Revolutionizing Alkylbenzothiazole Synthesis: Metal-Free Visible Light Technology for Commercial Scale
The pharmaceutical and fine chemical industries are constantly seeking innovative synthetic methodologies that balance efficiency with environmental sustainability. Patent CN112979580B introduces a groundbreaking approach for the preparation of alkylbenzothiazole derivatives, a crucial class of heterocyclic compounds widely used in drug discovery and agrochemical applications. This technology leverages visible light irradiation to drive intramolecular cross-coupling reactions, eliminating the need for expensive and toxic transition metal catalysts or external photosensitizers. By utilizing simple household compact fluorescent lamps and inorganic bases like sodium phosphate, this method represents a paradigm shift towards greener chemistry. For R&D directors and procurement managers, this patent signals a potential reduction in raw material costs and a simplification of purification processes, directly impacting the bottom line of pharmaceutical intermediates manufacturing. The ability to operate at room temperature further enhances the safety profile and energy efficiency of the synthesis, making it an attractive candidate for commercial scale-up of complex heterocycles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of benzothiazole derivatives has relied heavily on transition metal catalysis, often involving precious metals like ruthenium or iridium complexes such as [Ru(bpy)3]2+ or [Ir(ppy)3]. These conventional methods, while effective, introduce significant logistical and financial burdens to the supply chain. The reliance on rare earth metals creates vulnerability to price volatility and supply disruptions, which is a critical concern for any procurement manager overseeing long-term production contracts. Furthermore, the presence of heavy metals in the reaction mixture necessitates rigorous purification steps to meet stringent pharmaceutical purity specifications, often requiring expensive scavenging resins and extensive analytical testing via ICP-MS.  These additional downstream processing steps not only increase the overall cost reduction in pharmaceutical intermediates manufacturing but also extend the production lead time, potentially delaying time-to-market for new drug candidates. The environmental footprint of disposing of metal-contaminated waste streams also poses compliance challenges for modern chemical facilities aiming for sustainability goals.
These additional downstream processing steps not only increase the overall cost reduction in pharmaceutical intermediates manufacturing but also extend the production lead time, potentially delaying time-to-market for new drug candidates. The environmental footprint of disposing of metal-contaminated waste streams also poses compliance challenges for modern chemical facilities aiming for sustainability goals.
The Novel Approach
In stark contrast, the novel approach disclosed in CN112979580B circumvents these issues by employing a metal-free photocatalytic system driven by visible light. This method utilizes N-(2-bromophenyl) alkylthioamides as substrates, which undergo cyclization under the irradiation of a standard 45W household compact fluorescent lamp. The elimination of transition metals means that the resulting alkylbenzothiazole derivatives are inherently free from heavy metal contamination, drastically simplifying the workup procedure. This technological advancement allows for a more streamlined manufacturing process where the focus shifts from complex purification to efficient reaction management. For supply chain heads, this translates to enhanced supply chain reliability as the dependency on specialized catalytic reagents is removed. The use of common inorganic bases like sodium phosphate and readily available solvents like DMSO ensures that raw material sourcing is robust and less susceptible to geopolitical or market fluctuations. This simplicity is key to reducing lead time for high-purity alkylbenzothiazole derivatives, enabling faster response to market demands.
Mechanistic Insights into Visible Light Promoted Cyclization
The core of this innovation lies in the unique mechanistic pathway that allows for bond formation without external photocatalysts. The reaction proceeds through a visible light-promoted intramolecular cross-coupling mechanism where the energy from the light source directly activates the substrate or facilitates a radical pathway in the presence of the inorganic base. 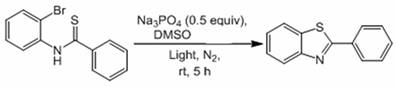 Detailed analysis of the patent data suggests that the base plays a crucial role in deprotonating the intermediate species, facilitating the nucleophilic attack or radical coupling required to close the thiazole ring. This mechanism is highly efficient, as evidenced by the high yields reported across various substrates, including those with electron-donating and electron-withdrawing groups. For R&D teams, understanding this mechanism is vital for optimizing reaction conditions and expanding the substrate scope to include more complex molecular architectures. The robustness of this mechanism under ambient conditions indicates a high tolerance for functional groups, which is essential for late-stage functionalization in drug synthesis.
Detailed analysis of the patent data suggests that the base plays a crucial role in deprotonating the intermediate species, facilitating the nucleophilic attack or radical coupling required to close the thiazole ring. This mechanism is highly efficient, as evidenced by the high yields reported across various substrates, including those with electron-donating and electron-withdrawing groups. For R&D teams, understanding this mechanism is vital for optimizing reaction conditions and expanding the substrate scope to include more complex molecular architectures. The robustness of this mechanism under ambient conditions indicates a high tolerance for functional groups, which is essential for late-stage functionalization in drug synthesis.
Impurity control is another critical aspect where this mechanism offers distinct advantages. In traditional metal-catalyzed reactions, side products often arise from metal-ligand interactions or incomplete catalyst turnover. However, in this metal-free system, the impurity profile is significantly cleaner, primarily consisting of unreacted starting materials or simple hydrolysis byproducts that are easier to separate. The absence of metal residues means that the final high-purity alkylbenzothiazole derivatives require less intensive chromatographic purification, which directly correlates to higher overall throughput and lower solvent consumption. This purity advantage is paramount for pharmaceutical applications where regulatory bodies impose strict limits on elemental impurities. By minimizing the formation of hard-to-remove byproducts, this technology ensures that the commercial scale-up of complex heterocycles can proceed with consistent quality and reduced risk of batch failure.
How to Synthesize Alkylbenzothiazole Derivatives Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting involves a straightforward protocol that aligns with standard organic synthesis practices while offering superior efficiency. The process begins with the preparation of the reaction vessel under an inert atmosphere, typically nitrogen, to prevent oxidative side reactions that could compromise yield. The substrate, N-(2-bromophenyl) alkylthioamide, is combined with the inorganic base and solvent, creating a homogeneous mixture ready for irradiation. This simplicity in operation reduces the training burden on technical staff and minimizes the potential for human error during setup. The standardized nature of the reagents means that sourcing is uncomplicated, further supporting the goal of cost reduction in pharmaceutical intermediates manufacturing.
- Prepare the reaction vessel under inert gas protection and add N-(2-bromophenyl) alkylthioamide substrate.
- Introduce inorganic base such as sodium phosphate and dimethyl sulfoxide (DMSO) solvent to the mixture.
- Irradiate the reaction with a 45W household compact fluorescent lamp at room temperature for 24 hours to yield the product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this visible light technology offers substantial strategic benefits for procurement and supply chain management. The primary advantage lies in the drastic simplification of the bill of materials. By removing the need for precious metal catalysts and specialized photosensitizers, the direct material costs are significantly reduced. This cost structure is more stable and predictable, allowing for more accurate budgeting and pricing strategies for long-term supply agreements. Additionally, the simplified purification process reduces the consumption of solvents and chromatography media, which are often significant cost drivers in fine chemical production. These factors combined contribute to a more competitive pricing model for the final alkylbenzothiazole derivatives, enhancing market positioning.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes the need for expensive metal scavenging steps and reduces the burden of analytical testing for heavy metal residues. This leads to substantial cost savings in both reagents and quality control operations. The use of household-grade light sources instead of specialized high-energy lamps further lowers capital expenditure and energy costs. These efficiencies compound over large production volumes, resulting in a significantly lower cost of goods sold. The overall process efficiency allows for better resource allocation, directing funds towards other critical areas of development and production.
- Enhanced Supply Chain Reliability: Relying on common inorganic bases and standard solvents mitigates the risk of supply chain disruptions associated with specialized reagents. The robustness of the reaction conditions ensures consistent output even with minor variations in raw material quality. This reliability is crucial for maintaining continuous production schedules and meeting delivery commitments to downstream pharmaceutical clients. The reduced complexity of the process also means that technology transfer to manufacturing sites is faster and smoother, ensuring that supply can be scaled up rapidly to meet demand spikes without compromising quality.
- Scalability and Environmental Compliance: The room temperature operation and use of visible light make this process inherently safer and easier to scale than high-temperature or high-pressure alternatives. The green chemistry profile, characterized by the absence of toxic metals and reduced waste generation, aligns with increasingly stringent environmental regulations. This compliance reduces the risk of regulatory penalties and enhances the corporate sustainability profile. The ease of scale-up ensures that the transition from laboratory to commercial production is seamless, supporting the long-term viability of the supply chain for these critical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis method, based on the detailed data provided in the patent documentation. These insights are intended to clarify the operational capabilities and strategic benefits for potential partners and stakeholders. Understanding these details is essential for making informed decisions about integrating this technology into existing production workflows.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, the patented method explicitly operates without any photosensitizer or transition metal catalyst, utilizing only visible light and inorganic base.
Q: What are the yield advantages of this visible light method?
A: The method achieves high yields, with specific examples demonstrating HPLC yields up to 99% and isolated yields ranging significantly higher than conventional metal-catalyzed routes.
Q: Is this process scalable for industrial pharmaceutical intermediate production?
A: Yes, the use of household compact fluorescent lamps and room temperature conditions suggests high scalability and reduced energy consumption compared to high-temperature or high-pressure methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alkylbenzothiazole Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the visible light synthesis method described in CN112979580B for producing high-quality alkylbenzothiazole derivatives. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch meets the highest industry standards. We understand the critical nature of pharmaceutical intermediates and are equipped to handle the specific challenges associated with heterocyclic synthesis, delivering products that support your drug development timelines.
We invite you to collaborate with us to leverage this advanced technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and quality targets. We encourage you to contact us to request specific COA data and route feasibility assessments that demonstrate how we can optimize your supply chain. By partnering with us, you gain access to a reliable alkylbenzothiazole supplier dedicated to driving efficiency and innovation in your manufacturing operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
