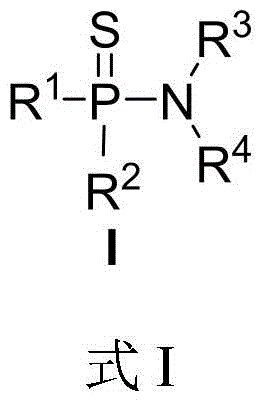
Advanced Electrochemical Route for High-Purity Thiophosphoramide Intermediates
Introduction to Patent CN116284117A
The landscape of organophosphorus chemistry is undergoing a significant transformation driven by the urgent need for greener, safer, and more efficient synthetic methodologies. Patent CN116284117A, filed in June 2023, presents a groundbreaking three-component electrochemical synthesis method for thiophosphoramide derivatives. This technology addresses critical bottlenecks in the production of high-value phosphorus-containing small molecules, which are pivotal in the fields of medicinal chemistry, agrochemicals, and materials science. By leveraging electricity as a traceless oxidant, this invention enables the direct coupling of trivalent phosphine hydrides, amines, and elemental sulfur in a single step. This approach not only circumvents the use of hazardous chlorinating agents but also streamlines the process workflow, offering a robust pathway for the commercial scale-up of complex organophosphorus compounds. For R&D directors and procurement managers alike, this patent represents a strategic opportunity to enhance supply chain resilience while adhering to increasingly stringent environmental regulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of thiophosphoramides has been plagued by significant safety and environmental challenges. The most prevalent industrial route involves the use of phosphorus trichloride sulfide (PSCl3) as a starting material. While effective, PSCl3 is extremely toxic and corrosive, posing severe risks to personnel and requiring specialized containment infrastructure that drives up capital expenditure. Furthermore, the reaction generates stoichiometric amounts of hydrochloric acid and other chloride by-products, necessitating complex neutralization and waste treatment procedures that increase the overall cost of goods sold. Alternative methods utilizing thio-reagents such as Lawesson's reagent are equally problematic due to their notorious foul odor and thermal instability, which complicate handling and storage. These traditional pathways often suffer from poor atom economy and limited functional group tolerance, restricting the chemical space accessible to medicinal chemists and forcing reliance on multi-step protection-deprotection sequences that erode overall process efficiency.
The Novel Approach
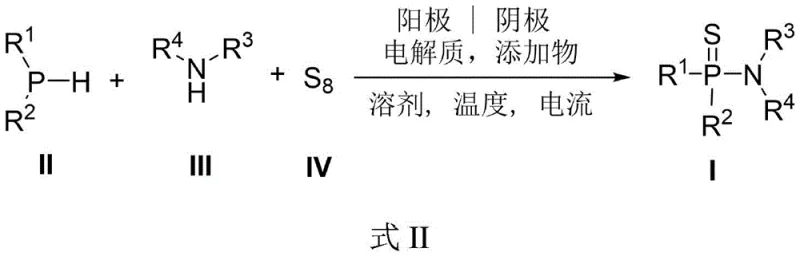
In stark contrast, the electrochemical methodology disclosed in CN116284117A offers a paradigm shift towards sustainable manufacturing. By employing an undivided cell setup with inexpensive graphite and platinum electrodes, the reaction activates the P-H bond of phosphine hydrides and facilitates the insertion of elemental sulfur without the need for external chemical oxidants. This "reagent-free" oxidation strategy inherently minimizes waste generation, as electrons serve as the primary oxidant, producing hydrogen gas as the only by-product at the cathode. The process operates under mild conditions, typically at room temperature, which preserves sensitive functional groups that would otherwise degrade under the harsh thermal or acidic conditions of conventional protocols. This innovation directly supports cost reduction in fine chemical manufacturing by simplifying downstream processing and eliminating the need for expensive catalysts or hazardous reagents, thereby creating a cleaner and more economically viable production model.
Mechanistic Insights into Electrochemical Oxidative Coupling
The core of this technology lies in the anodic oxidation mechanism that drives the formation of the P-S and P-N bonds simultaneously. In the proposed catalytic cycle, the trivalent phosphine hydride undergoes single-electron oxidation at the anode surface to generate a phosphorus-centered radical cation. This highly reactive intermediate is subsequently trapped by elemental sulfur, which acts as both a sulfur source and an electron mediator, forming a transient P-S species. Concurrently, the amine nucleophile attacks the electrophilic phosphorus center, facilitated by the presence of additives like hexafluoroisopropanol (HFIP) which enhance the acidity of the N-H bond and stabilize charged intermediates through hydrogen bonding. The use of tetrabutylammonium iodide as an electrolyte further promotes the redox mediation process, ensuring efficient electron transfer throughout the reaction medium. This intricate interplay of electrochemical parameters allows for precise control over the reaction trajectory, minimizing side reactions such as over-oxidation to phosphine oxides or homocoupling of the phosphine substrates.
From an impurity control perspective, this mechanism offers distinct advantages over traditional nucleophilic substitution routes. Since the reaction does not involve halogenated leaving groups, the final product is free from inorganic salt contaminants and chloride residues that are notoriously difficult to remove to ppm levels required for pharmaceutical applications. The high chemoselectivity of the electrochemical oxidation ensures that other oxidizable functional groups on the amine or phosphine scaffolds, such as sulfides or electron-rich aromatics, remain intact. This high fidelity in bond construction translates to a cleaner crude reaction profile, significantly reducing the burden on purification teams. For quality assurance departments, this means a more consistent impurity profile across different batches, facilitating easier regulatory filing and validation. The ability to tune the reaction potential by adjusting the current density provides an additional handle for optimizing selectivity, making this method adaptable to a wide array of structurally diverse substrates.
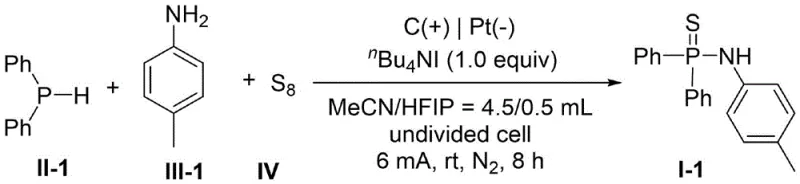
How to Synthesize P,P-Diphenyl-N-(p-tolyl)phosphinothioic amide Efficiently
To demonstrate the practical applicability of this invention, the patent details a standardized protocol for the synthesis of representative thiophosphoramide derivatives, specifically highlighting the preparation of P,P-Diphenyl-N-(p-tolyl)phosphinothioic amide. The procedure utilizes readily available starting materials and standard electrochemical equipment, making it accessible for both laboratory-scale optimization and pilot plant operations. The reaction is conducted in an undivided cell using acetonitrile as the solvent, with hexafluoroisopropanol serving as a crucial proton shuttle additive. Optimization studies indicate that a constant current of 6 mA provides the ideal balance between reaction rate and selectivity, preventing the degradation of sensitive intermediates. The molar ratio of phosphine hydride to amine is carefully controlled at 2:1 to ensure complete conversion of the limiting amine reagent while compensating for any minor oxidative losses of the phosphine component.
- Combine phosphine hydride (Compound II), amine (Compound III), elemental sulfur, electrolyte (e.g., nBu4NI), and additive (e.g., HFIP) in an organic solvent like acetonitrile within an undivided cell equipped with graphite anode and platinum cathode.
- Apply a constant direct current (e.g., 6 mA) at room temperature under nitrogen atmosphere until the amine starting material is completely consumed, typically requiring approximately 8 hours.
- Remove the organic solvent under reduced pressure and purify the crude residue via silica gel column chromatography using a petroleum ether and ethyl acetate mixture to isolate the target thiophosphoramide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this electrochemical synthesis route offers tangible strategic benefits that extend beyond mere technical novelty. The elimination of toxic and regulated precursors like phosphorus trichloride sulfide simplifies the logistics of raw material sourcing, as elemental sulfur and common amines are commodity chemicals with stable global supply chains. This shift reduces the regulatory burden associated with the storage and transport of hazardous materials, lowering insurance costs and facility compliance overheads. Furthermore, the mild reaction conditions reduce energy consumption compared to high-temperature reflux processes, contributing to a lower carbon footprint for the manufacturing site. These factors collectively enhance the long-term viability of the supply chain, mitigating risks associated with regulatory crackdowns on hazardous chemical usage.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven primarily by the simplification of the process workflow. By removing the need for expensive transition metal catalysts or specialized thio-reagents, the direct material costs are significantly lowered. Additionally, the absence of halogenated by-products means that wastewater treatment costs are drastically reduced, as there is no need for extensive dehalogenation or neutralization steps. The high yields reported, reaching up to 88%, maximize the utilization of raw materials, reducing the cost per kilogram of the active pharmaceutical ingredient (API) intermediate. This efficiency gain allows for more competitive pricing strategies in the global market while maintaining healthy profit margins.
- Enhanced Supply Chain Reliability: Reliance on niche, hazardous reagents often creates single points of failure in the supply chain. By switching to a three-component system based on abundant feedstocks, manufacturers can diversify their supplier base and reduce lead times. The robustness of the electrochemical method against variations in substrate structure means that the same production line can be easily adapted to synthesize a library of analogues without major retooling. This flexibility is crucial for responding rapidly to changing market demands or clinical trial requirements. Moreover, the operational safety of the process reduces the risk of unplanned shutdowns due to safety incidents, ensuring consistent delivery schedules for downstream customers.
- Scalability and Environmental Compliance: Scaling electrochemical reactions has historically been a challenge, but recent advancements in flow electrochemistry and electrode design have made kilogram-to-tonne production feasible. This patent's method is inherently scalable because the reaction rate is controlled by current rather than reagent addition rates, allowing for precise control even in large reactors. From an environmental standpoint, the process aligns perfectly with green chemistry principles by using electricity as a renewable energy input and generating minimal waste. This compliance with strict environmental standards future-proofs the manufacturing asset against tightening global regulations on chemical emissions, securing the license to operate for years to come.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrochemical synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in patent CN116284117A, providing clarity for stakeholders evaluating the feasibility of adopting this route for their specific projects. Understanding these nuances is essential for making informed decisions about process development and resource allocation.
Q: Why is this electrochemical method preferred over traditional phosphorus trichloride routes?
A: Traditional methods often rely on highly toxic phosphorus trichloride (PSCl3) or foul-smelling thio-reagents like Lawesson's reagent. This electrochemical approach eliminates the need for these hazardous chemicals, utilizing elemental sulfur and electricity as clean oxidants, thereby significantly improving operational safety and environmental compliance.
Q: What are the typical reaction conditions and yields for this synthesis?
A: The reaction proceeds under mild conditions, typically at room temperature with a constant current of 6 mA. It does not require transition metal catalysts or inert gas protection beyond initial setup. Yields are generally high, with specific examples demonstrating isolated yields reaching up to 88% for various substituted thiophosphoramides.
Q: Does this method support a wide range of functional groups?
A: Yes, the methodology exhibits excellent functional group tolerance. It is compatible with various substituents including alkyl, alkoxy, halogens (F, Cl, Br), trifluoromethyl groups, and heterocycles. This broad substrate scope makes it highly versatile for synthesizing diverse intermediates for drug discovery and agrochemical development.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thiophosphoramide Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the electrochemical synthesis methods described in CN116284117A for the production of high-value organophosphorus intermediates. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate these innovative laboratory protocols into robust, GMP-compliant manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from gram-scale discovery to industrial supply. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of thiophosphoramide derivative meets the highest quality standards required by the global pharmaceutical industry.
We invite you to collaborate with us to leverage this cutting-edge technology for your next drug development program. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific molecule, helping you identify the most efficient route to market. Please contact our technical procurement team today to request specific COA data for our existing catalog or to discuss route feasibility assessments for your custom synthesis needs. Let us help you secure a reliable, cost-effective, and sustainable supply of critical thiophosphoramide intermediates.
