Advanced Catalytic Process for Manufacturing High-Purity N,N-Dicyanoethyl Arylamine Intermediates
The global demand for high-performance disperse dyes continues to drive innovation in the synthesis of key intermediates, specifically N,N-dicyanoethyl arylamine derivatives. A pivotal advancement in this domain is documented in Chinese Patent CN1166628C, which discloses a highly efficient preparation method utilizing a novel dual-component catalyst system comprising aluminum chloride (AlCl3) and zinc chloride (ZnCl2). This technological breakthrough addresses long-standing inefficiencies in the industrial production of arylamine-based dye intermediates, shifting away from harsh, copper-catalyzed protocols toward a more sustainable and economically viable pathway. By leveraging this specific catalytic synergy, manufacturers can achieve reaction conversions and yields that significantly surpass traditional benchmarks, thereby optimizing the entire value chain from raw material consumption to final product isolation. For R&D directors and process engineers, understanding the nuances of this patent is critical for modernizing production lines and securing a competitive edge in the fine chemical market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of N,N-dicyanoethyl arylamine has relied heavily on copper-based catalytic systems, typically employing cupric chloride (CuCl2) in the presence of acetic acid (HAc). These legacy processes are fraught with significant operational drawbacks that hinder cost-effective manufacturing and scalability. The most prominent limitation is the requirement for severe reaction conditions, necessitating temperatures ranging from 110°C to 140°C within high-pressure autoclaves, which imposes substantial capital expenditure on specialized equipment and increases safety risks associated with high-pressure operations. Furthermore, the catalytic efficiency of the traditional copper system is suboptimal, often resulting in reaction conversion rates and isolated yields that struggle to exceed 80%, leading to considerable waste of valuable arylamine and acrylonitrile feedstocks. The accumulation of copper residues also complicates downstream purification, often requiring extensive treatment to meet the stringent purity specifications demanded by the textile and dye industries.
The Novel Approach
In stark contrast, the methodology outlined in patent CN1166628C introduces a paradigm shift by utilizing a synergistic AlCl3-ZnCl2 catalyst system that operates under markedly milder conditions. This innovative approach enables the cyanoethylation reaction to proceed efficiently at temperatures between 60°C and 100°C, eliminating the absolute necessity for high-pressure vessels and allowing the use of standard glass-lined or stainless steel reactors. The visual representation of this transformation highlights the direct addition of two acrylonitrile molecules to the arylamine nitrogen, facilitated by the Lewis acidity of the aluminum and zinc salts.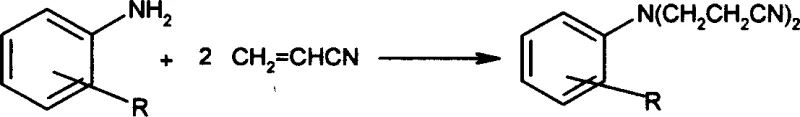 This reduction in thermal severity not only enhances process safety but also drastically lowers energy consumption per kilogram of product. Moreover, the new protocol demonstrates exceptional efficacy, with reported yields reaching between 88% and 94%, representing a substantial improvement in atom economy and resource utilization compared to the legacy copper-catalyzed routes.
This reduction in thermal severity not only enhances process safety but also drastically lowers energy consumption per kilogram of product. Moreover, the new protocol demonstrates exceptional efficacy, with reported yields reaching between 88% and 94%, representing a substantial improvement in atom economy and resource utilization compared to the legacy copper-catalyzed routes.
Mechanistic Insights into AlCl3-ZnCl2 Dual-Component Catalysis
The superior performance of the AlCl3-ZnCl2 system can be attributed to the enhanced Lewis acidity and specific coordination geometry provided by the combination of these two metal halides. In the mechanism of cyanoethylation, the catalyst functions by activating the electron-deficient double bond of the acrylonitrile molecule, making it more susceptible to nucleophilic attack by the lone pair of electrons on the arylamine nitrogen. The presence of both aluminum and zinc species likely creates a more robust electrophilic environment than copper salts alone, stabilizing the transition state and lowering the activation energy barrier for the Michael addition. This allows the reaction to initiate and propagate rapidly even at the lower temperature range of 60°C to 80°C, preventing the thermal degradation of sensitive functional groups that might occur at the higher temperatures required by conventional methods. The stoichiometry is carefully balanced, with mass ratios of arylamine to AlCl3 to ZnCl2 maintained between 1:0.1-0.5:0.05-0.2, ensuring optimal catalytic turnover without excessive metal loading that could complicate waste disposal.
From an impurity control perspective, this mechanistic pathway offers distinct advantages in minimizing side reactions such as polymerization of acrylonitrile or over-alkylation. The controlled reactivity of the dual-catalyst system ensures that the addition of the second cyanoethyl group proceeds smoothly after the first, leading to the desired N,N-disubstituted product with high selectivity. The workup procedure described in the patent further reinforces purity control; by employing vacuum distillation to recover unreacted acrylonitrile prior to aqueous workup, the process prevents hydrolysis of the nitrile groups which can occur under acidic or basic conditions if volatile reactants are not removed. Subsequent acid pickling effectively solubilizes the metal catalyst residues, allowing them to be washed away into the aqueous phase, leaving behind the organic product with purity levels consistently exceeding 98.8% and often reaching 99.4%. This level of purity is critical for downstream dye synthesis, where trace metal impurities can adversely affect color strength and hue.
How to Synthesize N,N-Dicyanoethyl Arylamine Efficiently
Implementing this patented synthesis route requires precise adherence to the specified addition sequences and thermal profiles to maximize yield and safety. The process begins with the preparation of the catalytic solution, where aluminum chloride and zinc chloride are dissolved in acrylonitrile under controlled cooling to manage the exotherm of dissolution. Once the catalyst system is homogenous, the arylamine substrate is introduced, and the mixture is agitated while maintaining the temperature within the 60°C to 100°C window for a duration of 4 to 30 hours, depending on the specific reactivity of the arylamine substituent. Following the reaction completion, the recovery of excess acrylonitrile is a critical step for both economic and environmental reasons, achieved through reduced-pressure distillation before the crude product undergoes acid-base extraction.
- Prepare the dual-component catalyst system by mixing aluminum chloride (AlCl3) and zinc chloride (ZnCl2) with acrylonitrile, ensuring full dissolution at temperatures below 80°C.
- Add the arylamine substrate to the catalyst mixture and maintain the reaction temperature between 60°C and 100°C for 4 to 30 hours under stirring.
- Recover unreacted acrylonitrile via vacuum distillation, followed by acid washing and neutralization to isolate the high-purity N,N-dicyanoethyl arylamine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this AlCl3-ZnCl2 catalytic technology translates into tangible strategic benefits that extend beyond mere chemical yield. The shift away from high-pressure, high-temperature processing fundamentally alters the cost structure of manufacturing this key dye intermediate, offering a pathway to significant cost reduction in dye intermediate manufacturing through lowered utility consumption and reduced equipment maintenance. The ability to utilize standard reactor configurations rather than specialized high-pressure autoclaves reduces capital depreciation costs and increases the flexibility of production scheduling, allowing facilities to switch between different campaigns with greater agility. Furthermore, the dramatic increase in yield from below 80% to over 90% implies a direct reduction in the volume of raw materials required per ton of finished goods, insulating the supply chain against volatility in the prices of aniline derivatives and acrylonitrile.
- Cost Reduction in Manufacturing: The elimination of high-pressure requirements and the reduction in reaction temperature from 140°C to roughly 80°C results in substantially lower energy costs for heating and cooling cycles. Additionally, the higher conversion efficiency means less raw material is wasted in side products or unreacted starting materials, directly improving the gross margin per batch. The simplified catalyst system, utilizing common industrial salts like aluminum chloride and zinc chloride, avoids the premium pricing and supply chain complexities often associated with specialized transition metal catalysts, further driving down the variable cost of production.
- Enhanced Supply Chain Reliability: By simplifying the equipment requirements to standard atmospheric or low-pressure vessels, manufacturers can reduce the lead time for high-purity dye intermediates by minimizing downtime associated with complex pressure vessel inspections and certifications. The robustness of the reaction conditions allows for more predictable batch cycles, reducing the risk of failed batches due to thermal runaway or pressure deviations. This reliability ensures a steady flow of material to downstream dye blenders, securing the continuity of supply for the textile industry even during periods of high demand.
- Scalability and Environmental Compliance: The process is inherently scalable, as the heat management requirements are less stringent than those of the exothermic high-pressure copper-catalyzed variants, facilitating the commercial scale-up of complex dye intermediates from pilot plants to multi-ton production. From an environmental standpoint, the ability to recover and recycle unreacted acrylonitrile via distillation minimizes volatile organic compound (VOC) emissions and hazardous waste generation. The use of aluminum and zinc salts, which are easier to treat in wastewater streams compared to heavy copper residues, simplifies compliance with increasingly rigorous environmental regulations regarding heavy metal discharge.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel catalytic process for N,N-dicyanoethyl arylamine. These insights are derived directly from the experimental data and claims presented in patent CN1166628C, providing a factual basis for evaluating the technology's fit within your existing manufacturing infrastructure. Understanding these details is essential for technical teams assessing the feasibility of technology transfer or licensing.
Q: What are the advantages of the AlCl3-ZnCl2 catalyst over traditional CuCl2 systems?
A: The novel AlCl3-ZnCl2 dual-component catalyst allows the reaction to proceed at significantly lower temperatures (60-100°C vs 110-140°C) and achieves superior yields (88-94% vs <80%), reducing energy costs and equipment stress.
Q: Is high-pressure equipment required for this synthesis?
A: No, unlike traditional methods that require high-pressure kettles, this patented process can operate under normal pressure or mild pressurization, simplifying equipment requirements and enhancing operational safety.
Q: How is the product purity ensured in this process?
A: High purity (up to 99.4%) is achieved through a streamlined workup procedure involving vacuum distillation to recover excess acrylonitrile, followed by precise acid pickling and washing to remove catalyst residues.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N,N-Dicyanoethyl Arylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global fine chemicals market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory patent data to industrial reality is seamless and efficient. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch against the highest industry standards. Our facility is equipped to handle the specific thermal and material handling requirements of the AlCl3-ZnCl2 catalytic process, guaranteeing a consistent supply of high-quality intermediates for your dye synthesis operations.
We invite you to collaborate with us to leverage these technological advancements for your specific application needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this optimized route for your supply chain. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities align with your quality and volume requirements, ensuring a partnership built on transparency and technical excellence.
