Advanced Catalytic Synthesis of 2-(Hydroxymethyl)cyclopropanecarboxylic Acid for Agrochemical Applications
The pharmaceutical and agrochemical industries continuously seek robust synthetic pathways for complex cyclic intermediates, and patent CN1681767A presents a significant breakthrough in the manufacturing of 2-(hydroxymethyl)cyclopropanecarboxylic acid compounds. These specific molecular scaffolds serve as critical precursors for the synthesis of chrysanthemic acid derivatives, which are foundational components in the production of pyrethroid insecticides. The disclosed technology addresses long-standing inefficiencies in prior art by introducing a direct catalytic hydrogenolysis strategy that converts protected ether precursors into the desired free alcohol functionality with exceptional efficiency. By leveraging transition metal catalysis, this method bypasses the cumbersome multi-step sequences traditionally associated with amino acid-derived syntheses, offering a streamlined route that is both economically and environmentally superior for large-scale production facilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-(hydroxymethyl)cyclopropanecarboxylic acid derivatives has been plagued by inefficient methodologies that hinder cost-effective manufacturing. Prior art, such as the methods described in Tetrahedron: Asymmetry (1995), relied heavily on glutamic acid as a chiral pool starting material, necessitating a lengthy sequence of protection, cyclization, and functional group manipulation steps. This approach not only suffers from low atom economy due to the extensive use of protecting groups but also incurs high operational costs associated with multiple isolation and purification stages. Furthermore, alternative strategies involving the acetylation and selective hydrolysis of diester compounds often struggle with regioselectivity issues, leading to mixtures of products that require resource-intensive chromatographic separation, thereby rendering these methods unsuitable for the rigorous demands of industrial agrochemical supply chains.
The Novel Approach
In stark contrast to these legacy techniques, the novel approach detailed in the patent utilizes a direct catalytic hydrogenolysis of aryl-substituted alkoxy methyl groups to unveil the target hydroxymethyl functionality. This method employs readily available 2-(substituted oxymethyl)cyclopropanecarboxylic acid esters, where the protecting group is typically a benzyl or substituted benzyl moiety, and subjects them to hydrogen donor conditions in the presence of specific transition metal catalysts. 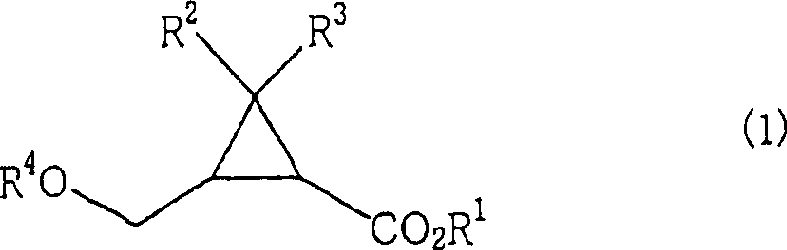 The reaction proceeds with remarkable chemoselectivity, cleaving the benzylic ether bond while leaving the sensitive cyclopropane ring and ester functionalities intact. This single-step transformation drastically simplifies the process flow, eliminating the need for harsh acidic or basic hydrolysis conditions that could compromise the structural integrity of the strained three-membered ring, thus providing a cleaner and more reliable pathway for generating high-purity intermediates.
The reaction proceeds with remarkable chemoselectivity, cleaving the benzylic ether bond while leaving the sensitive cyclopropane ring and ester functionalities intact. This single-step transformation drastically simplifies the process flow, eliminating the need for harsh acidic or basic hydrolysis conditions that could compromise the structural integrity of the strained three-membered ring, thus providing a cleaner and more reliable pathway for generating high-purity intermediates.
Mechanistic Insights into Pd/C-Catalyzed Hydrogenolysis
The core of this technological advancement lies in the precise mechanistic action of the selected catalysts, particularly palladium-based systems, which facilitate the activation of the hydrogen donor species. When using palladium on carbon (Pd/C) with hydrogen gas or formic acid, the catalyst surface adsorbs the hydrogen species, generating active hydride equivalents that attack the benzylic carbon-oxygen bond. This heterolytic or homolytic cleavage mechanism is highly specific to the activated benzylic position, ensuring that other potentially reducible groups within the molecule, such as the ester carbonyl or the cyclopropane ring itself, remain unaffected under the optimized reaction conditions. The use of heterogeneous catalysts further enhances the process by allowing for a clear phase separation post-reaction, which is critical for minimizing residual metal impurities in the final product intended for sensitive biological applications.
Furthermore, the preservation of stereochemical integrity is a paramount feature of this catalytic system, addressing a major concern for R&D directors focused on enantiomeric purity. The mild reaction temperatures, typically ranging from 0°C to 100°C, prevent the thermal equilibration or racemization of the chiral centers located on the cyclopropane ring. Whether the starting material is a pure cis-isomer, a trans-isomer, or a specific optical enantiomer, the reaction kinetics are tuned to maintain the original stereochemical configuration throughout the transformation. This fidelity ensures that the downstream synthesis of chrysanthemic acid derivatives proceeds with the expected biological activity, as the spatial arrangement of the substituents on the cyclopropane ring is strictly conserved from the precursor to the final hydroxymethyl product.
How to Synthesize 2-(Hydroxymethyl)cyclopropanecarboxylic Acid Efficiently
To implement this synthesis effectively, operators must first secure high-quality precursors where the hydroxymethyl group is masked as a stable aryl-alkyl ether, such as a benzyloxymethyl group. The process involves suspending the chosen heterogeneous catalyst, typically palladium on carbon or Raney nickel, in a compatible solvent system like methanol or ethanol, followed by the addition of the substrate.
- Prepare the reaction vessel by charging the protected 2-(substituted oxymethyl)cyclopropanecarboxylic acid ester substrate along with a heterogeneous catalyst such as palladium on carbon (Pd/C) or Raney nickel.
- Introduce the hydrogen donor system, selecting either pressurized hydrogen gas or a chemical donor like formic acid in a suitable solvent such as methanol or ethanol, ensuring thorough mixing.
- Maintain the reaction mixture at a controlled temperature between 0°C and 100°C until conversion is complete, followed by catalyst filtration and solvent removal to isolate the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalytic hydrogenolysis route offers transformative benefits regarding cost structure and supply reliability. The shift away from complex amino acid-derived starting materials to simpler olefin-based precursors significantly lowers the raw material entry cost, as the upstream synthesis of the protected cyclopropane esters is well-established and scalable. Additionally, the use of heterogeneous catalysts eliminates the need for expensive and difficult-to-remove homogeneous metal complexes, reducing both the cost of goods sold and the environmental burden associated with heavy metal waste disposal, which aligns perfectly with modern green chemistry mandates and regulatory compliance requirements.
- Cost Reduction in Manufacturing: The streamlined nature of this single-step deprotection strategy removes the necessity for multiple intermediate isolations, solvent swaps, and extensive purification columns that characterize older synthetic routes. By consolidating the transformation into a single reactor operation with simple filtration workup, manufacturers can achieve substantial reductions in labor hours, energy consumption, and solvent usage. The high yields reported, often exceeding ninety percent, further contribute to cost efficiency by maximizing the output per batch and minimizing the loss of valuable chiral materials, ultimately driving down the unit cost of the final agrochemical intermediate.
- Enhanced Supply Chain Reliability: The reliance on robust, commercially available catalysts like Pd/C and common hydrogen donors ensures that the supply chain is not vulnerable to the bottlenecks often associated with specialized reagents. Since the reaction tolerates a wide range of solvents and conditions, production facilities have the flexibility to source materials locally without compromising process performance. This resilience is crucial for maintaining continuous production schedules for key agrochemical actives, preventing delays that could ripple through the global supply chain for crop protection products, especially during peak seasonal demand periods.
- Scalability and Environmental Compliance: From a scale-up perspective, the exothermic nature of hydrogenolysis is easily managed in standard industrial reactors equipped with appropriate cooling and pressure control systems. The ability to run the reaction under atmospheric or moderate pressure simplifies the equipment requirements compared to high-pressure hydrogenation processes. Moreover, the generation of benign byproducts, such as toluene from benzyl deprotection, facilitates easier waste stream management and recycling, helping manufacturers meet stringent environmental discharge standards while maintaining high throughput capabilities for commercial tonnage production.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this patented synthesis route. These insights are derived directly from the experimental data and scope defined within the patent documentation, providing clarity on catalyst selection, stereochemical outcomes, and process parameters.
Q: What are the primary advantages of this hydrogenolysis method over traditional glutamic acid routes?
A: The patented hydrogenolysis method significantly reduces the number of synthetic steps compared to traditional routes starting from glutamic acid. It eliminates the need for complex multi-step protection and deprotection sequences, resulting in higher overall yields and simplified purification processes suitable for industrial scale-up.
Q: Does the catalytic process affect the stereochemistry of the cyclopropane ring?
A: No, the process is highly stereospecific. The catalytic hydrogenolysis conditions are mild enough to preserve the cis/trans ratio and optical purity of the starting material. If an optically active substrate is used, the resulting 2-(hydroxymethyl)cyclopropanecarboxylic acid retains its stereochemical configuration without racemization.
Q: Which catalysts are most effective for this transformation?
A: The patent highlights palladium catalysts, particularly palladium on carbon (Pd/C), as highly effective. Nickel catalysts, such as Raney nickel, are also viable alternatives. These heterogeneous catalysts allow for easy removal via filtration, minimizing metal contamination in the final agrochemical intermediate.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(Hydroxymethyl)cyclopropanecarboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your agrochemical development programs. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to full-scale manufacturing is seamless and efficient. We adhere to stringent purity specifications and utilize rigorous QC labs to verify that every batch of 2-(hydroxymethyl)cyclopropanecarboxylic acid meets the exacting standards required for downstream synthesis of pyrethroids, guaranteeing consistency and performance in your final formulations.
We invite you to collaborate with us to optimize your supply chain for these essential building blocks. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced catalytic capabilities can enhance your production efficiency and reduce overall manufacturing costs.
