Scalable Synthesis of Maxacalcitol Intermediates: A Technical Breakthrough for Commercial Production
Scalable Synthesis of Maxacalcitol Intermediates: A Technical Breakthrough for Commercial Production
The pharmaceutical industry continuously seeks robust manufacturing pathways for active pharmaceutical ingredients (APIs) that balance high purity with economic viability. Patent CN114728874A introduces a transformative methodology for the preparation of Maxacalcitol, a potent Vitamin D analog used extensively in the treatment of psoriasis and secondary hyperparathyroidism. This technical insight report analyzes the novel synthetic route which bypasses the historical reliance on microbial fermentation and photochemical isomerization. By leveraging a sequence of precise organic transformations including ozonolysis, Baeyer-Villiger oxidation, and Horner-Wadsworth-Emmons coupling, this process offers a streamlined approach to generating high-purity intermediates. For R&D directors and procurement strategists, understanding this shift from biological to purely chemical synthesis is critical for securing long-term supply chain stability and reducing the cost of goods sold (COGS) in dermatology and endocrinology therapeutic areas.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Vitamin D analogs like Maxacalcitol has been plagued by significant upstream dependencies and process inefficiencies. As illustrated in prior art such as US Patent No. 5,436,401, traditional routes often commence with Dehydroepiandrosterone (DHEA) obtained through microbial fermentation. This biological starting point introduces inherent variability in raw material quality and creates a supply bottleneck that is difficult to scale rapidly in response to market demand. Furthermore, these legacy processes frequently rely on photochemical irradiation and thermal isomerization to construct the secosteroid backbone. Such photochemical steps are notoriously difficult to control on an industrial scale, often resulting in complex mixtures of geometric isomers that require extensive and yield-eroding chromatographic purification. The combination of biological sourcing constraints and the technical challenges of photochemistry renders these conventional methods suboptimal for modern, cost-sensitive commercial manufacturing environments.
The Novel Approach
In stark contrast, the methodology disclosed in CN114728874A establishes a fully chemical synthetic trajectory that decouples production from biological feedstocks. The core innovation lies in the utilization of a readily accessible Vitamin D derivative (Formula 2) as the starting material, which undergoes a controlled oxidative cleavage to set the stage for side-chain modification. This approach eliminates the unpredictability of fermentation and the safety hazards associated with large-scale photochemical reactors. Instead, it employs well-understood unit operations such as low-temperature ozonolysis and nucleophilic substitutions that are easily transferred from pilot plant to commercial production scales. By restructuring the synthesis to avoid light-induced isomerization, the process inherently reduces the formation of difficult-to-remove impurities, thereby enhancing the overall efficiency and throughput of the manufacturing campaign. This strategic pivot to a purely chemical route represents a significant advancement in process chemistry for complex steroidal intermediates.
Mechanistic Insights into the Ozonolysis and HWE Coupling Strategy
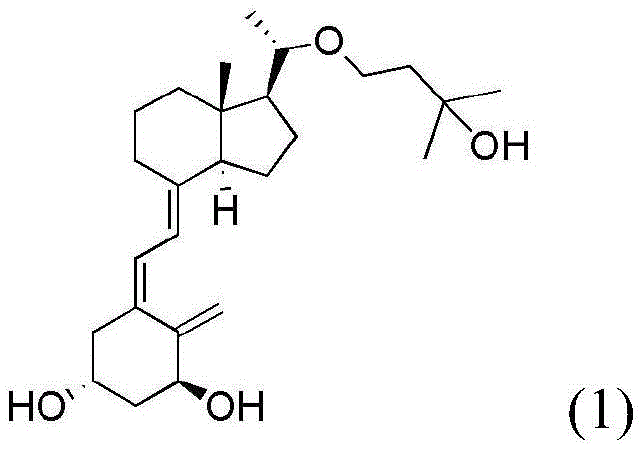
The heart of this novel synthesis is a meticulously orchestrated sequence of reactions designed to maximize stereocontrol and yield. The process initiates with the ozonolysis of the exocyclic methylene group on the starting material (Formula 2) at cryogenic temperatures ranging from -60°C to -70°C. This step is critical for cleaving the carbon-carbon double bond to generate the requisite aldehyde functionality (Formula 3) without degrading the sensitive triene system of the Vitamin D scaffold. Following the oxidative cleavage, a reductive workup using dimethyl sulfide ensures the selective formation of the carbonyl group. Subsequently, the aldehyde undergoes a Baeyer-Villiger oxidation using performic acid, which inserts an oxygen atom to form the formate ester (Formula 4). This intermediate is then subjected to a Horner-Wadsworth-Emmons (HWE) reaction with a specialized phosphonate reagent (Formula 6). The HWE coupling is performed under strictly anhydrous conditions using n-butyllithium as a base at -60°C to -50°C, ensuring the formation of the desired Z-alkene geometry in the final diene system (Formula 5) with high stereoselectivity.
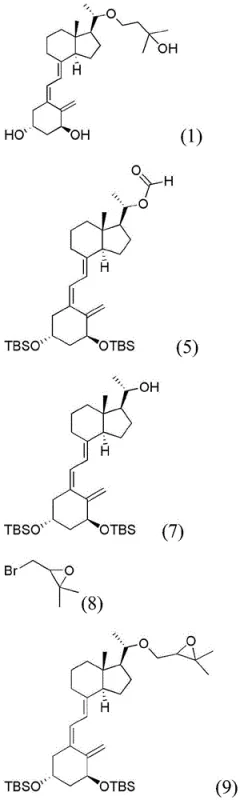
Beyond the initial backbone construction, the process demonstrates exceptional control over the side-chain installation and final deprotection. The formate group on the key intermediate (Formula 5) is hydrolyzed under mild basic conditions to reveal the alcohol (Formula 7), which then serves as a nucleophile in an alkylation reaction with a chiral epoxide (Formula 8). This step is crucial for introducing the specific hydroxy-alkyl side chain characteristic of Maxacalcitol. The subsequent ring-opening of the epoxide is achieved using lithium tri-sec-butylborohydride (L-Selectride), a bulky reducing agent chosen specifically for its ability to direct the hydride attack to the less hindered position, thereby establishing the correct stereochemistry at the new chiral center. Finally, the removal of the tert-butyldimethylsilyl (TBS) protecting groups is accomplished using tetra-n-butylammonium fluoride (TBAF). This fluoride-mediated deprotection is highly chemoselective, cleaving the silyl ethers without affecting the sensitive conjugated double bonds, ultimately delivering the target molecule (Formula 1) with the rigorous purity specifications required for pharmaceutical applications.
How to Synthesize Maxacalcitol Efficiently
Implementing this synthesis requires strict adherence to the reaction parameters outlined in the patent to ensure reproducibility and safety. The process begins with the preparation of the aldehyde intermediate via ozonolysis, where temperature control is paramount to prevent over-oxidation. Operators must monitor the saturation of ozone visually, indicated by a blue coloration of the solution, before quenching with argon and adding the reducing agent. Following the formation of the key diene intermediate (Formula 5), the downstream processing involves a series of functional group interconversions that must be managed to minimize exposure to moisture and air, particularly during the organolithium and borohydride steps. The detailed standardized synthesis steps, including specific solvent volumes, molar equivalents, and work-up procedures for each transformation from Formula 2 to Formula 1, are provided in the technical guide below to facilitate technology transfer and process validation.
- Perform ozonolysis on the starting Vitamin D derivative (Formula 2) at -60 to -70°C followed by reduction with dimethyl sulfide to obtain the aldehyde intermediate (Formula 3).
- Subject the aldehyde to Baeyer-Villiger oxidation using performic acid to generate the formate ester (Formula 4), then couple with a phosphonate via Horner-Wadsworth-Emmons reaction to form the key diene intermediate (Formula 5).
- Hydrolyze the formate group, alkylate with an epoxide side chain, reduce the epoxy group using L-Selectride, and finally deprotect the silyl groups to yield pure Maxacalcitol (Formula 1).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel chemical synthesis offers profound strategic benefits that extend beyond simple yield improvements. By eliminating the dependency on microbial fermentation for starting materials, manufacturers can secure a more reliable and consistent supply of raw inputs, insulating the production schedule from the biological variabilities and long lead times associated with fermentation batches. This shift significantly de-risks the supply chain, ensuring that production timelines are driven by chemical processing capacity rather than biological growth cycles. Furthermore, the avoidance of photochemical steps removes the need for specialized and expensive photoreactor equipment, allowing the synthesis to be conducted in standard stainless steel reactors found in most multipurpose API facilities. This compatibility with existing infrastructure drastically reduces capital expenditure requirements for scale-up and accelerates the time-to-market for generic or biosimilar versions of the drug.
- Cost Reduction in Manufacturing: The economic advantages of this route are driven by the simplification of the purification train. Traditional photochemical routes often necessitate multiple rounds of preparative HPLC or flash chromatography to separate isomers, which consumes vast quantities of silica and solvents while sacrificing yield. In contrast, the high stereoselectivity of the HWE coupling and L-Selectride reduction in this new process minimizes the generation of structural impurities. This allows for the use of simpler purification techniques such as crystallization and standard aqueous work-ups, leading to substantial reductions in solvent consumption and waste disposal costs. Additionally, the use of commodity chemicals like ozone and hydrogen peroxide, rather than exotic biological enzymes or precious metal catalysts, further optimizes the variable cost profile of the manufacturing process.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the use of commercially available or easily synthesized starting materials. Unlike the DHEA route, which ties the API supply to the agricultural and fermentation sectors, this chemical route relies on petrochemical-derived precursors that are available from a broad global supplier base. This diversification of the raw material supply base mitigates the risk of shortages and price volatility. Moreover, the robustness of the chemical steps, which tolerate a wider range of operating conditions compared to sensitive biological or photochemical processes, ensures higher batch success rates. This reliability translates directly into more predictable delivery schedules for downstream formulation partners and reduces the need for excessive safety stock inventory.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this process aligns well with green chemistry principles by improving atom economy and reducing hazardous waste. The replacement of chromatographic purifications with crystallizations significantly lowers the volume of organic solvent waste generated per kilogram of product. Furthermore, the process avoids the use of heavy metal catalysts that often require stringent removal steps to meet ICH Q3D guidelines for elemental impurities. The scalability is evidenced by the use of industrial ozone generators and standard exothermic reaction controls, making the transition from kilogram-scale development to multi-ton commercial production seamless. This ease of scale-up ensures that the manufacturing capacity can be expanded rapidly to meet surging market demand without compromising on quality or environmental compliance standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These answers are derived directly from the experimental data and beneficial effects described in patent CN114728874A, providing clarity on the operational advantages and chemical rationale behind the process. Understanding these details is essential for technical teams evaluating the feasibility of adopting this methodology for their own production lines or for procurement teams assessing the long-term viability of suppliers utilizing this technology.
Q: Why is the ozonolysis route preferred over the traditional DHEA microbial fermentation method?
A: The traditional DHEA route relies on microbial fermentation which presents significant supply chain bottlenecks and variability in starting material quality. The novel ozonolysis pathway utilizes chemically synthesized starting materials that are commercially available or easily prepared, ensuring consistent supply continuity and eliminating the biological variability associated with fermentation processes.
Q: How does the new process improve impurity control compared to photochemical methods?
A: Conventional photochemical methods often generate complex mixtures of isomers due to non-selective light irradiation, requiring difficult and costly purification steps. The new chemical synthesis route avoids photochemical isomerization entirely, relying on stereoselective reactions like L-Selectride reduction and specific HWE coupling, which drastically simplifies the impurity profile and enhances the final purity of the API intermediate.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the process is explicitly designed for scalability. It replaces laboratory-scale constraints with industrial-friendly conditions, such as the use of industrial ozone generators and standard solvent systems like dichloromethane and tetrahydrofuran. The elimination of complex chromatographic purifications in favor of crystallization and standard work-ups makes it highly viable for commercial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Maxacalcitol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our technical team has thoroughly analyzed the pathway described in CN114728874A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this complex Vitamin D analog to life. We are committed to delivering high-purity Maxacalcitol intermediates that meet stringent purity specifications, leveraging our rigorous QC labs to ensure every batch complies with international regulatory standards. Our facility is equipped to handle the specialized low-temperature reactions and sensitive organometallic steps inherent in this synthesis, guaranteeing a consistent and high-quality supply for your API manufacturing needs.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our implementation of this novel process can enhance your project's success and accelerate your time to market.
