlike pyrophoric Raney nickel which requires careful handling and generates aluminum waste, the supported nano-nickel catalyst is stable, non-pyrophoric, and allows for the use of safer aqueous ammonia instead of hazardous liquid ammonia.
Q: What is the expected catalyst lifetime in continuous operation?
A: Patent data indicates that the optimized catalyst maintains stable performance with yields around 95% for over 500 hours of continuous operation without significant deactivation.
Q: Does this process require downstream filtration steps?
A: No, because the catalyst is fixed in the bed reactor, the product stream exits free of catalyst particles, eliminating the need for complex filtration or centrifugation steps required in batch slurry processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N,N-Diethyl-1,4-Pentanediamine Supplier
As the pharmaceutical industry continues to evolve towards more sustainable and efficient manufacturing practices, the availability of advanced intermediates produced via cutting-edge technologies becomes a key differentiator. NINGBO INNO PHARMCHEM stands at the forefront of this transformation, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to deliver exceptional value to our global partners. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that ensure every batch of N,N-diethyl-1,4-pentanediamine meets the highest international standards. We understand that in the competitive landscape of API manufacturing, consistency and reliability are paramount, and our state-of-the-art facilities are designed to provide exactly that level of assurance for your critical supply chains.
We invite you to explore how our advanced catalytic capabilities can optimize your procurement strategy and reduce your overall cost of goods. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing you to make informed decisions that will strengthen your position in the market. Partner with us to secure a stable, high-quality supply of this essential chloroquine intermediate and drive your pharmaceutical projects forward with confidence.
