Advanced Manufacturing of N-Benzyl Azetidine-2-Carboxylate for Scalable Pharmaceutical Intermediates
The pharmaceutical industry continuously seeks robust and scalable pathways for synthesizing complex amino acid analogs, particularly those serving as critical building blocks for bioactive peptides. Patent CN1182112C introduces a significant advancement in this domain by detailing a highly efficient process for the production of N-benzyl azetidine-2-carboxylate derivatives. These compounds are pivotal intermediates in the synthesis of Azetidine-2-carboxylic acid (AzeOH), a well-known proline analog used extensively in the construction of high molecular weight polypeptides. The disclosed methodology overcomes historical limitations associated with natural source extraction and earlier synthetic attempts, offering a route that is both chemically elegant and commercially viable for large-scale manufacturing operations.
Traditionally, the synthesis of azetidine rings has been fraught with challenges regarding yield optimization and reaction control. Prior art methods, such as those described by Fowden or Duplan, often relied on the use of strong inorganic bases like barium hydroxide or sodium hydroxide to effect cyclization of 4-amino-2-halobutanoic acids. These classical approaches frequently suffered from harsh reaction conditions that could degrade sensitive functional groups or lead to complex mixtures of by-products. Furthermore, alternative routes involving 2,4-dibromobutyrate derivatives, while effective, often necessitated prolonged reflux periods extending up to 24 hours, which is energetically inefficient and detrimental to throughput in a commercial setting. The reliance on such demanding parameters historically constrained the economic feasibility of producing high-purity azetidine intermediates.
In stark contrast, the novel approach detailed in the patent leverages a specific halogenated precursor, 2-bromo-4-chlorobutyrate, which exhibits superior reactivity profiles compared to its di-halogenated counterparts. By reacting this specific ester with benzylamine, the process achieves cyclization with unexpectedly high efficiency and yield. The strategic placement of the bromine and chlorine atoms facilitates a smoother nucleophilic substitution and subsequent ring closure, allowing the reaction to proceed under milder thermal conditions. This innovation not only accelerates the reaction kinetics but also simplifies the downstream purification processes, resulting in a final product with a markedly improved chemical purity profile that is essential for pharmaceutical grade applications.
Mechanistic Insights into Halobutyrate Cyclization
The core of this synthetic breakthrough lies in the precise mechanistic pathway governing the formation of the four-membered azetidine ring. The reaction initiates with the nucleophilic attack of the benzylamine nitrogen on the carbon bearing the bromine atom of the 2-bromo-4-chlorobutyrate ester. This step forms an acyclic amino-ester intermediate, which then undergoes an intramolecular nucleophilic substitution where the secondary amine displaces the chloride ion at the 4-position. This cyclization is critically dependent on the leaving group ability of the halogens and the conformational flexibility of the carbon chain. The use of an iodide source, such as sodium iodide, acts as a potent catalyst in this transformation, likely facilitating the displacement of the chloride through a Finkelstein-type mechanism or by enhancing the electrophilicity of the carbon center, thereby lowering the activation energy required for ring closure.
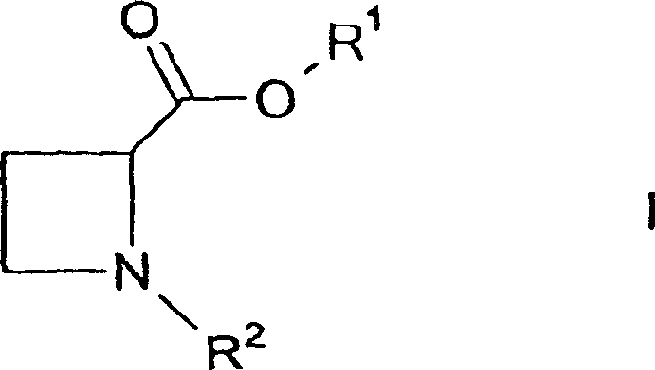
Controlling the impurity profile during this cyclization is paramount for ensuring the quality of the final API intermediate. The patent highlights that the specific choice of solvent, particularly polar aprotic solvents like acetonitrile, plays a crucial role in stabilizing the transition states and solubilizing the inorganic salts generated during the reaction. Furthermore, the careful regulation of temperature, typically ranging from 50°C to reflux, ensures that the cyclization proceeds to completion without promoting side reactions such as elimination or polymerization. The ability to conduct this transformation in a telescoped manner, where the amination and cyclization occur sequentially without isolating the intermediate, significantly reduces the exposure of reactive species to potential degradation pathways, thus maintaining a clean impurity spectrum that simplifies final crystallization and drying steps.
How to Synthesize N-Benzyl Azetidine-2-Carboxylate Efficiently
Implementing this synthesis requires a disciplined approach to reaction parameter control to maximize yield and safety. The process generally begins with the preparation of the key 2-bromo-4-chlorobutyrate ester, often derived from 4-chlorobutyryl chloride via bromination and subsequent esterification. Once the precursor is secured, it is reacted with benzylamine in the presence of a base and catalyst system. The following guide outlines the standardized operational framework derived from the patent examples, providing a clear roadmap for R&D teams looking to replicate or scale this chemistry. Detailed standard operating procedures for each unit operation are provided in the section below to ensure consistency and reproducibility across different manufacturing batches.
- Preparation of 2-bromo-4-chlorobutyrate ester by reacting 4-chlorobutyryl chloride with bromine, followed by esterification with an alcohol.
- Reaction of the resulting 2-bromo-4-chlorobutyrate ester with benzylamine in a suitable solvent like acetonitrile.
- Cyclization of the intermediate using a base such as potassium carbonate and an iodide catalyst to form the final N-benzyl azetidine ring.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented methodology offers substantial strategic advantages over legacy manufacturing routes. The primary benefit stems from the utilization of readily available and cost-effective starting materials, such as 4-chlorobutyryl chloride and benzylamine, which are commodity chemicals with stable global supply chains. By eliminating the need for exotic reagents or complex multi-step protection-deprotection sequences early in the synthesis, the overall cost of goods sold (COGS) is significantly optimized. This reduction in raw material complexity translates directly into lower purchasing costs and reduced inventory holding risks, making the final azetidine intermediate more price-competitive in the global marketplace for peptide building blocks.
- Cost Reduction in Manufacturing: The process achieves cost efficiency primarily through the elimination of expensive transition metal catalysts and the reduction of processing time. Unlike older methods that required extended reflux periods or harsh conditions necessitating specialized reactor linings, this route operates under standard glass-lined or stainless steel conditions with common solvents. The high conversion rates observed minimize the loss of valuable starting materials, and the ability to potentially skip the isolation of intermediates reduces solvent consumption and waste disposal costs. These cumulative efficiencies result in a drastically simplified production workflow that lowers the overall manufacturing expenditure without compromising on product quality.
- Enhanced Supply Chain Reliability: The robustness of this chemical route ensures a high degree of supply continuity, which is critical for long-term pharmaceutical contracts. Because the reaction tolerances are broad regarding temperature and concentration, the risk of batch failure due to minor process deviations is substantially mitigated. This reliability allows for more accurate production planning and shorter lead times for order fulfillment. Furthermore, the use of standard solvents like acetonitrile and ethyl acetate means that supply chain disruptions related to niche solvent availability are avoided, ensuring that production schedules can be maintained even during periods of market volatility.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward due to the absence of highly exothermic runaway risks or the generation of difficult-to-treat hazardous by-products. The reaction generates inorganic salts as the primary waste stream, which are easily managed through standard aqueous workup and disposal protocols. This environmental compatibility simplifies regulatory compliance and reduces the burden on wastewater treatment facilities. Consequently, the process is highly amenable to scale-up from pilot plant quantities to multi-ton annual production capacities, supporting the growing demand for azetidine-based therapeutics.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of N-benzyl azetidine-2-carboxylate. These answers are derived directly from the technical specifications and experimental data provided in the patent literature, ensuring that stakeholders receive accurate and actionable information. Understanding these nuances is essential for evaluating the feasibility of integrating this intermediate into your specific drug development pipeline or manufacturing portfolio.
Q: What distinguishes this cyclization method from prior art techniques?
A: Unlike conventional methods that often utilize harsh bases like barium hydroxide or require prolonged reflux times with dibromo-derivatives, this patented approach employs a specific 2-bromo-4-chlorobutyrate precursor. This unique halogen arrangement facilitates unexpectedly high yields and efficiency under milder conditions, significantly reducing reaction time and improving the impurity profile of the final azetidine product.
Q: Can the intermediate amine be isolated during the process?
A: Yes, the process technically allows for the isolation of the aminated intermediate (2-(benzylamino)-4-chlorobutyrate) before cyclization. However, the preferred industrial embodiment utilizes a one-pot or telescoped procedure where the amination and subsequent ring-closing cyclization occur sequentially in the same reactor, thereby minimizing handling losses and maximizing overall throughput.
Q: How does this route impact the production of L-Azetidine-2-carboxylic acid?
A: This synthetic route provides a robust racemic precursor that can be efficiently deprotected to yield Azetidine-2-carboxylic acid (AzeOH). The resulting acid serves as a critical substrate for subsequent resolution processes, such as diastereomeric salt formation with tartaric acid, enabling the production of enantiomerically pure L-AzeOH which is essential for high-value peptide synthesis applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Benzyl Azetidine-2-Carboxylate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of modern drug discovery and development programs. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistent quality. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of N-benzyl azetidine-2-carboxylate meets the exacting standards required for pharmaceutical synthesis. Our commitment to process excellence means we can deliver this complex intermediate with the reliability and transparency that global supply chains demand.
We invite you to collaborate with us to optimize your sourcing strategy for azetidine derivatives. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs, demonstrating how our manufacturing capabilities can reduce your overall development costs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us support your next breakthrough in peptide therapeutics with our superior manufacturing expertise and dedicated customer service.
