Advanced Optical Resolution and Recycling Technology for High-Purity Atomoxetine Intermediates
The pharmaceutical industry continuously seeks robust methodologies for the production of chiral active pharmaceutical ingredients, particularly for central nervous system disorders where enantiomeric purity is critical. Patent CN1950326A introduces a transformative approach to the optical resolution of racemic tomoxetine, specifically targeting the isolation of the therapeutically active (R)-(-)-enantiomer, known commercially as atomoxetine. This technology addresses long-standing inefficiencies in chiral separation by integrating a high-yield crystallization protocol with a novel recycling mechanism for the unwanted stereoisomer. By leveraging a specific solvent system comprising aromatic hydrocarbons and lower alcohols, the process achieves superior optical purity while mitigating the environmental hazards associated with traditional ether-based extractions. For global procurement teams, this patent represents a pivotal shift towards more sustainable and economically viable manufacturing routes for ADHD treatments. The integration of an epimerization step further enhances the overall material efficiency, ensuring that valuable chiral precursors are not discarded as waste but are instead converted back into the production cycle. This comprehensive strategy not only improves the bottom line through yield optimization but also aligns with modern green chemistry principles demanded by regulatory bodies worldwide.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the optical resolution of racemic tomoxetine has been plagued by suboptimal yields and reliance on hazardous chemical environments. Prior art, such as the methods disclosed in European Patent No. 0 052 492, typically yielded only about 18% of the desired (R)-(-)-tomoxetine (S)-(+)-mandelate salt. This low efficiency implies that a substantial portion of the synthesized racemic material remains unrecovered in the mother liquor, representing a significant loss of raw materials and increased production costs. Furthermore, these legacy processes often necessitate the use of volatile and environmentally detrimental solvents like diethyl ether and methylene chloride, which pose serious safety risks in large-scale operations. The requirement for strictly anhydrous conditions in subsequent racemization steps, often involving dangerous reagents like butyl lithium, further complicates the operational workflow and increases the barrier to entry for safe commercial manufacturing. These factors collectively contribute to a fragmented supply chain where consistency and cost-effectiveness are frequently compromised by the inherent limitations of the chemistry employed.
The Novel Approach
In stark contrast, the methodology outlined in CN1950326A offers a streamlined and robust alternative that drastically improves both yield and operational safety. By utilizing a mixture of racemic tomoxetine, a C1-4 alcohol such as methanol, and an aromatic solvent like toluene, the new process facilitates the crystallization of the target diastereomeric salt with yields estimated at least around 35%, with experimental examples demonstrating yields exceeding 40%. This approach eliminates the need for distilling toluene under rigorous conditions and avoids the use of harmful anhydrous media, thereby simplifying the equipment requirements and reducing energy consumption. The introduction of a biphasic system option, employing aqueous base and acetic acid, provides additional flexibility for process engineers to optimize phase separation and product recovery. Moreover, the ability to recycle the unwanted (S)-(+)-enantiomer through a mild epimerization process using potassium hydroxide in dimethyl sulfoxide ensures that the overall atom economy of the synthesis is maximized. This holistic improvement in process design directly translates to enhanced supply chain stability and reduced cost of goods sold for the final API.
Mechanistic Insights into Chiral Resolution and Epimerization
The core of this technological advancement lies in the precise manipulation of solubility differences between diastereomeric salts and the controlled stereochemical inversion of the unwanted isomer. The resolution process relies on the formation of a less soluble salt between the (R)-(-)-tomoxetine and (S)-(+)-mandelic acid in a specific solvent matrix. Heating the mixture to approximately 60°C to 80°C ensures complete dissolution, creating a homogeneous phase from which the target salt can selectively crystallize upon cooling to temperatures between -5°C and 20°C. This thermal cycling is critical for excluding the (S)-(+)-tomoxetine mandelate from the crystal lattice, thereby achieving high enantiomeric excess. Following isolation, the mother liquor containing the enriched (S)-(+)-enantiomer is subjected to epimerization. This step involves heating the mixture with a base possessing a highly ionized counterion, such as potassium hydroxide, in an aprotic polar solvent like DMSO at temperatures ranging from 85°C to 90°C. Under these conditions, the chiral center adjacent to the amine functionality undergoes racemization, effectively resetting the stereochemistry and allowing the material to re-enter the resolution cycle.
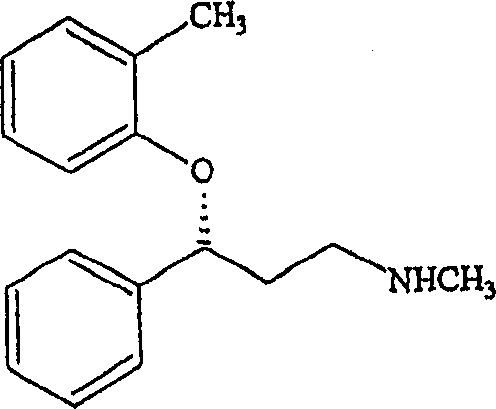
Understanding the structural nuances of the molecule is paramount for R&D directors aiming to replicate or scale this process. As illustrated in the chemical structure, the chiral center at the 3-position of the propylamine chain is the focal point of the resolution strategy. The steric and electronic interactions between the phenoxy group, the phenyl ring, and the amine moiety dictate the stability of the diastereomeric salts formed with mandelic acid. The success of the recrystallization step depends heavily on the solvent ratio; for instance, maintaining a specific ratio of toluene to methanol is essential to prevent oiling out and to promote the formation of well-defined crystals. Additionally, the conversion of the isolated mandelate salt into the final hydrochloride salt involves a straightforward acid-base extraction followed by treatment with hydrogen chloride gas. This final step ensures the removal of the resolving agent and yields the pharmaceutically acceptable salt form with high purity, ready for formulation into dosage forms for the treatment of Attention Deficit/Hyperactivity Disorder.
How to Synthesize Atomoxetine Efficiently
The synthesis of high-purity atomoxetine via this patented route requires careful attention to solvent ratios, temperature gradients, and mixing efficiencies to ensure consistent quality. The process begins with the preparation of the racemic amine, followed by the critical resolution step where the chiral integrity of the product is established. Operators must strictly adhere to the heating and cooling profiles to maximize the precipitation of the desired enantiomer while keeping the impurity in solution. Detailed standard operating procedures regarding the handling of the epimerization mother liquors are also essential to maintain the closed-loop efficiency of the plant. For a comprehensive breakdown of the specific reagent quantities, reaction times, and workup procedures validated in the patent examples, please refer to the technical guide below.
- Prepare a mixture of racemic tomoxetine, a C1-4 alcohol such as methanol, an aromatic solvent like toluene, and (S)-(+)-mandelic acid.
- Heat the mixture to approximately 60°C to 80°C to ensure complete dissolution of all solid components.
- Cool the solution to between -5°C and 20°C to induce crystallization of the desired (R)-(-)-tomoxetine (S)-(+)-mandelate salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this resolution technology offers profound strategic benefits that extend beyond simple chemical yield improvements. The shift away from hazardous solvents like diethyl ether and methylene chloride significantly reduces the regulatory burden and safety costs associated with storage, handling, and waste disposal. By utilizing common industrial solvents such as toluene and methanol, manufacturers can leverage existing infrastructure without needing specialized containment systems for highly volatile or toxic chemicals. This compatibility with standard equipment lowers the capital expenditure required for facility upgrades and accelerates the timeline for technology transfer from the lab to the pilot plant. Furthermore, the ability to recycle the unwanted enantiomer means that the effective consumption of starting materials is drastically reduced, insulating the production cost from fluctuations in raw material pricing. This material efficiency creates a more resilient supply chain capable of sustaining high-volume production runs without the bottleneck of waste accumulation.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents, combined with the recycling of the (S)-(+)-enantiomer, leads to substantial cost savings in the overall production budget. By avoiding the need for complex anhydrous conditions and dangerous alkyl lithium bases, the process reduces utility costs related to drying and inert atmosphere maintenance. The higher yield per batch means fewer batches are required to meet the same production target, effectively lowering the fixed cost allocation per kilogram of API produced. Additionally, the simplified workup procedures reduce labor hours and solvent consumption during the purification stages. These cumulative efficiencies result in a significantly lower cost of goods sold, providing a competitive edge in the generic pharmaceutical market where price sensitivity is high.
- Enhanced Supply Chain Reliability: The use of readily available and stable solvents ensures that production is not vulnerable to supply disruptions of niche or regulated chemicals. The robustness of the biphasic system allows for greater flexibility in scaling operations, as phase separation is generally easier to manage in large reactors compared to single-phase crystallizations that are sensitive to minor temperature fluctuations. The recycling loop for the unwanted enantiomer acts as a buffer, ensuring that variations in the initial resolution yield do not result in permanent material loss. This reliability is crucial for maintaining continuous supply to downstream formulation partners and meeting strict delivery schedules. Consequently, pharmaceutical companies can secure a more stable source of high-quality intermediates, reducing the risk of stockouts and production delays.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing unit operations such as crystallization, filtration, and distillation that are standard in the fine chemical industry. The avoidance of chlorinated solvents and the reduction of hazardous waste streams align perfectly with increasingly stringent environmental regulations globally. This compliance minimizes the risk of regulatory fines and shutdowns, ensuring long-term operational continuity. The greener profile of the synthesis also enhances the corporate social responsibility standing of the manufacturer, which is becoming a key criterion for selection by major multinational pharmaceutical clients. The ability to scale from kilogram to multi-ton production without fundamental changes to the chemistry ensures a smooth transition from clinical trial material to commercial supply.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this optical resolution technology. They are derived from the specific advantages and procedural details outlined in the patent documentation to assist decision-makers in evaluating the feasibility of this route for their specific manufacturing needs. Understanding these nuances is essential for conducting accurate risk assessments and cost-benefit analyses prior to technology adoption.
Q: What represents the primary advantage of this resolution method over conventional techniques?
A: The primary advantage is the significant increase in yield, achieving over 35% compared to the approximate 18% yield of older methods, alongside the elimination of hazardous solvents like diethyl ether.
Q: How is the unwanted (S)-(+)-enantiomer managed in this process?
A: The process includes an efficient epimerization step using potassium hydroxide in DMSO at 85°C to 90°C, converting the unwanted (S)-(+)-enantiomer back into racemic tomoxetine for re-processing.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the method utilizes environmentally compatible solvents like toluene and avoids strictly anhydrous conditions, making it highly scalable and safer for industrial manufacturing environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Atomoxetine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes to maintain competitiveness in the global pharmaceutical market. Our team of expert chemists has extensively evaluated the resolution and recycling protocols described in CN1950326A and possesses the technical capability to implement them at an industrial scale. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory success to manufacturing reality is seamless. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to guarantee enantiomeric excess levels exceeding 99%. We are committed to delivering high-purity atomoxetine intermediates that meet the exacting standards required for FDA and EMA filings.
We invite potential partners to engage with our technical procurement team to discuss how this optimized process can benefit your specific supply chain requirements. By collaborating with us, you gain access to a Customized Cost-Saving Analysis that quantifies the economic impact of switching to this high-yield resolution method. We encourage you to request specific COA data and route feasibility assessments to validate the quality and consistency of our output. Let us help you secure a reliable, cost-effective, and compliant source of atomoxetine intermediates for your next generation of ADHD therapeutics.
