Advanced Electrochemical Manufacturing of High-Purity Arenopyrazole Intermediates
Advanced Electrochemical Manufacturing of High-Purity Arenopyrazole Intermediates
The pharmaceutical and fine chemical industries are constantly seeking more sustainable and efficient pathways to access complex heterocyclic scaffolds, particularly those found in active pharmaceutical ingredients (APIs). A significant breakthrough in this domain is detailed in Chinese Patent CN115928114B, which discloses a novel electrochemical synthetic method for arenopyrazole compounds. This technology represents a paradigm shift from traditional thermal and stoichiometric oxidation methods to a greener, electricity-driven approach. By utilizing inexpensive ammonia as a nitrogen source and leveraging anodic oxidation to drive N-N bond formation, this process achieves high efficiency under mild conditions. For R&D directors and procurement managers alike, this patent offers a compelling alternative that addresses critical pain points regarding metal contamination, reaction safety, and overall process economics in the manufacturing of valuable indazole and pyrazole derivatives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of arenopyrazole skeletons, which serve as the core structure for drugs like Bendazac L-lysine and Axitinib, has relied on harsh and chemically demanding protocols. Traditional routes often involve the use of o-aminobenzonitrile reacting with Grignard reagents, where the Grignard species acts as a nucleophile to attack the cyano group. This approach presents severe logistical and safety challenges; Grignard reagents are notoriously moisture-sensitive, requiring strictly anhydrous conditions and specialized handling equipment, which drastically increases operational complexity. Furthermore, subsequent N-N bond formation typically necessitates transition metal catalysts, such as copper acetate, along with molecular oxygen as an oxidant at elevated temperatures around 85°C. The reliance on transition metals introduces a significant burden on downstream processing, as rigorous purification steps are mandatory to remove trace metal impurities to meet stringent pharmaceutical regulatory standards. These factors collectively result in higher production costs, increased waste generation, and potential supply chain bottlenecks due to the sensitivity of the reagents involved.
The Novel Approach
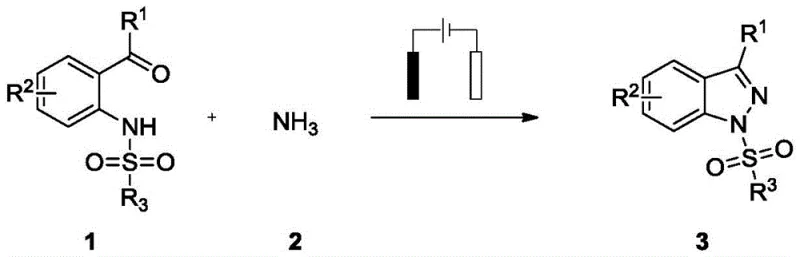
In stark contrast, the methodology described in patent CN115928114B introduces a streamlined, one-pot electrochemical strategy that circumvents these traditional hurdles. The process initiates with the in-situ condensation of a 2-sulfonylaminobenzaldehyde or ketone with small molecule ammonia to form an imine intermediate. Instead of using chemical oxidants or metal catalysts, the reaction utilizes a constant current in an undivided cell to drive the cyclization. This electrochemical N-N bond formation occurs at room temperature, eliminating the energy costs associated with heating and removing the risks linked to exothermic thermal runaways. The system employs simple iodide salts as redox mediators rather than expensive precious metals, and the only by-products generated are hydrogen gas and water, aligning perfectly with green chemistry principles. This transition from stoichiometric chemical oxidation to catalytic electrochemical oxidation not only simplifies the workflow but also fundamentally alters the cost structure of producing these high-value intermediates.
Mechanistic Insights into Electrochemical N-N Bond Formation
Understanding the mechanistic underpinnings of this transformation is crucial for R&D teams evaluating its robustness and scope. The reaction proceeds through a sophisticated interplay between electrochemical events at the electrode surface and homogeneous chemical steps in the bulk solution. Initially, the carbonyl group of the starting ketone or aldehyde reacts with ammonia to generate an imine intermediate, a step confirmed by control experiments where the isolated imine successfully converted to the product under standard conditions. The core of the innovation lies in the anodic oxidation of iodide ions (I⁻) present in the electrolyte. At the anode, iodide is oxidized to elemental iodine (I₂), which then acts as a mild electrophilic mediator in the solution phase. This electro-generated iodine interacts with the deprotonated amine nitrogen of the imine intermediate, facilitating the critical intramolecular N-N coupling. The cathode simultaneously reduces protons to evolve hydrogen gas, maintaining charge balance without introducing reducing agents that could interfere with the oxidative cyclization. This indirect electrolysis mechanism ensures that the organic substrate is not directly oxidized at the electrode surface, which often leads to decomposition, thereby enhancing the selectivity and yield of the desired arenopyrazole product.
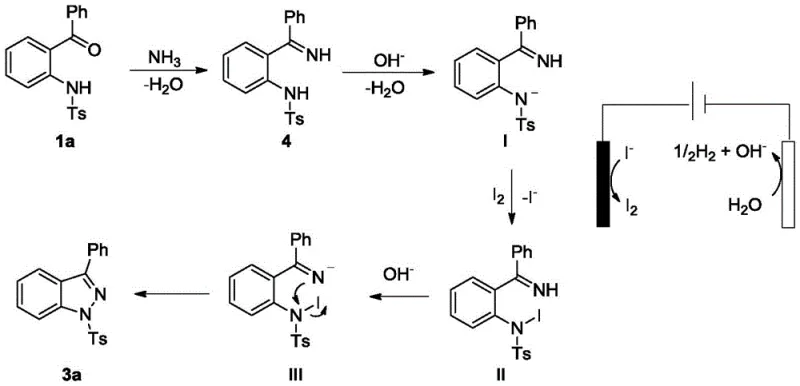
From an impurity control perspective, this mechanism offers distinct advantages over radical-based or metal-catalyzed pathways. Because the oxidation potential is carefully tuned via the iodide mediator, over-oxidation of the sensitive aromatic rings or the sulfonyl group is minimized. The absence of transition metals eliminates the risk of metal-ligand complexation side reactions that can trap intermediates or generate difficult-to-remove organometallic impurities. Furthermore, the use of ammonia as a direct nitrogen source avoids the introduction of bulky leaving groups or protecting groups that would otherwise need to be cleaved in subsequent steps, each of which carries a risk of generating degradation products. The result is a cleaner reaction profile that simplifies the isolation of high-purity intermediates, a critical factor for pharmaceutical applications where impurity profiles must be tightly controlled to ensure patient safety and regulatory compliance.
How to Synthesize Arenopyrazole Efficiently
The practical implementation of this electrochemical synthesis is designed for ease of operation, making it accessible for both laboratory-scale optimization and pilot-scale production. The standard protocol involves charging an undivided electrolytic cell with the 2-sulfonylaminobenzophenone substrate, a catalytic amount of ammonium iodide, and a stoichiometric base such as potassium hydroxide. Methanol serves as the preferred solvent, providing excellent solubility for the reagents and supporting the conductivity of the electrolyte solution. Upon addition of the ammonia source, typically a 7M solution in methanol, the reaction mixture is subjected to a constant current of 5 mA using platinum electrodes. The process runs at ambient temperature for approximately 10 hours, after which the solvent is evaporated, and the crude product is purified via column chromatography. This straightforward procedure eliminates the need for inert atmosphere gloveboxes or cryogenic cooling, significantly lowering the barrier to entry for adopting this technology in a manufacturing setting.
- Prepare the reaction mixture by combining 2-sulfonylaminobenzaldehyde/ketone substrate, ammonium iodide catalyst, and potassium hydroxide base in methanol solvent within an undivided electrolytic cell.
- Introduce ammonia source (7M in MeOH) and insert platinum electrodes (anode and cathode) into the solution, ensuring proper electrical connection.
- Apply a constant current of 5 mA at room temperature for approximately 10 hours to facilitate the N-N bond formation cyclization, followed by solvent evaporation and chromatographic purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this electrochemical technology translates into tangible strategic benefits that extend beyond mere technical novelty. The shift away from traditional chemical oxidants and transition metal catalysts directly impacts the cost of goods sold (COGS) by removing expensive reagent lines from the bill of materials. Moreover, the simplified purification process reduces the consumption of silica gel and solvents required for metal scavenging, leading to substantial cost savings in waste disposal and raw material usage. The reliance on commodity chemicals like ammonia and iodide salts ensures a stable and resilient supply chain, mitigating the risks associated with sourcing specialized or geopolitically sensitive catalysts. Additionally, the mild reaction conditions and the generation of benign by-products like water and hydrogen simplify environmental compliance and reduce the capital expenditure required for specialized waste treatment infrastructure, making this a highly attractive option for sustainable manufacturing initiatives.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts such as copper and the avoidance of stoichiometric chemical oxidants significantly lowers raw material costs. Furthermore, the removal of heavy metal scavenging steps reduces the consumption of auxiliary materials and shortens the overall processing time, leading to improved throughput and lower operational expenditures per kilogram of product produced.
- Enhanced Supply Chain Reliability: By utilizing ammonia and simple iodide salts as key reagents, the process relies on globally available commodity chemicals rather than specialized, single-source catalysts. This diversification of the supply base reduces the risk of production delays caused by reagent shortages and ensures consistent availability of critical inputs for continuous manufacturing operations.
- Scalability and Environmental Compliance: The electrochemical nature of the reaction allows for precise control over the oxidation potential, which facilitates safer scale-up compared to exothermic chemical oxidations. The generation of only hydrogen and water as by-products minimizes the environmental footprint, simplifying regulatory approvals and reducing the costs associated with hazardous waste management and disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the electrochemical synthesis of arenopyrazoles, based on the data and findings presented in the patent literature. These insights are intended to clarify the operational parameters and potential benefits for stakeholders considering the integration of this technology into their existing production workflows.
Q: What are the primary advantages of this electrochemical method over traditional Grignard-based synthesis?
A: The electrochemical method eliminates the need for unstable Grignard reagents and toxic transition metal catalysts like copper. It operates at room temperature rather than elevated temperatures (85°C), significantly improving safety profiles and reducing energy consumption while simplifying downstream purification by avoiding heavy metal residues.
Q: Is this process scalable for industrial production of pharmaceutical intermediates?
A: Yes, the process utilizes a simple undivided cell setup with commercially available electrode materials (Pt/Pt) and common solvents like methanol. The absence of sensitive reagents and the generation of only hydrogen and water as by-products make it highly suitable for scale-up in compliance with green chemistry principles.
Q: What is the role of the iodide catalyst in the reaction mechanism?
A: The iodide species acts as a redox mediator. At the anode, iodide ions are oxidized to elemental iodine, which then chemically mediates the intramolecular N-N bond formation of the imine intermediate, regenerating iodide ions to complete the catalytic cycle without being consumed stoichiometrically.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Arenopyrazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in the production of high-value pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN115928114B can be seamlessly translated into robust industrial processes. Our state-of-the-art facilities are equipped with advanced electrochemical reactors and rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of arenopyrazole intermediate delivered meets the highest quality standards required by global regulatory agencies.
We invite you to collaborate with us to leverage this green technology for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this metal-free route can optimize your manufacturing budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable and cost-effective supply of these critical building blocks.
