Advanced Synthesis of Cyclopentenol Derivatives for Scalable Agrochemical Production
The pharmaceutical and agrochemical industries constantly seek robust synthetic routes for complex intermediates that balance high purity with economic viability. Patent CN1100406A, filed in 1995, presents a significant technological breakthrough in the preparation of cyclopentenol derivatives, specifically targeting compounds useful as intermediates for sterilants and pesticides. This patent discloses a novel multi-step synthesis that overcomes the limitations of prior art, particularly the low yields associated with traditional Wittig reactions. By utilizing a specialized titanium-mediated methylenation strategy on protected cyclopentenolone precursors, the invention achieves superior productivity and stereochemical control. For R&D teams and supply chain managers, understanding this chemistry is crucial for securing a reliable agrochemical intermediate supplier capable of delivering high-quality materials at scale.
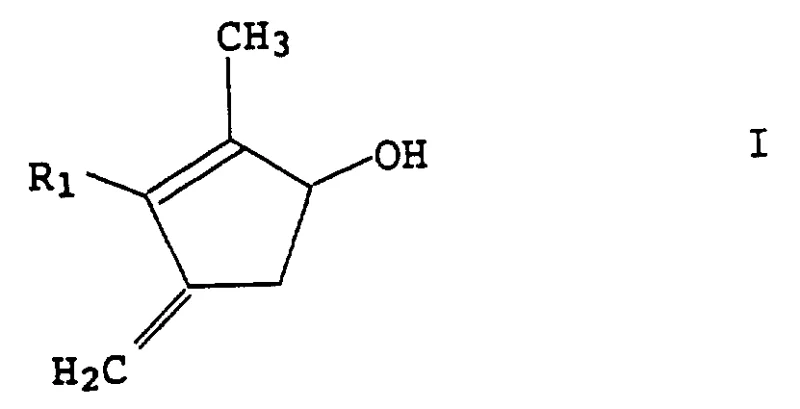
The core innovation lies in the efficient transformation of a cyclopentenolone scaffold into a methylenated cyclopentenol structure. The target molecule, represented as Compound I in the patent documentation, features a critical exocyclic methylene group and a hydroxyl functionality, structural motifs often found in bioactive natural products and synthetic fungicides. The ability to construct this specific carbon-carbon double bond with high regioselectivity and yield is a persistent challenge in organic synthesis. This patent provides a definitive solution that replaces cumbersome phosphorus-based chemistry with a more atom-economical and operationally simple protocol involving zinc, methylene halides, and titanium tetrachloride.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this invention, the standard industrial approach for synthesizing similar cyclopentenol compounds relied heavily on the Wittig reaction. As noted in the background of the patent, earlier Japanese patent publications disclosed methods using halomethyl triphenyl phosphonium salts in the presence of strong bases. While chemically feasible, these conventional methods suffer from significant drawbacks that impact commercial feasibility. The primary issue is the unsatisfactory production yield, which directly inflates the cost of goods sold (COGS) for the final active ingredient. Furthermore, the Wittig reaction generates stoichiometric amounts of triphenylphosphine oxide, a byproduct that is notoriously difficult to remove from the reaction mixture, often requiring extensive chromatography or recrystallization steps that reduce overall throughput. These inefficiencies create bottlenecks for procurement managers seeking cost reduction in agrochemical manufacturing.
The Novel Approach
The novel approach described in CN1100406A circumvents these issues by employing a reagent system comprising titanium tetrachloride, zinc, and methylene bromide or iodide. This combination generates a highly reactive carbenoid species in situ, which effectively methylenates the ketone functionality of the protected precursor. Unlike the Wittig reaction, this method does not produce heavy phosphorus waste, simplifying the workup procedure to basic filtration and extraction. The patent data demonstrates that this route can be applied to both racemic and optically active starting materials without loss of stereochemical integrity. For example, the conversion of protected ketones to the methylenated intermediates proceeds with yields often exceeding 60-80%, a substantial improvement over the roughly 40% yields observed in comparative Wittig examples. This shift represents a paradigm change in how complex cyclopentenol scaffolds are assembled for commercial scale-up.
Mechanistic Insights into Titanium-Mediated Methylenation
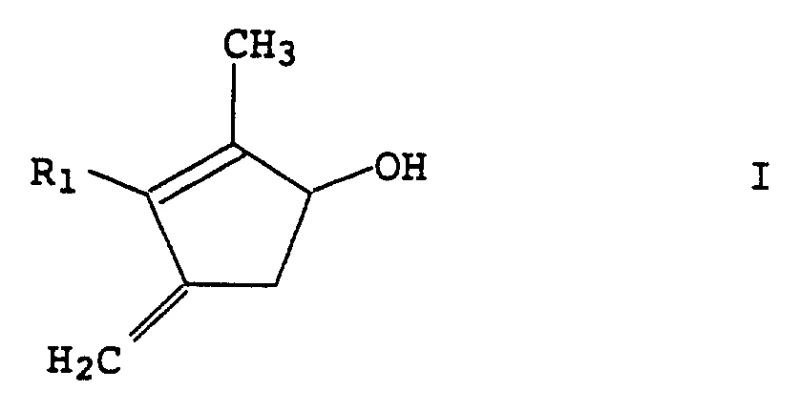
The mechanistic pathway of this synthesis is rooted in the formation of a titanium-carbene complex, often referred to as a Lombardo-type reagent. The process begins with the activation of zinc metal by titanium tetrachloride in the presence of a dihalomethane source. This interaction creates a zinc-carbenoid species coordinated to titanium, which acts as a potent nucleophile towards the carbonyl group of the substrate. The reaction is typically conducted in inert organic solvents such as tetrahydrofuran (THF) or dichloromethane at controlled temperatures ranging from -40°C to 50°C, with a preferred window of -20°C to 10°C to minimize side reactions. The mildness of these conditions is a key factor in preserving sensitive functional groups elsewhere on the molecule, such as the allylic or propargylic side chains denoted as R1 in the general formula.
Crucially, the success of this mechanism depends on the prior protection of the hydroxyl group on the cyclopentenolone ring. The patent details several effective protecting groups, including tert-butyldimethylsilyl (TBS), trimethylsilyl (TMS), and tetrahydropyranyl (THP) ethers. This protection step prevents the acidic titanium species from interacting with the free alcohol, which could lead to decomposition or polymerization. Once the methylenation is complete, forming the exocyclic double bond seen in Compound IV, the protecting group is removed under mild acidic or fluoridic conditions. This strategic use of orthogonal protection ensures that the final deprotection step yields the target cyclopentenol I with high purity and minimal impurity generation, satisfying the rigorous quality standards required for pharmaceutical and agrochemical applications.
How to Synthesize Cyclopentenol Derivatives Efficiently
The synthesis protocol outlined in the patent offers a clear roadmap for laboratory and pilot-scale production. It involves three distinct chemical transformations: protection of the starting ketone, titanium-mediated methylenation, and final deprotection. Each step has been optimized with specific reagents and conditions to maximize yield and safety. For instance, the use of imidazole as a base during silylation or p-toluenesulfonic acid for THP protection allows for precise control over reaction kinetics. The subsequent methylenation step requires careful handling of titanium tetrachloride, typically added dropwise to a suspension of zinc and methylene bromide to control exotherms. Detailed standardized synthetic steps see the guide below.
- Protect the hydroxyl group of the starting cyclopentenolone compound (II) using silyl or acetal protecting groups to form derivative III.
- Generate the active methylenating reagent by reacting zinc powder and methylene bromide with titanium tetrachloride in an inert solvent like THF or DCM.
- React the protected ketone III with the reagent mixture to form the methylenated intermediate IV, followed by deprotection to yield the final cyclopentenol I.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers tangible benefits beyond mere chemical elegance. The shift from phosphorus-based chemistry to a titanium-zinc system fundamentally alters the cost structure and operational risk profile of the manufacturing process. By eliminating the generation of triphenylphosphine oxide, the process removes a major purification bottleneck, thereby reducing solvent consumption and waste disposal costs. This efficiency translates directly into a more competitive pricing structure for the final intermediate, supporting cost reduction initiatives without compromising on quality. Furthermore, the robustness of the reaction conditions suggests a high degree of reproducibility, which is essential for maintaining consistent supply continuity.
- Cost Reduction in Manufacturing: The elimination of expensive phosphorus reagents and the associated waste treatment significantly lowers the variable costs per kilogram. The higher yields achieved in the methylenation step mean that less raw material is required to produce the same amount of product, enhancing overall material efficiency. Additionally, the simplified workup procedures reduce the demand for labor and equipment time, further driving down operational expenditures. These factors combine to create a leaner manufacturing process that is highly resilient to fluctuations in raw material pricing.
- Enhanced Supply Chain Reliability: The reagents used in this process, such as zinc powder, titanium tetrachloride, and common solvents like THF and DCM, are commodity chemicals with stable global supply chains. Unlike specialized catalysts that may face sourcing constraints, these materials are readily available from multiple vendors, reducing the risk of supply disruption. The ability to use various protecting groups also provides flexibility; if one reagent becomes scarce, the process can be adapted to use alternative protection strategies without redesigning the entire synthetic route, ensuring uninterrupted production schedules.
- Scalability and Environmental Compliance: The reaction conditions are amenable to large-scale batch processing, with temperature controls that are easily managed in standard stainless steel reactors. The absence of heavy metal catalysts like palladium or platinum simplifies the regulatory compliance landscape, as there is no need for stringent residual metal testing or complex scavenging steps. This environmental friendliness aligns with modern green chemistry principles, making the process more attractive for facilities operating under strict environmental regulations. The scalability ensures that production can be ramped up from 100 kgs to 100 MT/annual commercial production to meet growing market demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. They are derived from the specific experimental data and claims found within the patent documentation, providing clarity on the practical aspects of the technology. Understanding these details helps stakeholders assess the feasibility of integrating this intermediate into their broader supply chain.
Q: What is the primary advantage of this method over the traditional Wittig reaction?
A: The traditional Wittig reaction using halomethyl triphenyl phosphonium often results in unsatisfactory yields and difficult purification due to triphenylphosphine oxide byproducts. The patented titanium-mediated method offers significantly higher productivity and cleaner reaction profiles.
Q: Can this process maintain optical purity for chiral intermediates?
A: Yes, the process is designed to retain the original configuration of optically active starting materials. When using optically active cyclopentenolone compounds, the method produces the corresponding optically active cyclopentenol derivatives without racemization.
Q: What types of protecting groups are compatible with this synthesis?
A: The method supports a wide range of protecting groups including tri(C1-C6)alkylsilyl groups (like TBS, TMS), tetrahydropyranyl (THP), tetrahydrofuranyl, and alkoxymethyl groups, allowing flexibility based on downstream processing needs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cyclopentenol Derivative Supplier
The synthesis of cyclopentenol derivatives described in CN1100406A represents a high-value opportunity for companies developing next-generation agrochemicals. At NINGBO INNO PHARMCHEM, we possess the technical expertise to translate this patented chemistry into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves seamlessly from the bench to the plant. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of cyclopentenol intermediate meets the exacting standards required for sterilant and pesticide formulation.
We invite you to discuss how our capabilities can support your specific project needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized processes can enhance your supply chain efficiency and reduce your overall manufacturing costs.
